Introduction
Diabetes mellitus is the most common cause of autonomic neuropathy. And neuropathy is the most common complication of diabetes mellitus and may have both somatic and autonomic features.1 Autonomic decline includes (in order) peripheral autonomic neuropathy (PAN), then diabetic autonomic neuropathy (DAN), and finally cardiovascular autonomic neuropathy (CAN).2,3
There are three stages to the CAN. Early stage: abnormality of heart rate response during deep breathing alone. Intermediate stage: an abnormality of Valsalva response. Severe stage: the presence of postural hypotension.4
Clinical symptoms of autonomic neuropathy generally do not occur until long after the onset of diabetes.3,5 Although symptoms suggestive of autonomic dysfunction may be common, they may frequently be due to other causes rather than to the true autonomic neuropathy. Subclinical autonomic dysfunction can, however, occur within a year after diagnosis among type 2 diabetes patients and within two years among type 1 diabetes patients. 6 Because of its association with a variety of adverse outcomes including cardiovascular deaths, the CAN is the most clinically important and well-studied form of the DAN.
The number of bedside tests used to diagnosed patients with the CAN are different from one study to another ranging from one to six.3 If more strict criteria were used (i.e., abnormalities present in at least three of six autonomic function tests), the prevalence of CAN was 16.8% for individuals with type 1 diabetes and 22.1% for individuals with type 2 diabetes.3
Results from earlier research suggest that using a battery from cardiovascular tests (some indicating parasympathetic involvement and others indicating possible sympathetic involvement) would make it possible to follow the progression of autonomic function over time.7
The three tests recommended were heart rate response to 1) deep breathing, 2) standing, and 3) the Valsalva maneuver. Two tests of blood pressure control were also recommended: blood pressure response to 1) standing or passive tilting and 2) sustained handgrip. These tests were judged suitable for both routine screening and monitoring of the progress of autonomic neuropathy.4
In 1992, a consensus development conference on standardized measures in diabetic neuropathy review the state-of-the-art diabetic neuropathy measures used in epidemiological and clinical studies including cross-sectional, longitudinal, and therapeutic trials .The panel revised its recommendation to include three tests for the longitudinal testing of the CAN: 1) heart rate response during deep breathing, 2) Valsalva maneuver, and 3) postural blood pressure testing .8
Aims of this study was to determine the prevalence of CAN in diabetic patients in Basrah by conducting cardiac autonomic function tests and to detect the predictors of the CAN in diabetic patients .
Methods
The study was conducted at the Al-Faiha Diabetes Endocrine and Metabolism Center (FDEMC) in Basrah for the period from January to December 2011.Sixty eight patients were selected to participate in this study. Written informed consent was obtained from each patient. Patients were selected irrespective of the duration of disease and therapeutic status. All patients were examined by a single investigator. Diabetes mellitus was defined as plasma glucose level being 200 mg/dl or higher independent of fasting time, fasting plasma glucose of ≥ 126 mg/dl on two occasions or, use of anti-diabetes agents or a combination of these. Current smoker was defined as any person who smoked cigarettes regardless of the number in the preceding three months. Hypertension was defined as systolic blood pressure being 140 mmHg or higher, diastolic blood pressure being 90 mmHg or higher, use of antihypertensive agents or a combination of these. Blood pressure was measured in a seated position after 5 minutes at rest, three times. The average of the second and third readings was defined as the subject’s blood pressure. Diagnosis of peripheral neuropathy was based on quantitative assessment of symptoms and physical finding.9 Body mass index (BMI, kg/m2) was computed from height and weight measurements. Exclusion criteria included those with poor functional capacity, amputation of any of the lower limbs, acute ischemic heart disease with admission to contrary care unit in the preceding month and cerbrovasuclar disease interfering with walking , standing or speech.
Clinical symptoms of autonomic neuropathy were assessed on the basis of presence or absence of various symptoms, such as nausea, vomiting, nocturnal diarrhea, constipation, early satiety, stool incontinence, gustatory sweating, bloating, fullness, dry skin, erectile dysfunction, urine retention, sterility or infertility, palpitation, syncope, or dizziness. The definitions of above symptoms were based on recommendation of American diabetic association (ADA).3
The tests for autonomic cardiovascular function performed as side procedures are described below. The tests were carried out in the morning. The participants arrived at 9 a.m, in the fasting state. Examination started with the patient resting in lying position for 5 minutes. A 12-lead ECG was performed with a standard device. The whole examination lasts 15-20 minutes per patient and no time for preparation is needed. The autonomic nervous system was examined using a battery from the five function tests.
