Although the progression of the disease may be slowed, it is not considered to be preventable in all patients due to the multifactorial nature of the process; however, glycemic control remains the most important goal in this aspect (American Diabetes Association, 2014,Park, Park & Baek, 2004). While all medication used in the management of neuropathy is implemented on the basis of altering pathophysiologic mechanisms of the disease, no protocols have been established regarding drug combinations in this context (Boucek, 2006). One of the most interesting aspects of this case was the remission of pain-related symptoms after starting on duloxetine and capsaicin, remaining still after suspension of both drugs, suggesting reversibility of neuronal damage even in cases as severe as ours.
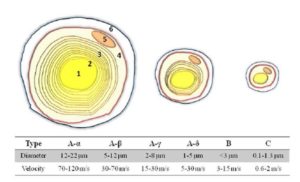
In order to assess this progression, DSP should be evaluated annually utilizing methods such as (American Diabetes Association, 2014): a) Exploration of pain sensation to pinching (mechanic pain threshold) and touch sensation to cotton swabs, both transmitted through Type C and A-ï¤ fibers; b) Vibration perception threshold of Type A-ï¢ fibers, assessed with 128 Hz tuning forks; c) Semmes-Weinstein monofilament test for the evaluation of deep protective sensation by Pacinian corpuscules; and d) Calcaneal/Patellar reflex exploration, assessing proprioceptive sensation by Golgi tendon organs. The combination of these techniques offers over 87% sensitivity for detecting DSP (American Diabetes Association, 2014). The utilization of tuning forks renders higher sensitivity and positive predictive value when compared to neurothesiometers (Kästenbauer et al, 2004). Furthermore, vibratory methods offer 53% sensitivity and 99% specificity, while superficial pain assessment through the pinching method offers 59% sensitivity and 97% specificity (Perkins et al, 2001).
Nevertheless, the Semmes-Weinstein monofilament test appears to offer the smallest margin of error, as well as require minimal training (Lee et al, 2003). This technique encompasses evaluation of 10 distinct sites in feet, with sensitivity varying between 57-93%, specificity 75-100%, positive predictive value 84-100% and negative predictive value 36-94% for the diagnosis of DSP (Freng, Schlösser & Sumpio, 2009). Moreover, different kinds of filaments yield distinct predictive results depending on the sites they are used in; for example, application of the 4.3/2-g monofilament renders remarkable diagnostic values in the hallux and the 5th metatarsal, with 60% sensitivity and 73.8% specificity (Kamei et al, 2005) with positive results being linked to alterations in vibratory perception and syncope, reflecting small-fiber damage (Perkins et al, 2001). These data have led to the proposal of limiting the sites of evaluation in order to guarantee more homogeneous results (Boucek, 2006). Particularly, the use of the 10g monofilament in 3 areas offers several advantages, including a higher risk in ulceration depth (Tan, 2010) and a lack of perception of this filament is regarded as an independent risk factor for ulceration, due to the loss of protective sensitivity (Tan, 2010).
Finally, nerve conduction assessment techniques and electromyography (Brij et al, 1996) may be implemented in the diagnosis of DSP, regarded as the most objective tests to this end (Hsu et al, 2012; Said 2007), returning documentation on the type of neuronal injury and its localization (Boulton 2005; Said 2007). In symptomatic DN, findings include longer conduction speeds due to demyelination, and lower action potentials, owing to axonal loss (Boulton 2005; Tan, 2010; Said 2007). The application of needle electromyography is reserved for assessment of abnormal spontaneous potentials and exploration of motor unit configuration, as required in cases of generalized neuropathy, atrophy of proximal muscle groups, and radiculopathy (Boulton 2005; Said 2007).
Additionally, nerve conduction studies are valuable tools in the evaluation of nerve preservation, yet they may not be applicable in all cases, such as in polyneuropathy affecting small fibers, since these do not participate in sensorial action potentials (Said 2007). In DN, axonal loss with reduction or absence of action potentials may be observed, predominating in lower limbs, although according to the stage of progression, it may involve the hands; however, many DM patients may present nerve conduction abnormalities without any clinical signs. Weisman et al. (2013) reported the predictive profile of the conduction speed of individual nerves and combinations in the diagnosis of DSP, where the conduction speed in the peroneal and the sural nerve identified cases with sensitivities of 80% and 83% respectively, and 89% and 72% specificity, correspondingly. Furthermore, the sum of the F wave of tibial latency plus peroneal conduction speed, and the sum of 3 conduction speeds of the lower limb (sural peroneal and tibial) predicted a 4-year incidence with 79-81% sensitivity and 63-77% specificity.
There are other methods for pain and quality of life assessment, such as DN4 (Douleur Neuropathique 4 Questions) for pain, which boasts 95% sensitivity and 96.6% specificity (Unal-Cevik, Sarioglu-Ay & Evcik, 2010); the LANSS Pain Scale (Leeds Assessment of Neuropathic Symptoms and Signs) with 70.2% sensitivity and 96.6% specificity (Unal-Cevik, Sarioglu-Ay & Evcik, 2010; Bennet, 2001); and the McGill Pain Questionnaire (Burckhardt & Jones, 2003). Although these methods allow for a quick screening and personal assessment of pain in correlation to clinical findings, confirmation through tuning fork assessment, monofilament test or anatomic-specific pulse and reflex exploration should always be sought.
