Introduction
The human placenta is a fetomaternal organ, developed by both fetal and maternal tissue. It plays a very essential role in growing embryo by facilitating gas and nutrient exchange between the mother and fetus (Parolini and Soncini, 2006; Parolini et al., 2008; Parolini and Caruso, 2011; Bhattacharya et al., 2011; Abdulrazzak et al., 2010). Besides this, human placenta is a possible source of progenitor or stem cells and these progenitor or stem cells are termed as placenta derived multipotent stem cells (PDMCs) (Linju Yen et al., 2005). However, studies have shown that placenta contains several times more stem cells than umbilical cord blood therefore, the use of placental stem cells can increase the chance for making stem cell from placenta for stem cell therapies in near future. PDMCs are unlikely to have the differentiation and proliferative potentials of embryonic stem cells (ESCs) though they may be more superior to adult stem cells in many aspects (Yumi et al., 2004). Furthermore, placenta is generally discarded after birth of neonate and can be available easily in great supply, secondly the recovery of placental cells from this tissue does not involve any invasive procedures and their use does not put up any ethical problems (Evangelista et al., 2008). Similarly, the accessibility, phenotypic plasticity and immunomodulatory properties of placenta-derived stem cells are useful characteristics for their use in stem cell therapy and tissue engineering (Ilancheran et al., 2009; Nazarov et al., 2012). Recently Parolini et al. (2011) have explained updates on studies on placenta derived stem cells and their potential clinical application in various diseases (Parolini et al., 2011). Many researchers are still working on placental stem cells which acquired multipotent properties that can help using them in transplantation, reprogramming to make induced pluripotent stem (iPS) cells and in therapeutic application (Ilancheran et al., 2009; Nazarov et al., 2012). Similarly, many investigators have been extensively worked on human fetal MSCs and MSCs derived from Wharton jelly. These isolated MSCs were well characterized and evaluated for their self renewal, proliferative, differentiating capacity and proved to be a promising feature for immune-modulatory and immune-regulatory capacities with a potential transplantable advantages for various disease and further clinical applications ( O’ Donoghue et al, 2006; Wang H. S., et al., 2004; Hass, R., 2011;Kim, D., 2013; Cardosa, T. C., 2012).
Multipotency of PDMCs can be evaluated well by using molecular markers. Numbers of molecular stem cell markers are being used to characterize stem cell populations derived from specific tissues along with combinations of markers which identify a particular cell type of stem cells (Potdat and Sutar, 2010; Potdar and Chaugule, 2011). Several studies have been carried out to demonstrate pluripotency of stem cells and have shown that Oct4, Nanog and Sox 2 are major genes expressed by these cells (Olamura-Nakanishi et al., 2005; Shi et al., 2006). Similarly, mesenchymal and heamatopoietic phenotypes can be confirmed by expression of CD105, CD73, CD13 (Zhang et al., 2006; Soncini et al., 2007; In’t Anker et al., 2004) and CD34, CD45 markers respectively (Catherine et al, 2009). Cytokine and chemokine play very important roles in stem cell proliferation and differentiation and also their use in cell based therapies therefore evaluation of cytokine markers like IL-6, TNFα and chemokine markers CXCR4, CXCR7, CCR5and SDF1α are studied by various investigators (Dimitriadis et al., 2005;Keelan et al., 2003). All these markers are indications of specific phenotypes of stem cells. Differential potential of placental stem cells in to cardiomyocytes and osteocytes can be confirmed by expression of cardiac β heavy chain myosin, cardiac troponin and cardiac actin and ALP and OCN markers respectively (Sabapathy et al., 2012; Sun and ji, 2012; Susman et al., 2010; Chien et al., 2006;Okamoto et al., 2007). Few studies have shown an importance of some of the genes such as CD73, CD45, CD100, Vimentin and Ki67 in maintenance of human pregnancy. However there are many lacunae to understand exact mechanism of spontaneous abortion in these women and need further investigation.
Therefore, in this study we have proposed to isolate multipotent stem cells from placenta of aborted fetus and characterized these cells by using various specific stem cell molecular markers along with the markers involved in development and implantation of fetus. Thus, this study will provide an excellent in vitro model system to study the exact mechanism of spontaneous abortion as well as if these cells found to be multipotent then these cells can be used for regenerative therapies for various disorders.
Material and Method
Isolation of Stem Cells from Human Placental Tissue
The aborted fetal placental tissue from miscarriage pregnancy and normal placental tissues specimens were sent to molecular diagnostic lab for DNA extraction which were collected in sterile 1X PBS after delivery with prior consent according to the guidelines of the ethical committee of the Jaslok Hospital and Research Centre, Mumbai, India. The part of this tissue was then used for isolation of PDMCs stem cell line. Placental tissue was initially harvested and washed thoroughly in sterile phosphate buffered saline (PBS) and processed within 1-2 hours after delivery. The washed placental tissue was then minced and enzymatically digested with 0.25% Trypsin-EDTA (HiMedia) for approximately 30 minutes at 37oC. The digested tissue pieces were explanted in 65 mm culture dishes (nunc) and were fed with Dulbecco’s modified Eagle’s medium (DMEM, HiMedia) supplemented with 10% fetal bovine serum (FBS, Invitrogen), 1% penicillin-streptomycin (HiMedia), 1μl/ml insulin (Sigma) and 2μl/ml L-glutamine (HiMedia). Explant cultures were maintained and incubated in CO2 incubator at 37oC with 5% CO2.
Explant cultures were daily observed under an inverted phase contrast microscope for the outgrowth of cells from digested tissue. After 7-8th days, many cells were seen outgrowing from tissue, adhered to the bottom of the culture dish without any substratum. Culture medium was replaced 2-3 times every week. The areas of cell colonies were located and marked and these marked cells were isolated separately by using the puck cylinder method as described in previously published research article (Potdar and Chaugule, 2012). These isolated cells readily initiated adhesion and proliferation and reached to confluence within 15 to 20 days. After confluency, these cells were passaged by trypsinization method to 65 mm culture flasks for expansion. The same procedure was repeated at every confluency and cells were cryopreserved at –850C for further experimentation.