Cardiovascular Autonomic Function Tests
- Beat-to-beat heart rate variation (E/I) ratio (With the patient at rest and supine (no overnight coffee or hypoglycemic episodes), breathing 6 breaths/min, heart rate monitored by ECG , a difference in heart rate of > 15 beats/min is normal and 1.17 is abnormal). (Parasympathetic activity).
- Heart rate response to Valsalva maneuver {(The subject forcibly exhales into the mouthpiece of a manometer to 40 mmHg for 15 s during ECG monitoring. Healthy subjects develop tachycardia and peripheral vasoconstriction during strain and an overshoot bradycardia and rise in blood pressure with release. The ratio of longest R-R shortest R-R should be > 1.2)}. (Parasympathetic activity)
- Heart rate response to standing {(30:15 ratio) (During continuous ECG monitoring, the R-R interval is measured at beats 15 and 30 after standing. Normally, a tachycardia is followed by reflex bradycardia. The 30:15 ratio is normally > 1.03)}. (Parasympathetic activity)
- Systolic blood pressure response to standing (systolic blood pressure is measured in the supine subject. The patient stands and the systolic blood pressure is measured after 2 min. Normal response is a fall of 30 mmHg with symptoms). (Sympathetic activity)
- Diastolic blood pressure response to isometric exercise (handgrip) {(The subject squeezes a handgrip dynamometer to establish a maximum. Grip is then squeezed at 30% maximum for 5 min. The normal response for diastolic blood pressure is a rise of > 16 mmHg in the other arm)}. (Sympathetic activity)
If two of the aforementioned test results were abnormal, the presence of cardiovascular autonomic neuropathy was assumed.10
Biochemical Studies
Fasting serum samples were analyzed in the same local laboratory. Each subject’s blood was sampled: 10 mL was drawn into a tube after the subject had fasted for at least 10 hours. Serum total cholesterol,high density lipoprotein cholesterol (HDL-C), and triglyceride (TG) were measured by the enzymatic method. Low density lipoprotein cholesterol (LDL-C) concentrations were calculated using the Friedewald formula.Hb A1c was measured using Bio-Rad D10 analyzer (HPLC cation-exchange chromatography) .
An ethical approval for this study was obtained from the Basrah directorate of health.
Statistics
The group data are presented as mean± standard deviation unless otherwise specified .A p value <0.05was considered significant. All analyses were performed using SPSS software version 18.0 (SPSS, Chicago, IL, USA). Univariate analysis was performed to determine each variable’s association with autonomic neuropathy. In the sixty eight patients, univariate analysis was performed for each of the factors with the presence or absence of autonomic neuropathy, then the presence of autonomic neuropathy used as dependent variable. Variables found associated with autonomic neuropathy in univariate analyses were then entered into a multivariable model using logistic regression to determine the strength of each variable in association with autonomic neuropathy.
Results
Autonomic neuropathy was presents in twenty nine patients (42.6%) as shown in table one. Abnormalities of two tests were seen in thirteen patients (19.1%), three tests in six ( 8.8%), four tests in nine (13.2%) and five tests in one (1.5%).
Autonomic test results are summarized in table 2. Parasympathetic function: abnormal beat-to-beat heart rate variation (E/I) ratio are present in 61.8%; abnormal heart rate response to Valsalva maneuver in 33.8 %; and abnormal heart rate response to standing in 23.5%. Sympathetic function: abnormal diastolic blood pressure response to isometric exercise are present in 25%, and orthostatic hypotension (defined as systolic blood pressure fall of >30 mmHg within 2 minutes of standing) in 5.1%.
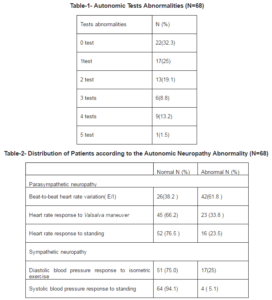
The complete demographic data for all sixty eight patients are shown in table three. The total enrolled was sixty eight patients (all were type 2 diabetes mellitus except three) of them thirty five men and thirty three women. Variables associated with autonomic neuropathy with statistical significance were gastroparesis, nocturnal diarrhea, constipation, areas of symmetrical anhydrosis, gustatory sweating, dry skin, erectile dysfunction for men, urine retention or incontinence, palpitation, exercise intolerance, peripheral neuropathy, Insulin use, duration of diabetes, and LDL- cholesterol level. The other variables are not associated with neuropathy like gender, smoking, dizziness or syncope, BMI, hypertension, the use of hypertension medication, Hba1c level, plasma glucose, total cholesterol, triglyceride, HDL-cholesterol levels.