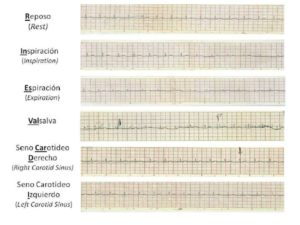
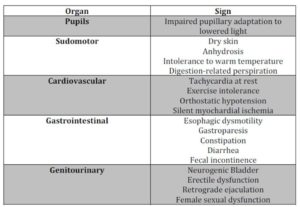
On the other hand, DAN assessment is recommended starting 5 years after diagnosis of DM1 (American Diabetes Association, 2014), surveying the following manifestations: Tachycardia at rest, exercise intolerance, orthostatic hypotension constipation, gastroparesis, erectile dysfunction, intolerance to hypoglycemia and sweating dysfunction (American Diabetes Association, 2014). The RINES-VALCARDI test (Chacín, Jatem & Rojas, 2009) is a diagnostic tool developed in Venezuela by Dr. Luis Chacín in 1981, wherein heart rate is registered with a conventional 12-lead electrocardiograph in 6 1-minute cycles: at rest, during deep inspiration, during expiration, while performing the Valsalva maneuver, and while softly massaging the left and right carotid sinuses (Figure 2). Results ≤15 points are compatible with cardiac DAN. Indeed, in comparison to non-diabetic subjects, diabetic individuals with RINES-VALCARDI scores ≤15 points presented greater frequency of postural hypotension (40%), loss of morning urinary urgency (53,3%), periodic nocturnal diarrhea (40%) and erectile dysfunction (44,4%) (Figuera et al, 1997). A new, less cumbersome version of the test has been recently proposed by the same research group (Chacín et al, 2009), which involves the utilization of a pulse oximeter for heart rate monitoring during each of the 6 cycles, registering this parameter at 0”, 15”, 30”, 45” and 60”. For each cycle, the difference between the highest and lowest values is calculated, and finally all 6 differences are added to obtain a final score. With this form of test, scores <27 points appear to detect cardiac DAN with 60.86% sensitivity, 86.61% specificity, 92.45% positive predictive value and 45.6% negative predictive value.
Regarding pharmacologic therapy of DN, a wide array of drugs with diverse mechanisms of action may be utilized, including (Attala et al, 2010): Inhibition of monoamine reuptake, calcium channel blockers N-Methyl-D-Aspartate receptor antagonists, Substance P-depleting agents, opioid agonists, GABA modulators, and NSAIDs. Indication of the drugs is issued in a progressive, stratified fashion including combinations, since monotherapy appears to offer relief to less than 50% of cases (Weisman et al, 2013). First-line medication includes (Vinik & Casellini, 2013): ï¡2-ï¤ agonists (pregabalin or gabapentin), serotonin-norepinephrine reuptake inhibitors (SNRI) (duloxetine) and tricyclic antidepressants (TCA). If pain management is inadequate or these are contraindicated, second-line treatment involves the following combinations (64): TCA with SNRI, TCA with ï¡2-ï¤ agonists or SNRI with ï¡2-ï¤ agonists. Finally, if control remains insufficient, an opioid is added into the combination. The corresponding efficacy rates and numbers needed to treat (NNT) are (Vinik, 2010): a) TCA OR 22.2 and NNT 1.5-3.5; b) Duloxetine OR 2.6 and NNT 5.7-5.8; c) Traditional Anticonvulsants OR 5.3 and NNT 2-9-4.3; and e) Opioids OR 4.3 and NNT 2.6-3.9.
According to the American Neurology Academy (Bril et al, 2011) and the Toronto Consensus Panel on Diabetic Neuropathy Management (Tesfaye et al, 2011), Level A of recommendation is Pregabalin (300-600 mg/day), whereas all other medication is considered Level B: Gabapentin 900-3600 mg/day, Duloxetine 60-120 mg/day, Amitriptyline 25-100 mg/day, Sodium Valproate 500-1200 mg/day, Tramadol 210 mg/day, and 0.075% Capsaicin cream. TCA are contraindicated in patients with glaucoma, orthostatic hypotension, overweight and cardiovascular disease. Duloxetine is contraindicated in subjects with hepatic disease, and ï¡2-ï¤ agonists contraindicated in subjects with edema and weight gain (Bril et al, 2011).
Achievement of metabolic goals accompanied by possible reversibility of neural damage was observed in these cases, where patient reeducation, psychological therapy, adequate weight gain, adherence to insulin therapy, improvement of lipid profile and inclusion of physical activity in daily routine served as key components for restitution of the functional anatomy of nerve tissue. Evidence of neuropathic damage reversibility exists in contexts such as cachectic DN (Tesfaye et al, 2011), characterized by intense neuropathic pain and wasting syndrome associated with the initiation of insulin therapy and the sudden lowering of HbA1c (Weintrob et al, 1997). This clinical picture is accompanied by finings of nervous potential ablation and slower conduction speeds in both motor and sensitive nerves (Weintrob et al, 1997; Kihara et al, 1994). Although its pathophysiology is yet to be elucidated, Grewal et al. (2006) have described the first report of objective confirmation of damage reversibility through nerve conduction studies during pain crises within the cachectic phase, suggesting this reversion to be dependent on replenishment of neuronal normoxia.