Phase Contrast Microscopy
The morphology of cultured cells was examined under phase contrast microscope equipped with TSview soft-ware (Tucsen Imaging, Fuzhou, PR China) and capturing the images. Cells were regularly monitored and photographed for analysis.
Cell Growth Study by MTT Assay
Cell growth was assessed by monitoring the conversion of MTT (3-(4, 5-Dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide, a tetrazole) into formazan crystals. The MTT reduction is catalyzed by mitochondrial dehydrogenase enzymes and is therefore a measure for cell viability (Mosmann, 1983). Briefly, placental cells were seeded at densities of 1 X 103 cells in each well into 96 well plates (HiMedia) in triplicates for this experiment and these cells were incubated at 37oC with 5% CO2 for different time period of 24 hrs to 216 hrs. After each time point, growth medium was removed and replaced by 90 µl of fresh complete DMEM media and 10 µl of MTT solution (5 mg/ml in stock PBS) in each well (Final volume 100 µl). The plate was then incubated at 37oC for 4 hrs. Until purple formazan crystals were formed due to reduction of MTT by viable placental cells. Then media was discarded and 200 µl of Dimethyl sulfoxide (DMSO) was added to each well and mixed properly to dissolve formazan crystals. The plate was then swirled gently and absorbance was measured at 490 nm using a Microplate reader (TECAN, sunrise, India) including blank. The absorbance was plotted against a number of hrs to calculate growth rate for this cell line.
Cytogenetic Analysis
Karyotyping of human Placental Mesenchymal Stem Cells (hPMSCs) was carried out to verify the chromosomal integrity. Cytogenetic was performed according to the standard procedure (Cardosa T. C., 2012). Briefly, cell line was processed with colchicine (Himedia) at 10µg/ml for 10-14hrs for metaphases arrest. Cells that were under metaphase stage were submitted to KCl (0.075M) solution for 25 min followed by three washes in fixative (3 methanol: 1 glacial acetic acid) and then spread onto chilled slides for giemsa staining. Morphology was analyzed and chromosomes were counted.
Molecular Characterization of Placental Stem Cells
Total RNA was isolated from placental tissue (In vivo) and from cultured placental stem cells at passage3. RNA Extraction was performed using Trizol reagent (Invitrogen) according to manufactures’ instruction. Total RNA was transcribed into cDNA by using Applied Biosystems high capacity cDNA kit (Invitrogen, USA). Subsequently, cDNA obtained was stored at -20oC until it is used in PCR reaction amplification. Molecular study was carried out for specific stem cells and molecular markers. The primer sequences for CD105, CD13, CD34, CD45, LIF, Sox2, Oct4, Nanog, Keratin18, β actin, CD 44, EGFR, TNFα, IL 6, CXCR4 and the size of amplification product were described previously (Potdar and Sutar, 2010; Potdar and Chaugule, 2011) whereas, sequences and size of primers other than reported genes are indicated in the table below (Table 1). The PCR products were then separated by electrophoresis on 2% Agarose gel and photographed under UV transilluminator.
Table 1: Shows Respective Primer Sequences Used for Molecular Markers for Placental Stem Cells

Results
Development of Human Placenta Mesenchymal Stem Cell Line from Placenta of Aborted Fetus
Human placental Mesenchymal stem cell line was developed from human fetal placental tissue as per the protocol described in material and method. The tissue explants cultured in growth medium were regularly monitored under phase contrast microscope for the out – growth of the cells as shown in figure 1. The outgrowths of cells were extended and formed small colonies of cells. Single colony emerged from placental tissue explants were then isolated and cultured into 35mm culture dishes to generate stem cell line. Once these transferred cells became confluent, cells were passaged by trypsinization with 0.25% Trypsin-EDTA and transferred to 60mm culture dishes and latter in 50mm flasks for further expansion. This process was continued several times after cells reached confluency and cryopreserved at different passages at –85°C for future experimentation. Thus, this study has established fetal placental derived Mesenchymal stem cells and was designated as human Placental Mesenchymal Stem Cells (hPMSCs) line subsequent to confirmation of phenotypes by molecular markers. The morphology of these cells was observed under phase contrast microscope as shown in figure 1a, 1b, 1c and 1d.
Phase Contrast Microscopy of hPMSCs Cell Line
Morphologically, these cells were elongated epithelial like appearance with high degree of adherence to substratum without feeder layer as shown in Figure 1a and 1b and 1c. These placental stem cells also have shown the capacity of pluripotent clone formation after passage 7 as shown in figure 1d. Figure 2a and 2b showed spontaneous differential potential of placental stem cells into cardiomyocytes and adipocytes respectively. Geimsa stained placental stem cells showed general morphological features having large spindle shaped cells with large nucleus and one or two nucleoli with scanty cytoplasm as shown in figure 2c.
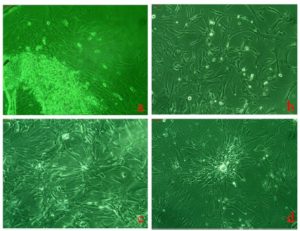
Figure 1a, 1b, 1c and 1d Show Phase Contrast Microphotographs (5X Magnification) of Isolated Human Placental Mesenchymal Stem Cells (hPMSCs). 1a and 1b Shows the Placental Cell Coming out of Explants and Form Epithelial Like Colonies, 1c Shows Confluent Placental Stem Cells and1d Shows Pluripotent Clone Formation after Passage 7.