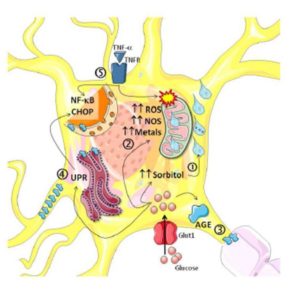
It is well-known that the application of intensive insulin therapy allows for the restoration of intraaxonal sodium ion transit patterns, in turn improving action potential capacity (Sima & Brismar, 1985). Furthermore, the reversion of hyperglycemia-triggered damage in DN may also be associated with the restoration of myoinositol metabolism defects and sodium permeability gradients (Brismar, Sima & Greene, 1987). Although several mechanisms are involved in axonal damage (Sytze Van Dam et al, 2013), these specific components share the polyol pathway as an etiopathogenic element. Williamson et al. (Williamson et al, 1993) have proposed hyperglycemia to generate the same effects as ischemia through the sorbitol pathway —termed hyperglycemic pseudohypoxia— such as an elevation of the NADH/NAD+ ratio, which induces electric, mechanic and vascular dysfunction with a consequent rise in lactate production. Moreover, it has been reported that the main mediator of oxidative neuronal damage is Aldose Reductase (AR), a key enzyme in the polyol pathway (Chung et al, 2003).
The increase of NADH due to AR and its consequent redox imbalance augment diacylglycerol synthesis and Protein Kinase C activity, leading to lowered Na+/K+ ATPase pump activity, myoinositol depletion, induction of prostaglandin synthesis, and production of reactive oxygen species and nitric oxide (Brismar, Sima & Greene, 1987). Moreover, this state of pseudohypoxia may trigger the expression of HIF-1 through nitric oxide acting on the genetic regulation of this protein (Marfella et al, 2002), enabling its participation in aberrant angiogenesis, a classic feature found in diabetic patients (Kota et al, 2012). Because AR is predominantly expressed in Schwann cells (Tomlinson & Gardiner, 2008), this aspect of neuropathic damage does not begin in neurons themselves, but rather in their myelination and support cellular system.
Considering this succession of cellular and clinical events, a possible interventional window may be outlined, wherein the main source of damage leading to conduction impairment first appears in Schwann cells (Zenker, Ziegler & Chrast, 2013); with this state of refractoriness and/or sodium gradient alteration being sufficient to modify axonal conduction patterns and “stunned” neurons, before the onset of overt clinical manifestation (Arnold et al, 2013). This phenomenon may also be observed in autonomic nerves, as reported by Kiyono et al. (2005) who found that a progressive potentiation of the polyol pathway induces sympathetic fiber dysfunction, associated with down-regulation of norepinephrine transporters, a loss of cardiac neuroautonomic control. The early detection of the neuropathic phenomenon and adequate, quick and precise control of hyperglycemia may allow for reversion of the events previously described, by rescuing neurons from stunning and impeding the development of the irreversible histopathologic changes seen in DN.
In conclusion, because DN is one of the most concerning complications — owing not only to its complex pathophysiology, but also to its booming incidence in the last decade (Mohsin et al, 2005) — early detection, strict glycemic control and management of amplifying factors are extremely valuable tools in order to delay progression and potentially favor reversibility of this disease.
Acknowledement
This work was supported by research grant Nº CC-0437-10-21-09-10 from the Technological, Humanistic, and Scientific Development Council, University of Zulia, and research grant Nº. FZ-0058-2007 from Fundacite-Zulia.
Disclosure
The authors have are no conflicts of interest to disclose.
References
1. Abd El Dayem, S. M., Battah, A. A, Soliman, R. A. (2011) Natural progression of cardiac autonomic neuropathyin patients with type 1 diabetes: a four-year follow-up study. Anadolu Kardiyol Derg, 11(3), 224-231.
Google Scholar
2. American Diabetes Association. (2014) Diagnosis and Classification of Diabetes Mellitus. Diabetes Care, 37 (Supplement 1), S81-S90.
3. Arnold, R., Kwai, N., Lin, CS., Poynten, A. M., Kiernan, M. C., Krishnan, A. V. (2013) Axonal dysfunction prior to neuropathy onset in type 1 diabetes. Diabetes Metab Res rev, 29 (1), 53-59.
Publisher – Google Scholar
4. Attala, N., Cruccu, G., Baron, R., Haanpä, M., Hansson, P., Jensen, T. S., Nurmiko, T. (2010) EFNA guidelines on pharmacology treatment of neuropathic pain: 2010 revision. Eur J Neurol, 17, 1113-1123.
Publisher – Google Scholar
5. Basic-Kes, V., Zavoreo, I., Bosnar-Puretie, M., Ivankovic, M., Birunjac, M., Govori, V., Demarin, V. (2009) Neuropathic pain. Acta Clin Croat, 48 (3), 359-365.
6. Bennet M. (2001) The LANSS pain scale: the Leeds assessment of neuropathic symptoms and signs. Pain, 92 (1-2), 147-157.
Publisher – Google Scholar
7. Blankenburg, M, Kraemer, N, Hirschfeld, G, Krumova, E. K., Maier, C, Hechler, T, Aksu, F, Magerl, W, Reinehr, T, Wiesel, T, Zernikow, B. (2012) Childhood diabetic neuropathy: functional impairment and non-invasive screening assessment. Diabet Med , 29 (11), 1425-1432.
Publisher – Google Scholar
8. Boucek P. (2006) Advanced diabetic neuropathy: a point of no return? Rev Diabetic Stud, 3 (3), 143-150.
Publisher – Google Scholar
9. Boulton, A. J. M. (2005) Managament of diabetic peripheral neuropathy. Clin Diabetes, 23 (1), 9-15.
Publisher – Google Scholar
10. Boulton, A. J. M., Vinik, A. I., Arezzo, J. C., Bril, V, Feldman, E. L., Freeman, R, Malik, R. A., Maser, R. E., Sosenko, J. M., Ziegler, D; American Diabetes Association. (2005) Diabetic Neuropathies. A statement by the American Diabetes Association. Diabetes Care, 28 (4), 956-962.
Publisher – Google Scholar
11. Brij, V., Werb, M.R., Greene, D. A., Sima, A. A. (1996) Single-fiber electromyography in diabetic peripheral polyneuropathy. Muscle Nerve, 19 (1), 2-9.
Publisher – Google Scholar
12. Bril, V., England, J., Franklin, G.M., Backonja, M., Cohen, J., Del Toro, D., Feldman, E., Iverson, D.J., Perkins, B., Russell, J.W., Zochodne, D; American Academy of Neurology; American Association of Neuromuscular and Electrodiagnostic Medicine; American Academy of Physical Medicine and Rehabilitation. (2011) Evidence-based guideline: treatment of painful diabetic neuropathy: report of the American Academy of Neurology, the American Association of Neuromuscular and Electrodiagnostic Medicine, and the American Academy of Phys¬ical Medicine and Rehabilitation. Neurology, 76 (20), 1758—1765.
Publisher – Google Scholar
13. Brismar, T., Sima, A. A, Greene, D. A. (1987) Reversible and irreversible nodal dysfunction in diabetic neuropathy.Ann Neurol, 21 (5), 504-507.
Publisher – Google Scholar
14. Burckhardt, C. S., Jones, K. D. (2003) Adult Measures of Pain: The McGill Pain Questionnaire (MPQ), Rheumatoid Arthritis Pain Scale (RAPS), Short-Form McGill Pain Questionnaire (SF-MPQ), Verbal Descriptve Scale (VDS), Visual Analog Scale (VAS), and West Haven-Yale Multidisciplinary Pain Inventory (WHYMPI). Arthritis Rheumatism, 49 (S5), S96-S104.
Publisher – Google Scholar
15. Burks, T. F., Buck, S. H., Miller, M. S. (1985) Mechanisms of depletion of substance P by capsaicin. Fed Proc, 44 (9), 2531-2534.
Google Scholar
16. Cameron, N. E. (2013) Role of endoplasmic reticulum stress in diabetic neuropathy. Diabetes, 62 (3), 696-697.
Publisher – Google Scholar
17. Casellini, C. M., Vinik, A. (2007) Clinical manifestations and current treatment options for diabetic neuropathy.Endocr Pract, 13 (5), 550-566.
Publisher – Google Scholar
18. Centers for Disease Control and Prevention, National diabetes fact sheet: national estimates and general information on diabetes and prediabetes in the United States. 2011 ed, Atlanta, GA: US Department of Health and human Services, Center for disease Control and Prevention, 2011.
Google Scholar
19. Chacín Ãlvarez, L. F., Jatem, E., Rojas, C. (2009) Diagnóstico de la neuropatía autonómica cardiovascular diabética mediante la monitorización de la frecuencia cardíaca (Rines Valcardi-Monitor). Diabetes Internacional, 1 (1), 9-14.
20. Chacín, L. F., Jatem, E, Rojas, C. (2009) Diagnóstico de la neuropatía autonómica cardiovascular diabética mediante la monitorización de la frecuencia cardíaca (Rines Valcardi-Monitor). Diabetes Int, 1 (1), 9-14.
21. Chaturvedi, N., Sjoelie, A. K., Porta, M., Aldington, S. J., Fuller, J. H., Songini, M., Kohner, E. M; EURODIAB Prospective Complications Study. (2001) Markers of insulin resistance are strong risk factors for retinopathy incidence in type 1 diabetes. Diabetes Care, 24 (2), 284— 289.
Publisher – Google Scholar
22. Chistiakov, D. A., Zotova, E. V., Savost´anov, K. V., Bursa, T. R., Galeev, I. V., Strokov, I. A., Nosikov, V. V. (2006) The 262T>C promoter polymorphism of the catalase gene is associated with diabetic neuropathy in type 1 diabetic Russian patients. Diabetes Metab, 32 (1), 63-68.
Publisher – Google Scholar
23. Chung, S. S., Ho E. C., Lam, K. S, Chung, S. K. (2003) Contribution of polyol pathway to diabetes-induced oxidative stress. J Am Soc Nephrol, 14 (8 Suppl 3), S233-S236.
Publisher – Google Scholar
24. Clayton, W, Elasy, T. A. (2009) A Review of the Pathophysiology, Classification, and Treatment of Foot Ulcers in Diabetic Patients. Clinical Diabetes, 27 (2), 52-58.
25. Eckersley, L. (2002) Role of the Schwann cell in diabetic neuropathy. Int Rev Neurobiol, 50, 293-321.
Publisher – Google Scholar
26. Edwards, J. L., Vincent, A. M., Cheng, H. T., Feldman, E. L. (2008) Diabetic neuropathy: mechanisms to management. Pharmacol Ther, 120 (1), 1-34.
Publisher – Google Scholar
27. Ewing, D. J., Clarke, B. F. (1982) Diagnosis and management of diabetic autonomic neuropathy. Br Med J, 285, 916-18.
Publisher – Google Scholar
28. Feldman, E. L. (2008) Diabetic neuropathy. Curr Drug Targets, 9 (1), 1-2.
Publisher – Google Scholar
29. Feng, Y., Schlösser, F. J., Sumpio, B. E. (2009) The Semmes Weinstein monofilament examination as a screening tool for diabetic peripheral neuropathy. J Vasc Surg, 50 (3), 675-682.