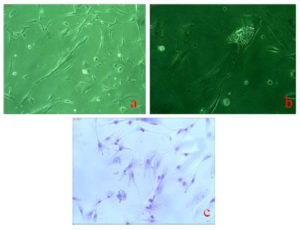
Figure 2a, 2b Show Phase Contrast Photographs (5X Magnification) of Spontaneous Differentiation of Placental Stem Cells into Cardiomyocytes and Adipocytes Respectively Whereas, 2c Shows Geimsa Stained Elongated Placental Stem Cells Having Faint Cytoplasm and Large Nucleus
Cell Proliferation Study by MTT Assay
Cell growth study by MTT assay is used to evaluate viability of cells and cell proliferation rate. The formations of resultant purple formazan crystals are directly proportional to the level of energy metabolism in the cells. The plot of MTT assay showed that these placental stem cells proliferation was increasing with the progression of days (up to 8 days) and cells were in exponential phase as shown in Figure 3.
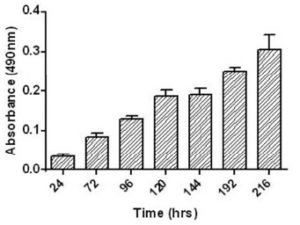
Figure 3 Shows Graph of in Vitro Placental Mesenchymal Stem Cells by MTT Assay. Placental Mesenchymal Stem Cells were Plated at Density of 1 x 103cells/ Well in Triplicate Cultures and were Harvested Every Day for 8 Days.
Cytogenetic Analysis of Human Placental Mesenchymal Stem Cell (hPMSCs) Line
Cell line was processed for cytogenetic analysis using standard chromosome preparation methods as described in Materials and method. More than 20 well spread metaphase cells were counted and found to have diploid 46 chromosomes as shown in figure 4.
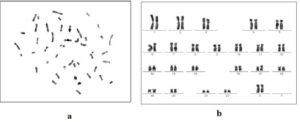
Figure 4a and 4b Show Cytogenetic Analysis of hPMSCs Cell Line by Giemsa Staining
Molecular Profiling of Human Placental Mesenchymal Stem Cell (hPMSCs) Line
Present work was undertaken to study multipotency of human placenta (In vivo) and human placental stem cells (In vitro) derived from the same tissue by using various stem cell molecular markers to evaluate their phenotypic and differentiating characteristic to confirm their mutipotent characteristics. The molecular markers defining their characteristic are defined as follow.
Selection of Positive Controls
RNA from CML (Chronic Myeloid leukemia) cells, Normal blood cells, hBCMSCs cell line (Potdar and Chaugule, 2011), hDMSCs cell line (Potdar and Prasanan, 2013) and chondrocytes cultures from articular cartilage (data was not published) developed in our laboratory were used as positive controls. CD45, CD34 were expressed in CML whereas, Oct4, Nanog, SOX2, LIF, Keratin18, CD100 and Ki 67 were expressed in normal blood cells. CD 105, CD13, CD73, CD44, CD133, Vimentin, CXCR4, CXCR7, CCR5, SDF-1α, c-Kit and IL-6 were expressed in hBCMSCs cell line. While β myosin heavy chain, cardiac troponin and cardiac actin were expressed in 5-Azacytidine treated hDMSCs cell line and ALP and OCN were expressed in chondrocytes culture cells. Confirmation of gene sequences of amplified PCR products was done by sequence analysis. The sequences obtained for each gene were confirmed from NCBI database using BLAST software [data not shown] and showed 100% match for these specific genes reported here. On the basis of this, positive controls were selected for this study.
Expression of Pluripotency and Differentiation Markers
Maintenance of pluripotency is an important characteristic of stem cells. In the present study, we have used very specific stem cell markers such as Oct 4, Nanog and SOX2 along with the differentiation makers LIF and Keratin 18 to define the pluripotency phenotypes of hPMSCs cells as well as placental tissue at in vitro and in vivo conditions respectively. Figure 5a showed that all these genes were expressed normally in placental tissue as well as in hPMSCs cell lines indicating that the cells in tissue as well as in culture are stem cells and maintain their undifferentiated phenotypes and pluripotent phenotypes at in vivo and in vitro conditions.
This study suggests that hPMSCs cells can be used for future regenerative therapies of various disorders. The expression of LIF clearly showed the undifferentiated status of these cells at in vivo and in vitro condition whereas; Keratin 18 is suggestive of their epithelial phenotypes.
Expression of Mesenchymal and Hematopoietic Stem Cell Markers
The common molecular markers used for identifying Mesenchymal and hematopoietic phenotypes are CD105, CD 73, CD 13 and CD 34, CD 45 respectively. Figure 5b showed that placental stem cells (In vitro) expressed CD 105, CD 13, CD 73 and CD 34, CD45 genes whereas, placental tissue (In vivo) also showed expression of all these markers except CD 73 which is one of the Mesenchymal stem cell marker. Overall it showed that human placental stem cells preserved both Mesenchymal and hematopoietic phenotypes at in vivo and in vitro condition. However, down regulation of CD 73 in placental tissue of aborted fetus is indicating its role in maintenance of fetus during implantation.
Expression of Additional Stem Cell Markers for Placental Cells
Figure 5cshows four additional stem cell markers expressed by the placental tissue and hPMSCs in culture. It was shown that placental tissue and hPMSCs cells strongly expressed c-Kit, SSEA 4, CD 133 and CD 44 genes showing multipotent stem cell property of human placental cells.
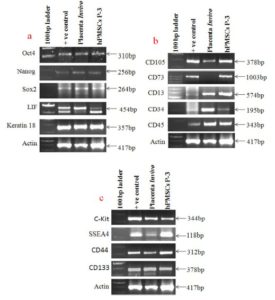
Figure 5a Shows Gene Expression of Pluripotency and Differentiation Markers, Whereas Figure 5b Shows Gene Expression Profile of Mesenchymal and Hematopoietic Stem Cells Markers in Placental Tissue Cells and in Cultured Placental Stem Cells. Down Regulation of CD73 is Clearly Observed in Aborted Placental Tissue, and Figure 5c Shows Gene Expression of Additional Stem Cell Markers in Placental Tissue Cells and Cultured Placental Stem Cells
Expression of Cytokine and Chemokine Marker
Several studies showed that cytokine and chemokine play very important role in normal tissue homeostasis. We therefore thought to study these markers in fetal placenta and hPMSCs cell line derived from the same tissue. Figure 6b showed expression of cytokines such as IL6 and TNF α in placenta and hPMSCs cell line. Both these genes normally expressed in placenta as well as in hPMSCs cell line.