Publisher – Google Scholar
30. Figuera Ch Y. J., Tovar, E., Tineo, A. (1997) Neuropatía autonómica cardiovascular en pacientes diabéticos no insulino dependientes. Saber, 9 (1), 25-31.
Google Scholar
31. Fujii, H., Kono, K., Nakai, K., Goto, S., Komaba, H., Hamada, Y., Shinohara, M., Kitazawa, R., Kitazawa, S., Fukagawa, M. (2010) Oxidative and nitrosative stress and progression of diabetic nephropathy in type 2 diabetes. Am J Nephrol, 31 (4), 342-352.
Publisher – Google Scholar
32. González-Clemente, J. M., Mauricio, D., Richart, C., Broch, M., Caixà s, A., Megia, A., Giménez-Palop, O., Simón, I., Martínez-Riquelme, A., Giménez-Pérez, G., Vendrell, J. (2005) Diabetic neuropathy is associated with activation of the TNF-alpha system in subjects with type 1 diabetes mellitus. Clin Endocrinol, 63 (5), 525-529.
Publisher – Google Scholar
33. Govindaraian, V. S. (1986) Capsicum–production, technology, chemistry, and quality. Chemistry of the color, aroma, and pungency stimuli. Crit Rev Food Sci Nutr, 24 (3), 245-355.
Publisher – Google Scholar
34. Grewal, J., Bril, V., Lewis, G., Perkins, B. A. (2006) Objective evidence for the reversibility of nerve injury in diabetic neuropathic cachexia. Diabetes Care, 29 (2), 472-473.
Publisher – Google Scholar
35. Heesom, A. E., Millward, A, Demaine, A. G. (1998) Susceptibility to diabetic neuropathy in patients with insulin dependent diabetes mellitus is associated with a polymorphism at the 5´ end of the aldose reductase gene. J Neurol Neurosug Psychiatry, 64 (2), 213-216.
Publisher – Google Scholar
36. Hilz, M. J., Marthol, H, Neundörfer, B. (2000) Diabetic somatic polyneuropathy. Pathogenesis, clinical manifestations and therapeutics concepts. Fortschr Neurol Psychiatr, 68 (6), 278-288.
Publisher – Google Scholar
37. Hökfelt, T., Pernow, B., Wahren, J. (2001) Substance P: a pioneer amongst neuropeptides. J Intern Med, 249 (1), 27-20.
Publisher – Google Scholar
38. Hovnik, T, Dolzan, V, Bratina, N. U., Podkrajsek, K. T., Battelino, T. (2009) Genetic polymorphisms in henes encoding antioxidant enzymes are associated with diabetic retinopathy in type 1 diabetes. Diabetes Care, 32 (12), 2258-2262.
Publisher – Google Scholar
39. Hsu, W.C., Chiu, S. Y., Yen, A. M., Chen, L. S., Fann, C. Y., Liao, C. S., Chen, H. H. (2012) Somatic neuropathy is an independent predictor of all- and diabetes-related mortality in type 2 diabetic patients: a population based 5 year follow up study (KCIS). Eur J Neurol, 19 (9), 1192-1198.
Publisher – Google Scholar
40. Imme, D. C., McClesky, E. W. (2001) Lactate enhances the acid sensing Na+ channel on ischemia-sensing neurons. Nat Neurosci, 4 (9), 869-870.
Publisher – Google Scholar
41. Jennings, P. E., Nightingale, S., Lawson, N., Hoffman, P., Williamson, J.R, Bernett, A. H. (1990) Impact of microangiopathy on chronic symptomatic peripheral neuropathy. Diabetes Res, 13 (2), 51-54.
Google Scholar
42. Kamei, N., Yamane, K., Nakanishi, S., Yamashita, Y., Tamura, T., Ohshita, K., Watanabe, H., Fujikawa, R., Okubo, M., Kohno, N. (2005) Effectiveness of Semmes-Weinstein monofilament examination of diabetic peripheral neuropathy screening. J Diabetes Complications, 19 (1), 47-53.
Publisher – Google Scholar
43. Kamenov, Z. A., Traykov, L.D. (2012) Diabetic autonomic neuropathy. Adv Exp Med Biol , 771, 176-193.
44. Karayannis, G, Giamouzis, G, Cokkinos, D.V., Skoularigis, J, Triposkiadis, F. (2012) Diabetic cardiovascular autonomic neuropathy: clinical implications. Expert Rev Cardiovasc Therapy, 10 (6), 747-765.
Publisher – Google Scholar
45. Kästenbauer, T, Sauseng, S, Brath, H, Abrahamian, H, Irsigler, K (2004) The value of the Rydel-Seiffer tuning fork as a predictor of diabetic polyneuropathy compared with the neurothensiometer. Diabet Med, 21 (6), 563-567.
Publisher – Google Scholar
46. Kihara, M., Zollman, P. J., Smithson, I. L., Lagerlund, T. D., Low, P. A. (1994) Hypoxic effect of exogenous insulin on normal and diabetic peripheral nerve. Am J Physiol, 266 (6 Pt 1), E980— E985.
Google Scholar
47. Kiyono, Y., Kajiyama, S., Fujiwara, H., Kanegawa, N., Saji, H. (2005) Influence of the polyol pathway on norepinephrine transporter reduction in diabetic cardiac sympathetic nerves: implications for heterogeneous accumulation of MIBG. Eur J Nucl Med Mol Imaging, 32 (8), 993-997.
Publisher – Google Scholar
48. Kota, S. K., Meher, L. K., Jammula, S., Kota, S. K., Krishna, S.V., Modi, K. D. (2012) Aberrant angiogenesis: the gateway to diabetic complications. Indian J Endocrinol Metab, 16 (6), 918-930.
Google Scholar
49. Kuehl, M, Stevens, M. J. (2012) Cardiovascular autonomic neuropathies as complications of diabetes mellitus. Nat Rev Endocrinol , 8(7), 405-416.
Publisher – Google Scholar
50. Kunde, D. A., Crawford, A., Geraghty, D. P. (2013) Tachykinin (NK1, NK2, and NK3) receptor; transient receptor potential vanilloid 1 (TRPV1) and early transcription factor, cFOS, mRNA expression in rat tissues following systemic capsaicin treatment. Regul Pept 183C, 35-41.
Publisher – Google Scholar
51. Lee, S, Kim, H, Choi, S, et al. (2003) Clinical usefulness of the two-site Semmes-Weinstein monofilament test for detecting diabetic peripheral neuropathy. J Korean Med Sci, 18 (1), 103-107.
Publisher – Google Scholar
52. Linley, J. E., Ooi, L., Pettinger, L., Kirton, H., Boyle, J. P., Peers, C., Gamper, N. (2012)
Reactive oxygen species are second messengers of neurokinin signaling in peripheral sensory neurons. Proc Natl Acad Sci U S A, 109 (24), E1578-E1586.
Publisher – Google Scholar
53. Lupachyk, S., Watcho, P., Hasanova, N., Julius, U., Obrosova, I. G. (2012) Triglyceride, NEFA and prediabetic neuropathy: role for oxidative-nitrosative stress. Free Radic Biol Med, 52 (8), 1255-1263.
Publisher – Google Scholar
54. Manzano, G. M., Giuliano, L. P., Nóbrega, J. A. (2008)
A brief historical note on the classification of nerve fibers. Arq NeuroPsiquiatr; 66 (1), 117-119.
Publisher – Google Scholar
55. Marfella, R., D’Amico, M., Di Filippo, C., Piegari, E., Nappo, F., Esposito, K., Berrino, L., Rossi, F., Giugliano, D. (2002) Myocardial infarction in diabetic rats: role of hyperglycaemia on infarct size and early expression of hypoxia-inducible factor 1. Diabetologia. 45 (8), 1172-1181.
Publisher – Google Scholar
56. Marvizón, J. C., Wang, X., Matsuka, Y., Neubert, J. K., Spigelman, I. (2003)
Relationship between capsaicin-evoke Substance P release and neurokinin 1 receptor internalization in the rat spinal cord. Neuroscience, 118 (2), 535-545.
Publisher – Google Scholar
57. Massin, M. M., Derkenne, B., Tallsund, M., Rocour-Brumioul, D., Ernould, C., Lebrethon, M.C., Bourguignon, J.P. (1999) Cardiac autonomic dysfunction in diabetic children. Diabetes Care, 22 (11), 1845-1850.
Publisher – Google Scholar
58. Mohsin, F., Craig, M. E., Cusumano, J., Chan, A. K., Hing, S., Lee, J. W., Silink, M., Howard, N. J., Donaghue, K. C. (2005) Discordant trends in microvascular complications in adolescents with type 1 diabetes from 1990 to 2002.Diabetes Care, 28 (8), 1974-1980.
Publisher – Google Scholar
59. Monastiriotis, C., Papanas, N., Trypsianis, G., Karanikola, K., Veletza, S., Maltezos, E. (2013)
The ε4 allele of the APOE gene is associated with more severe peripheral neuropathy in type 2 diabetic patients.Angiology, 64 (4), 451-5.
Publisher – Google Scholar
60. Nalysnyk, L, Hernández-Medina, M, Krishnarajah, G. (2010) Glycaemic variability and complications in patients with diabetes mellitus: evidence from a systematic review of the literature. Diabetes Obes Metab, 12 (4), 288-298.
Publisher – Google Scholar
61. Nikitin, A. G., Chudakova, D. A., Strokov, I. A., Bursa, T. R., Chistiakov, D. A., Nosikov, V. V. (2008) Leu54Phe and Val762Ala polymorphoisms in the poly(ADP-ribose)polymerase-1 gene are associated with diabetic polyneuropathy in Russian type 1 diabetic patients. Diabetes Res Clin Pract , 79 (3), 446-452.
Publisher – Google Scholar
62. O´Connor TM, O´Connell J, O´Brien DI, Goode T, Bredin CP, Shanahan F. The role of substance P in inflammatory disease. J Cell Physiol 2004;201:167-180.
Publisher – Google Scholar
63. Oates, P. J. (2002) Polyol pathway and diabetic peripheral neuropathy. Int Rev Neurobiol, 50, 329-392.
Publisher – Google Scholar
64. Papanas, N., Papatheodorou, K., Papazoglou, D., Kotsiou, S., Christakidis, D., Maltezos, E. (2007)
An insertion/deletion polymorphism in the alpha2B adrenoceptor gene is associated with peripheral neuropathy in patients with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes, 115 (5), 327-30.
Publisher – Google Scholar
65. Papanas, N, Maltezos, E. (2013) Etiology, pathophysiology and classifications of the diabetic Charcot foot. Diabetic Foot Ankle, 4; DOI: 10.3402/dfa.v4i0.20872.
Publisher – Google Scholar
66. Park, T. S., Park, J. H., Baek, H.S. (2004) Can diabetic neuropathy be prevented? Diabetes Res Clin Pract, 66 (Supplement 1), S53-S56.