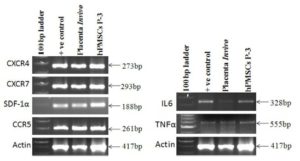
Figure 6a and 6b Show Gene Expression of Chemokine and Cytokine Markers in Placental Tissue and Cultured Placental Stem Cells
Similarly, chemokines are also an important for immunosurveillance and tissue homeostasis. Both the placental tissue and hPMSCs cells were strongly expressed all chemokines such as CXCR4, CXCR7, CCR5 and SDF-1α as shown in figure 6a.
Markers for Aborted Human Placental Cells
Several studies have shown that CD45, CD100, CD73, Ki67 and Vimentin are the specific genes involved in maintenance of pregnancy as well as responsible for the failure of the fetal immune system in aborted fetal cells (Lorenzi, et al., 2012; Kawashima, et al., 2000; Erfanian, et al. 2009; Hu, et al., 2004; Behman, et al. 1988). In the present study, we have seen expression of these genes in aborted fetal placenta and hPMSCs cells derived from the same tissue to compare their expression at in vivo and in vitro level. The present study has shown that glycoprotein CD45 is expressed in aborted fetus and hPMSCs cells whereas; CD 100 is completely down regulated in aborted fetus as well as in placental cells (figure 7a )indicating that the cells derived from aborted fetus retained this phenotype of tissue even at culture condition suggesting the use of hPMSCs cell line as an in vitro model system for understanding the mechanism of spontaneous abortions as well as being useful in developing drug discovery program for this abnormality. It was further shown that CD 73, Ki 67 and Vimentin were completely down regulated in aborted fetal tissue as shown in Figure 7a & b indicating their important role in maintenance of pregnancy in women which needs further investigation.
Similarly, we have analyzed the above genes expression in normal pregnant woman with normal pregnancy. We have investigated the expression of CD 45, CD 100, CD 73, Ki 67 and Vimentin genes in normal placenta which revealed that all above genes were normally expressed in vivo placental tissue which may proved the role of these genes in maintenance of pregnancy.
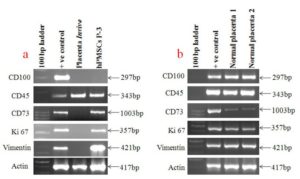
Figure 7a Shows Gene Expression of Markers in Placental Tissue and Cultured Placental Stem Cells and Figure 7b Shows the Same Markers’ Expression in Controlled Normal Pregnant Woman in Vivo Placental Tissues
Markers for Differentiated hPMSCs into Cardiomyocytes and Osteocytes
The multipotency of the placental stem cells has been reported in several studies. We have observed that some of the placental stem cells spontaneously differentiate into cardiomyocytes and osteocytes in culture condition. The spontaneous differentiation of hPMSCs cell line into cardiomyocytes cells and adipocytes was shown in figure 8a and 8b since theses cells were stained with Alizarin red and Oil red O as has been shown in figure 8a and 8b. Figure 8c showed the expression of β myosin heavy chain, cardiac troponin and cardiac actin in placenta as well as in hPMSCs cell line. Similarly, we have also checked specific markers for osteocytes differentiation. Figure 8d showed expression of alkaline phosphates (ALP) and osteocalcin (OCN) in both placenta as well as hPMSCs cell line indicating their presence in fetal placenta as well as in the cell line developed from this tissue.
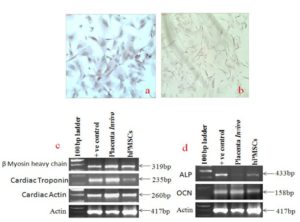
Figure 8a and 8b Show Alizarin Red and Oil red O Staining of hPMSCs Cells and Figure 8c and 8d Show Gene Expression of Cardiomyogenic and Osteogenic Markers in Placental Tissue Cells and Cultured Placental Stem Cells
Discussion
Placenta is a potent niche source of stem cells and has been studied extensively over the past few years. It has been shown that placenta can be easily available during delivery as well as from aborted fetus for stem cell studies (Parolini and Soncini, 2006; Parolini et al., 2008; Parolini and Caruso, 2011; Bhattacharya et al., 2012; Abdulrazzak et al., 2010). Recently researchers have proved that fetal and extra embryonic tissues have not only existed with multipotent stem cells but also contain pluripotent subpopulation of stem cells (O’Donoghue and Fisk, 2004; Cananzi et al., 2009). Therefore, placenta fulfills two main necessities for cell therapy; one is to obtain higher number of cells and second is the use of non-invasive procedures for their harvesting. Placental derived stem/progenitor stem cells show phenotypic plasticity, immunomodulatory properties which are useful characteristics of cell therapy and tissue engineering (Kanemura, 2010; Ilancheran et al., 2009; Nazarov et al., 2012;Drygalski and Adamson, 2000; Abdulrazzak et al., 2010). In the present study, we have taken a placenta of aborted fetus and succeeded in isolating and characterizing placental stem cells phenotypically and genotypically by using various molecular stem cell markers. It was observed that the placental stem cells showed high proliferative rate at in vitro level. This phenomenon was well correlated by formation of pluripotent clones within culture conditions while attending confluency. This cell line was designated as a human Placental Mesenchymal Stem Cell (hPMSCs) line which is presently at passage 10 and growing well in culture.