Publisher – Google Scholar
67. Pellerin, L., Magistretti, P. J. (2003) How to balance the brain budget while spending glucose differently. J Physiol, 546 (Pt 2), 325.
Publisher – Google Scholar
68. Perkins, B. A., Olaleye, D., Zinman, B., Bril, V. (2001) Simple screening tests for peripheral neuropathy in the diabetes clinic. Diabetes Care, 24 (2), 250-256.
Publisher – Google Scholar
69. Peters A, Laffel L, The American Diabetes Association Transitions Working Group. (2011)
Diabetes Care for emerging adults: recommendations for transition from pediatric to adult diabetes care systems.Diabetes Care, 34 (11), 2477-2485.
Publisher – Google Scholar
70. Pfeifer, M. A., Weinberg, C. R, Cook, D. L, Reenan, A, Halter, J. B., Ensinck, J. W., Porte, D. Jr. (1984) Autonomic neural dysfunction in recently diagnosed diabetic subjects. Diabetes Care, 7 (5), 447-453.
Google Scholar
71. Pop-Busui, R., Sima, A., Stevens, M. (2006) Diabetic neuropathy and oxidative stress. Diabetes Metab Res Rev , 22 (4), 257-273.
Google Scholar
72. Rafehi H, El-Osta A, Karagiannis T. C. (2011) Genetic and epigenetic events in diabetic wound healing. Int Wound J , 8 (1), 12-21.
Publisher – Google Scholar
73. Rafehi, H, El-Osta, A, Karagiannis, T. C. (2012) Epigenetic mechanisms in the pathogenesis of diabetic foot ulcers.J Diabetes Complications, 26 (6), 554-561.
Publisher – Google Scholar
74. Rogus, J. J., Warram, J. H., Krolewski, A. S. (2002) Genetic studies of late diabetic complications: the overlooked importance of diabetes duration before complication onset. Diabetes, 51 (6), 1655-1662.
Publisher – Google Scholar
75. Rolim, L. C., Sá, J. R., Chacra, A. R., Dib, S. A. (2009) Clinical heterogeneity and coexistence of diabetic neuropathies: difference and similarities between types 1 and 2 diabetes mellitus. Arq Bras Endocrinol Metabol, 53 (7), 818-824.
Google Scholar
76. Rosenbloom, A. L., Silverstein, J. H., Amemiya, S., Zeitler, P., Klingensmith, G. J.; International Society for Pediatric and Adolescent Diabetes. (2008) ISPAD Clinical Practice Consensus Guidelines 2006-2007.
Type 2 diabetes mellitus in the child and adolescent. Pediatric Diabetes, 9 (5), 512-526.
Publisher – Google Scholar
77. Said, G. (2007) Diabetic neuropathy — a review. Nat Clin Pract Neurol, 3 (6), 331-340.
Publisher – Google Scholar
78. Salzer, J. L. Brophy, P. J, Peles, E. (2008) Molecular domains of myelinated axons in the peripheral nervous system. Glia, 56 (14), 1532-1540.
Publisher – Google Scholar
79. Sfarti, C., Trifan, A., Hutanasu, C., Cojocariu, C., Singeap, A. M., Stanciu, C. (2010) Prevalence of gastroparesis in type 1 diabetes mellitus and its relationship to dyspeptic symptoms. J Gastrointestinal Liver Dis, 19 (3), 279-284.
Google Scholar
80. Shrestha, P, Ghimire, L. A. (2012) Review about the Effect of Lifestyle Modification on Diabetes and Quality of Life. Glob J Health Sci, 4 (6), 185-190.
81. Sima, A. A., Brismar, T. (1985) Reversible diabetic nerve dysfunction: structural correlates to electro´hysiological abnormalities. Ann Neurol, 18 (1), 21-29.
Google Scholar
82. Soedamah-Muthu, S. S., Abbring, S, Toeller, M. Diet, Lifestyle and Chronic Complications in Type 1 Diabetic Patients. S.S. Soedamah-Muthu, S. Abbring and M. Toeller (2011). Diet, Lifestyle and Chronic Complications in Type 1 Diabetic Patients, Type 1 Diabetes – Complications, Pathogenesis, and Alternative Treatments, Prof. Chih-Pin Liu (Ed.), ISBN: 978-953-307-756-7, InTech, DOI: 2011. Available at: http://www.intechopen.com/books/type-1-diabetes-complications-pathogenesis-and-alternative-treatments/diet-lifestyle-
and-chronic-complications-in-type-1-diabetic-patients
83. Sugimoto, K., Yusujima, M., Yagihahsi, S. (2008) Role of advanced glycation end products in diabetic neuropathy. Curr Pharm Des, 14 (10), 953-961.
Publisher – Google Scholar
84. Sytze Van Dam, P., Cotter, M. A., Bravenboer, B., Cameron, N. E. (2013) Pathogenesis of diabetic neuropathy: focus on neurovascular mechanisms. Eur J Pharmacol, 719 (1-3), 180-186.
Publisher – Google Scholar
85. Tan, L.S. (2010) The clinical use of the 10 g monofilament and its limitation: a review. Diabetes Res Clin Pract, 90 (1), 1-7.
Publisher – Google Scholar
86. Tesfaye, S., Vileikyte, L., Rayman, G., Sindrup, S., Perkins, B., Baconja, M., Vinik, A., Boulton, A; on behalf of the Toronto Expert Panel on Diabetic Neuropathy. (2011) Painful diabetic peripheral neuropathy: consensus recommendations on diagnosis, assessment and management. Diabetes Metab Res Rev 2011; DOI; 10.1002/dmrr.1225.
Google Scholar
87. Thorens, B., Mueckler, M. (2010) Glucose transporters in the 21st century. Am J Physiol Endocrinol Metab, 298 (2), E141-E145.
Publisher – Google Scholar
88. Tomlinson, D. R., Gardiner, N. J. (2008) Glucose neurotoxicity. Nat Rev Neurosci, 9 (1), 36-45.
Google Scholar
89. Unal-Cevik, I., Sarioglu-Ay, S., Evcik, D. A. (2010) comparison of the DN4 and LANSS questionnaires in the assessment of neuropathic pain: validity and reliability of the Turkish version of DN4. J Pain, 11 (11), 1129-1135.
Publisher – Google Scholar
90. Van Acker, K, Bouhassira, D, De Bacquer, D, Weiss, S, Matthys, K, Raemen, H, Mathiue, C, Colin, I. M. (2009)Prevalence and impact on quality of life of peripheral neuropathy with or without neuropathic pain in type 1 and type 2 diabetic patients attenting hospital outpatients clinics. Diabetes Metab, 35 (3), 206-213.
Publisher – Google Scholar
91. van Dieren, S, Beulens, J. W., van der Schouw, Y. T., Grobbee, D. E., Neal, B. (2010) The global burden of diabetes and its complications: an emerging pandemic. Eur J Cardiovasc Prev Rehabil,17 (Supplement 1), S3-S8.
Publisher – Google Scholar
92. Vazeou, A., Papadopoulou, A., Miha, M., Drakatos, A., Georgacopoulos, D. (2008) Cardiovascular impairment in children, adolescents, and young adults with type 1 diabetes mellitus (T1DM). Eur J Pediatr, 167 (8), 877-884.
Publisher – Google Scholar
93. Vazeou, A., Papadpoulou, A., Papadimitriou, A., Kitsou, E., Stathatos, M., Bartsocas, C. S. (2004) Autonomic neuropathy and gastrointestinal motility disorders in children and adolescents with type 1 diabetes mellitus. J Pediatr Gastroenterol Nutr, 38 (1), 61-65.
Publisher – Google Scholar
94. Vinik, A, Erbas, T. Neuropathy. In Handbook of Exercise in Diabetes. 2nd ed. Ruderman N, Devlin JT, Kriska A, Eds. Alexandria, VA, American Diabetes Association 2002, p. 463-496.
95. Vinik, A. (2010) The approach to the management of the patient with neuropathic pain. J Clin Endocrinol Metab, 95 (11), 4802—4811.
Publisher – Google Scholar
96. Vinik, A. I., Casellini, C. M. (2013) Guidelines in the management of diabetes nerve pain: clinical utility of pregabalin. Diabetes Metab Synd Obesity, 6:57-78.
Publisher – Google Scholar
97. Vinik, A. I., Freeman, R, Erbas, T. (2003) Diabetic autonomic neuropathy. Semin Neurol, 23 (4), 365-372
Publisher – Google Scholar
98. Vinik, A. I., Maser, R. E., Mitchell, B. D., Freeman, R. (2003) Diabetic autonomic neuropathy. Diabetes Care, 26 (5), 1553-1579.
99. Voulgari, C., Psallas, M., Kokkinos, A., Argiana, V., Katsilambros, N., Tentolouri,s N. (2011) The association between cardiac autonomic neuropathy with metabolic and other factors in subjects with type 1 and type 2 diabetes. J Diabetes Complications, 25 (3), 159-167.
Publisher – Google Scholar
100. Weintrob, N., Josefberg, Z., Galazar, A., Vardi, P., Karp, M. (1997) Acute painful neuropathic cachexia in a young type 1 diabetic woman: a case report. Diabetes Care, 20 (3), 290-291.
Google Scholar
101. Weisman, A., Bril, V., Ngo, M., Lovblom, L. E., Halpern, E. M., Orszag, A., Perkins, B. A. (2013) Identification and prediction of diabetic sensorimotor polyneuropathy using individual and simple combinations of nerve conduction study parameters. PLoS ONE, 8 (3), e58783.
Publisher – Google Scholar
102. Wiggin TD, Sullivan KA, Pop-Busui R., Amato, A., Sima, A. A., Feldman, E. L. (2009) Elevated triglycerides correlate with progression of diabetic neuropathy. Diabetes, 58 (7), 1634-1640.
Publisher – Google Scholar
103. Williamson, J. R., Chang, K., Frangos, M., Hasan, K.S., Ido, Y., Kawamura, T., Nyengaard, J. R., van den Enden, M., Kilo, C., Tilton, R. G. (1993) Hyperglycemic pseudohypoxia and diabetic complications. Diabetes, 42 (6), 801-813.
Publisher – Google Scholar
104. Yigit, S, Karakus, N, Inanir, A. (2013) Association of MTHFR gene C677T mutation with diabetic peripheral neuropathy and diabetic retinopathy. Mol Vis, 19, 1626-1630.
Google Scholar
105. Zelmanovitz, T, Gerchman, F, Balthazar, A. P., Thomazelli, F. C., Matos, J. D., Canani, L. H. (2009) Diabetic nephropathy. Diabetol Metab Syndr, 1 (1):10.
106. Zenker, J., Ziegler, D., Chrast, R. (2013) Novel pathogenic pathways in diabetic neuropathy. TRENDS Neurosciences, 36 (8), 439-449.
Publisher – Google Scholar