Role of Mesenchymal and Hematological Markers in Placental Stem Cells
Recent studies have shown that adult stem cells derived from tissues were expressed mesenchymal stem cell markers such as CD 105, CD 13, and CD 73 along with hematopoietic stem cell markers like CD 45 and CD 34 (Rusciucă et al., 2011; Potdar et al., 2010, 2011, 2012). In the present study, we have also studied the expression of mesenchymal and hematopoietic markers in placental and hPMSCs cells from the same placenta. It was observed that placental tissue expressed CD 105, CD 13, CD 45 and CD 34 except CD 73 whereas, hPMSCs cell line expressed all these markers. The study has shown that the CD 73 besides a Mesenchymal stem cell marker is highly regulated during pregnancy and maintain the fetal growth (Airas et al., 1997). Our finding has shown that there is a complete down regulation of CD 73 gene indicating that lack of CD73 expression may be responsible for the spontaneous fetal abortion. However, the hPMSCs cell line derived from the same fetal placenta expressed normal level of CD73 indicating that when the aborted fetal stem cells grew in cultures with growth medium, they maintain their normal phenotypes. Thus the factors such as insulin, growth factors etc. present in growth media can be useful in elevating the level of CD73 for maintenance of aborted fetus in women with high risk pregnancy which need further investigation.
Role of Pluripotency and Differentiation Markers in Placental Stem Cells
The crucial characteristic of stem cells is their pluripotency and their self-regeneration capacity which are confirmed by transcriptional factors, Oct3/4, Nanog and Sox2 (Matikainen et al., 2005; Barry et al., 2005). Similarly LIF helps in maintaining developmental potential of embryonic stem cells and protect them from differentiation (Keelan et al., 2003; Olamura-Nakanishi et al., 2005;Shi et al., 2006). In the present study, all these transcription factors such asOct4, Nanog, Sox 2 and LIF were well expressed by fetal placental cells as well as derived hPMSCs cell line indicating that these cells are stem cells and retain their stem cell nature in culture conditions and it showed that there is no effect shown on pluripotent properties of fetal stem cells in aborted placenta.
Role of Additional Stem Cell Markers in Placental Stem Cells
Placenta has been already described for having multipotent stem cells by many investigators. We therefore thought of studying some additional stem cell markers which may be useful in evaluating the multipotency of placental stem cells and their use in therapies for various disorders. Several key transcriptional factors and stem cell markers were studied including CD 133, CD 44, c-Kit, Ki 67, Vimentin and SSEA4 by various investigators (Sölder et al., 2012; Bhattacharya et al., 2012;Abdulrazzak et al., 2010; Erfanianet al., 2009;Ivaska et al., 2007; Irmaet al.,2013; Lim et al., 2012). In the present study, we have shown the expression of these markers in placental tissue as well as in hPMSCs line. Interestingly, it has been seen that placental tissue does not express vimentin which was highly expressed in hPMSCs line along with other stem cell markers as shown in Figure 6. Vimentin is a major intermittent filament protein which is shown to be responsible for implantation in human pregnancy (Magroet al., 1995; Korgunet al., 2007). Our study clearly showed that vimentin is totally down regulated in human aborted placental tissue. Thus this can be used as a marker to evaluate the miscarriage in women who have a history of spontaneous abortion.
Role of Cytokine and Chemokine Markers in Placental Stem Cells
Cytokines have been expressed in the placenta and associated fetal and maternal membranes during pregnancy and play important role in immunomodulatory functions (Keelan et al., 2003). Cytokines appear to be an important constituent of a regulatory network operating within the feto-maternal interface during normal pregnancy. Imbalances in the cytokine milieu during the time of implantation and invasion may play a causative role in disorders linked with early pregnancy failure and in trophoblastic disease (Dimitriadis et al., 2005; Keelan et al., 2003). Similarly, Chemokines are a large superfamily of structurally and functionally related cytokines with chemotactic activity induced in response to an inflammatory stimulus. Recent evidence showed the constitutive expressions of chemokines are responsible for immunosurveillance and tissue homeostasis (Dimitriadis et al., 2005). Studies on chemokines revealed that they are essential determinants of implantation and placentation by their chemoattracting actions which are critical factors at the embryo–maternal interface and by additional functions such as cell proliferation and modification of adhesion molecule expression (Dimitriadis et al., 2005). In present study, it has been shown that there is no change in the overall cytokine and chemokine expression in human aborted fetal placenta as well as hPMSCs line indicating that these factors may not be involved in the process of spontaneous abortion.
Expression of CD45 and CD100 in Fetal and hPMSCs Cell Line
Lorenzi et al., 2012 have shown that the expression of CD100 and CD45 in placental tissue plays a crucial role in the immune system during pregnancy, which has predictable factor in miscarriage. In present study, we have studied the expression of these two proteins in fetal as well as in hPMSCs cells derived from the same tissue. Our study has shown that there is an up regulation of CD45 gene and down regulation of CD100 in both tissue and hPMSCs cell line indicating that these factors may be responsible for spontaneous abortion of fetus in these women. As the status of genes expression in tissue and hPMSCs cells are similar which indicates that hPMSCs cell line can be used as an in vitro model system to study mechanism of abortion and also useful in drug discovery program for possible treatment for this disorder.
Expression of Cardiomyocytes and Osteocytes Genes in Fetal and hPMSCs Cells
Numerous studies have demonstrated the differential potential and multilineage capacity of the placental derived stem cells through in vitro characterization and differentiation experiments (Sabapathy et al., 2012; Sun and ji, 2012; Susman et al., 2010; Chien et al., 2006). Recently, it has been reported that the placental derived stem cells differentiate into osteocytes, chondrocytes, adipocytes, hepatocytes and cardiomyocytes under specific culture conditions (Sabapathy et al., 2012; Sun and ji, 2012; Susman et al., 2010; Chien et al., 2006;Okamoto et al., 2007).We have selected differentiation pathways which are more important for the future program of stem cell transplantation at our institute – one is the use of cardiomyocytes in myocardial infarction and the another one is the use of osteocytes for bone disorders and bone tissue engineering(Sharma et al., 2013 (In press); Gupta et al., 2013(In press); Potdar and S Kumar. 2013;Potdar and Prasanan, 2013). In the present study, we have shown that hPMSCs cells have a tendency to spontaneous differentiation into cardiomyocytes as well as osteocytes and further confirmed the same with molecular markers such as β myosin heavy chain, cardiac troponin and cardiac actin for cardiomyocytes and alkaline Phosphates (ALP) and osteocalcin (OCN) markers for Osteocytes differentiation indicating that the hPMSCs cells are multipotent stem cells and can be used for regenerative therapies in cardiac as well as bone disorders.
Overall, it shows that placental mesenchymal stem cells are multipotent stem cells and have the great advantages in the therapeutic program of stem cell research. Moreover, because placental stem cells can be expanded to large numbers compared with adult stem cells, they are ideal candidates for tissue engineering, cell based therapeutic applications, and for iPS generation. Besides this, the present study has clearly shown that the abnormal expression of CD 73, CD 100, CD 45, Ki 67and vimentin are involved in fetal abortion in high risk pregnancy, and hPMSCs cell line can be used as an in vitro model system to study the mechanism of fetal abortion as well as for future regenerative therapies.
Acknowledgment
We thank the management of Jaslok Hospital and Research Center, Mumbai, India for generously sanctioning and supporting financial assistance for this research Work and special thanks to O.T. Manager Dr. Khan, Jaslok hospital and research centre. We also extend thanks to Mrs. Vrushali Joshi, Navigene Genetic Science Pvt. Ltd. for helping in cytogenetic analysis.
(adsbygoogle = window.adsbygoogle || []).push({});
References
Abdulrazzak, H., Moschidou, D., Jones, G. & Guillot, P. V. (2010). “Biological Characteristics of Stem Cells from Foetal, Cord Blood and Extraembryonic Tissues,” Journal of the Royal Society Interface, 7 S689–S706.
Publisher – Google Scholar
Airas, L., Niemelä, J., Salmi, M. et al. (1997). “Differential Regulation and Function of CD73, a Glycosyl-Phosphatidylinositol–linked 70-kD Adhesion Molecule, on Lymphocytes and Endothelial Cells,” Journal of Cell Biology, 136(2) 421–431.
Publisher – Google Scholar
Barry, F. P., Murphy J. M., English, K. & Mahon B. P. (2005). “Immunogenicity of Adult Mesenchymal Stem Cells: Lessons from the Fetal Allograft,” Stem Cells and Development, 14 (3) 252–265.
Publisher – Google Scholar
Beham, A., Denk, H. & Desoye, G. (1988). “The Distribution of Intermediate Filament Proteins, Actin and Desmoplankins in Human Placental Tissue as Revealed by Polyclonal and Monoclonal Antibodies,” Placenta, 9 (5) 479-492.
Publisher – Google Scholar
Bhattacharya, N. & Stubblefield, P. (2011). Regenerative Medicine Using Pregnancy-Specific Biological Substances, Springer, New York, USA.
Publisher – Google Scholar
Cananzi, M., Atala, A. & De Coppi, P. (2009). “Stem Cells Derived from Amniotic Fluid: New Potentials in Regenerative Medicine,” Reproductive BioMedicine Online, 18 (1) 17–27.
Publisher – Google Scholar
Cardosa, T. C., Ferrari, H. F., Garcia, A. F., Novais, J. B. et al. (2012). “Isolation and Characterization of Wharton’s Jelly- Derived Multipotent Mesenchymal Stromal Cells Obtained from Bovine Umbilical Cord and Maintained in a Defined Serum Free Three Dimensional System,” BMC Biotechnology, 12 (18).
Publisher – Google Scholar
Chien, C.- C., Yen, B. L., Lee, F.- K., Lai, T.- H., Chen, Y.- C., Chan, S.- H. & Huang, H.- I. (2006). “In Vitro Differentiation of Human Placenta-Derived Multipotent Cells into Hepatocyte-Like Cells,” Stem Cells, 24 1759–1768.
Publisher – Google Scholar
Dimitriadis, E., White, C. A., Jones, R. L. & Salamonsen, L. A. (2005). “Cytokines, Chemokines and Growth Factors in Endometrium Related to Implantation,” Human Reproduction Update, 11 (6) 613–630.
Publisher – Google Scholar
Drygalski, A.V. & Adamson, J. W. (2000). “Placental/Umbilical Cord Blood (PCB) Stem Cells for Transplantation: Early Clinical Outcomes and the Status of Ex Vivo Expansion Strategies,” The Keio Journal of Medicine, 49 (4) 141-151.
Publisher – Google Scholar
Erfanian, M., Sharifi, N. & Omidi, A. (2009). “P63 and Ki-67 Expression in Trophoblastic Disease and Spontaneous Abortion,” Journal of Research in Medical Sciences 14 (6) 375-384.
Publisher – Google Scholar
Evangelista, M., Soncini, M. & Parolini, O. (2008). “Placenta-Derived Stem Cells: New Hope for Cell Therapy?,” Cytotechnology, 5833–42.
Publisher – Google Scholar
Fukuchi, Y., Nakajima, H., Sugiyama, D., Hirose, I., Kitamura, T. & Tsuji, K. (2004). “Human Placenta-Derived Cells Have Mesenchymal Stem/Progenitor Cell Potential,” Stem Cells, 22 (5) 649 – 658.
Publisher – Google Scholar
Gupta, S., Dinda, A., Potdar, P. & Mishra, N. (2013). ‘Modification of Decellularized Goat-Lung Scaffold with Chitosan/Nanohydroxyapatite Composition for Bone Tissue Engineering Application,’ Polymer Engineering and Science. (In press)
Hass, R., Kasper, C., Bohm, S. & Jacobs, R. (2011). “Different Populations and Sources of Human Mesenchymal Stem Cells (MSC): A Comparison of Adult and Neonatal Tissue Derived MSC,” Cell Communication and Signaling, 9 (12).
Publisher – Google Scholar
Hu, Z. Y., Fung, M. R., Shen, Z. F., Wang, Y. P., Wu, L. Z., Chen, L. & Li, J. C. (2004). “Relationship between Expression of Vimentin, Type IV Collagen and Fibronectin in Human Placenta and Pregnancy Induced Hypertension,” Zhonghua Fu Chan Za Zhi, 39 (9) 609-611.
Publisher – Google Scholar
Ilancheran, S., Moodley, Y. & Manuelpillai, U. (2009). “Human Fetal Membranes: A Source of Stem Cells for Tissue Regeneration and Repair?,” Placenta, (30) 2–10.
Publisher – Google Scholar
In’t Anker, P. S., Scherjon, S. A., Kleijburg-vanderKeur, C. et al. (2004). “Isolation of Mesenchymal Stem Cells of Fetal or Maternal Origin from Human Placenta,” Stem Cells, 22 1338 –1345.
Publisher – Google Scholar
Ivaska, J., Pallari, H.- M., Nevo, J. & Eriksson, J. E. (2007). “Novel Functions of Vimentin in Cell Adhesion, Migration, and Signaling,” Experimental Cell Research, 3132050–2062.
Publisher – Google Scholar
Kanemura, Y. (2010). “Development of Cell-Processing Systems for Human Stem Cells (Neural Stem Cells, Mesenchymal Stem Cells, and Ips Cells for Regenerative Medicine,” The Keio Journal of Medicine, 59 (2) 35-45.
Publisher – Google Scholar
Kawashima, Y., Nagasawa, T. & Ninomiya, H. (2000). “Contribution of Ecto 5’- Nucleotidase to the Inhibition of Platelet Aggregation by Human Endothelial Cells,” Blood, 96 (6) 2157-2162.
Publisher – Google Scholar
Keelana, J. A., Blumenstein, M., Helliwell, R. J. A., Sato, T. A., Marvin K. W. & Mitchell, M. D. (2003). “Cytokines, Prostaglandins and Parturition—A Review,” Placenta, 17 S33–S46.
Publisher – Google Scholar
Kim, D.- W., Staples, M., Shinozuka, K., Pantcheva, P., Kang, S.- D. & Borlongan, C. V. (2013). “Wharton’s Jelly Derived Mesenchymal Stem Cells: Phenotypic Characterization and Optimizing their Therapeutic Potential for Clinical Applications,” International Journal of Molecular Sciences, 14 11692-11712.
Publisher – Google Scholar
Korgun, E. T., Cayli, S., Asar, M. & Demir, R. (2007). “Distribution of Laminin, Vimentin and Desmin in the Rat Uterus during Initial Stages of Implantation,” Journal of Molecular Histology, 38(4)253-60.
Publisher – Google Scholar
Lim, M. N., Hussin, N. H., Othman, A., Umapathy, T., Baharuddin, P., Jamal, R. & Zakaria, Z. (2012). “Ex Vivo Expanded SSEA-4+ Human Limbal Stromal Cells are Multipotent and Do Not Express Other Embryonic Stem Cell Markers,” Molecular Vision, 18 1289-1300.
Publisher – Google Scholar
Lorenzi, T., Turi, A., Lorenzi, M., Paolinelli, F., Mancioli, F., Sala, L. et al. (2012). “Placental Expression of CD100, CD72 and CD45 Is Dysregulated in Human Miscarriage,” PLoS ONE 7 (5) e35232.
Publisher – Google Scholar
Magro, G. & Grasso, S. (1995). “Expression of Cytokeratins, Vimentin and Basement Membrane Components in Human Fetal Male Müllerian Duct and Perimüllerian Mesenchyme,” Acta Histochemica, 97(1)13-8.
Publisher – Google Scholar
Matikainen, T. & Laine, J. (2005). “Placenta –An Alternative Source of Stem Cells,” Toxicology and Applied Pharmacology, 207 (2) 544– 549.
Publisher – Google Scholar
Mosmann, T. (1983). “Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays,” Journal of Immunological Methods, 65 (1–2) 55–63.
Publisher – Google Scholar
Nazarov, I., Lee, J. W., Soupene, E., Etemad, S., Knapik, D. et al. (2012). “Multipotent Stromal Stem Cells from Human Placenta Demonstrate High Therapeutic Potential,” Stem Cells Translational Medicine, 1 359 –372.
Publisher – Google Scholar
O’Donoghue, K. & Chan, J. (2006). “Human Fetal Mesenchymal Stem Cells,” Current Stem Cell Research & Therapy, 1 (3) 371-386.
Publisher – Google Scholar
O’Donoghue, K. & Fisk, N. M. (2004). “Fetal Stem Cells,” Best Practice & Research Clinical Obstetrics & Gynaecology, 18 853–875.
Publisher – Google Scholar
Okamoto, K., Miyoshi, S., Toyod, M., Hida, N., Ikegami, Y., Makino, H., Nishiyama, N., Tsuji, H. et al. (2007). “Working’ Cardiomyocytes Exhibiting Plateau Action Potentials from Human Placenta-Derived Extraembryonic Mesodermal Cells,” Experimental Cell Research, 313 2550-2562.
Publisher – Google Scholar
Okumura-Nakanishi, S., Saito, M., Niwa, H. & Ishikawa, F. (2005). “Oct-3/4 and Sox2 Regulate Oct-3/4 Gene in Embryonic Stem Cells,” Journal of Biological Chemistry, 22 (7) 5307-5317.
Publisher – Google Scholar
Parolini, O., Alviano, F., Bagnara, G. P. et al. (2008). “Concise Review: Isolation and Characterization of Cells from Human Term Placenta: Outcome of the First International Workshop on Placenta Derived Stem Cells,” Stem Cells, 26 300–311.
Publisher – Google Scholar
Parolini, O. & Caruso, M. (2011). “Review: Preclinical Studies on Placenta-Derived Cells and Amniotic Membrane,” Placenta, 25 S186-S195.
Publisher – Google Scholar
Parolini, O. & Soncini, M. J. (2006). “Human Placenta: A Source of Progenitor/Stem Cells?,” Journal für Reproduktionsmedizin Endokrinologie, 3 (2) 117-126.
Publisher – Google Scholar
Potdar, P. D. & Chaugule, S. R. (2012). ‘Defining Heterogeneity of Human Non-Metastatic Breast Cancer Tumor by Identifying Individual Cell Types Using Cellular and Molecular Markers,’ Stem Cell Studies, 2 e4.
Google Scholar
Potdar, P. D. & Prasannan, P. (2013). “Differentiation of Human Mesenchymal Stem Cells into Cardiomyocytes by treatment with 5-Azacytidine: Concept for Regenerative Therapy in Myocardial Infarction,” ISRN Stem Cell, Article ID 687282, 1-9.
Publisher – Google Scholar
Potdar, P. D. & Sutar, J. P. (2010). “Establishment and Molecular Characterization of Mesenchymal Stem Cell Lines Derived From Human Visceral & Subcutaneous Adipose Tissues,” Journal of Stem Cells and Regenerative Medicine 6 (1) 1-10.
Publisher – Google Scholar
Potdar, P. & Chaugule, S. (2011). “Establishment and Molecular Characterization of Breast Cancermesenchymal Stem Cell Line Derived from Human Non-Metastasis Breast Cancer Tumor,” Stem Cell Discovery 1 (2) 21-28.
Publisher – Google Scholar
Potdar, P. & S. Kumar, K. S. (2013). “Establishment and Molecular Characterization of Human Dermal Mesenchymal Like Stem Cells Derived from Human Scalp Biopsy of Androgenic Alopecia Patient,” Stem Cell Discovery, 3 (2), 77-82.
Publisher – Google Scholar
Robin, C., Bollerot, K., Mendes, S., Haak, E., Crisan, M., Cerisoli, F., Lauw, I. et al. (2009). “Human Placenta is a Potent Hematopoietic Niche Containing Hematopoietic Stem and Progenitor Cells throughout Development,” Cell Stem Cell, 5 385–395.
Publisher – Google Scholar
Rus-ciucă, D., Soriţău, O., Suşman, S., Pop, V. I. & Mihu, C. M. (2011). “Isolation and Characterization of Chorionic Mesenchymal Stem Cells from the Placenta,” Romanian Journal of Morphology and Embryology , 52 (3) 803–808.
Publisher – Google Scholar
Sabapathy, V., Ravi, S., Srivastava, V., Srivastava, A. & Kumar, S. (2012). “Long-Term Cultured Human Term Placenta-Derived Mesenchymal Stem Cells of Maternal Origin Displays Plasticity,” Stem Cells International, 174328 11.
Publisher – Google Scholar
Sharma, C., Dinda, A., Potdar, P. & Mishra, N. (2013). ‘Fabrication of Quaternary Composite Scaffold from Silk Fibroin, Chitosan, Gelatin and Alginate and its Characterization for Bone and Skin Tissue Engineering Applications,’ Journal of Tissue Engineering and Regenerative Medicine. (In press)
Shi, W., Wang, H., Pan, G., Geng, Y., Guo, Y. & Pei, D. (2006). “Regulation of the Pluripotency Marker Rex-1 by NANOG and SOX2,” Journal of Biological Chemisty, 281 (33) 23319-25.
Publisher – Google Scholar
Sölder, E., Böckle, B. C., Nguyen, V. A., Fürhapter, C., Obexer, P. et al. (2012). “Isolation and Characterization of CD133 + CD34 + VEGFR-2 + CD45 − Fetal Endothelial Cells from Human Term Placenta,” Microvascular Research, 84 (1) 65–73.
Publisher – Google Scholar
Soncini, M., Vertua, E., Gibelli, L. et al. (2007). “Isolation and Characterization of Mesenchymal Cells from Human Fetal Membranes,” Journal of Tissue Engineering and Regenerative Medicine, 1 296 –305.
Publisher – Google Scholar
Sun, N. Z. & Ji, H. S. (2012). “In Vitro Differentiation of Osteocytes and Adipocytes from Human Placenta-Derived Cells,”Journal of International Medical Research, 40 761-767.
Publisher – Google Scholar
Şuşman, S., Soriţău, O., Rus-ciucă, D., Tomuleasa, C., Pop, V. I. & Mihu, C. M. (2010). “Placental Stem Cell Differentiation into Islets of Langerhans-Like Glucagon-Secreting Cells,” Romanian Journal of Morphology and Embryology, 51 (4) 733–738.
Publisher – Google Scholar
Virant-Klun, I., Skutella, T., Hren, M., Gruden, K., Cvjeticanin, B. et al. (2013). “Isolation of Small SSEA-4-Positive Putative Stem Cells from the Ovarian Surface Epithelium of Adult Human Ovaries by Two Different Methods,” BioMed Research International, Article ID 690415, 1-15.
Publisher – Google Scholar
Yen, B. L., Huang, H.- I., Chien, C.- C., Jui, H.- Y., Ko, B.- S. et al. (2005). “Isolation of Multipotent Cells from Human Term Placenta,” Stem Cells, 23 3–9.
Publisher – Google Scholar
Wang, H. S., Hung, S. C., Peng, S. T., Huang, C. C., Wei, H. M. et al. (2004). “Mesenchymal Stem Cells in Wharton’s Jelly of Human Umbilical Cord,” Stem Cells, 22 (1) 1330-1337.
Publisher – Google Scholar
Zhang, X., Mitsuru, A., Igura, K. et al. (2006). “Mesenchymal Progenitor Cells Derived from Chorionic Villi of Human Placenta for Cartilage Tissue Engineering,” Biochemical and Biophysical Research Communications, 340 944 –952.
Publisher – Google Scholar