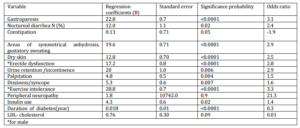
Predictors of autonomic neuropathy identified by the multivariate analysis are shown in table four. Variables which remain statistically significantly associated with autonomic neuropathy were gastroparesis, nocturnal diarrhea, areas of symmetrical anhydrosis, gustatory sweating, dry skin, erectile dysfunction for men, palpitation, exercise intolerance and insulin use.
Discussion
Forty two point six percent of the selected patients had the CAN (manifested as two abnormalities and above. The commonest parasympathetic neuropathy abnormality was abnormal beat-to-beat heart rate variation (E/I) in 61.8% and the commonest sympathetic neuropathy abnormality was diastolic blood pressure response to isometric exercise in 25% with 19.1% had two abnormalities. This finding has been confirmed in other studies, where the most informative test to assess cardiovascular autonomic neuropathy in diabetes mellitus is the abnormal beat-to-beat heart rate variation (E/I).11In Omdurman, Sudan, evidence suggests that the CAN is present in 70% of the diabetic patients. Those who had evidence of CAN had abnormal beat-to-beat heart rate variation (E/I) in 90%, abnormal response of heart rate to standing was positive in 81% of diabetic patients and abnormal heart rate response to Valsalva was found in 45% of the same studied patients.12
In one study conducted in India, the CAN was found in 54% of diabetic patients. About 28% had two abnormal cardiovascular reflexes.13 Abnormal beat-to-beat heart rate variation (E/I) ratio was the most sensitive test to determine autonomic neuropathy. It was abnormal in 32% patients, this was followed by abnormal heart rate response to standing, which was abnormal in 24% of the patients, Abnormal diastolic blood pressure response to isometric exercise (handgrip) test seen in 16% of the patients. Heart rate response to Valsalva maneuver ratio was abnormal in 22% of the patients. While in another study from India, the CAN was seen in 57.5%, abnormal beat-to-beat heart rate variation (E/I) ratio in 30% and abnormal heart rate response to standing in 27.5%. Abnormal heart rate response to Valsalva maneuver ratio in 25%, abnormal diastolic blood pressure response to isometric exercise (handgrip) in 20%, One abnormal cardiac reflex was seen in 15%, 2 in 32.5% and 3 or more in 10%.14
Systolic blood pressure response to standing abnormality was rare in the study where it was seen only in 5.1%. This result was not astonishing as the presence of postural hypotension was in the late stage in the natural history of autonomic neuropathy.4 In India, studies found the least sensitive test to detect autonomic neuropathy was postural hypotension. This was abnormal in 14-15% of the cases.13,14
The long-term predictive power of five simple function tests with regard to all-cause mortality in individuals with diabetes from the general population found that only heart rate response to Valsalva maneuver ratio, beat-to-beat heart rate variation (E/I ) ratio and diastolic blood pressure response to isometric exercise (handgrip) were predictors of survival.15
In this study, variables not associated with neuropathy were gender, smoking, dizziness or syncope, BMI, hypertension, the use of hypertension medication, HbA1c level, plasma glucose, total cholesterol, triglyceride, HDL-cholesterol levels. Several other studies have found significant relations between the CAN and peripheral neuropathy, systolic blood pressure, retinopathy and nephropathy, whereas no relationships with HbA1c, hyperlipidemia or BMI were found.16,17Duration of diabetes in this study was not predictor of autonomic neuropathy. The EURODIAB study concluded that duration of diabetes was no longer related to incidence of the CAN when adjusted for age and sex.18
Table-3- Univariate Association of Potential Confounding Variables with Autonomic Neuropathy (N=68)
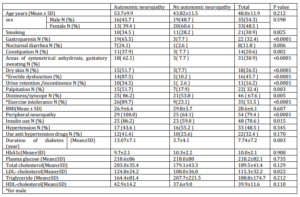
Table-4- Predictors of Autonomic Neuropathy Identified by Multivariate Analysis (N=68)
Conclusion
Various variables can predict the CAN, which affects more than 40% of diabetic patients in Basrah.
Reference
1- Vinik, A. I., Freeman, R. & Erbas, T. (2003). “Diabetic Autonomic Neuropathy,” Seminars in Neurology, Dec 2003; 23(4):365-72.
Publisher – Google Scholar
2- Boulton, A. J. M., Vinik, A. I., Arezzo, J. C., Bril, V., Feldman, E. L., Freeman, R., et al. (2006). “Etal.”Diabetic Neuropathies: A Statement by the American Diabetes Association,” Diabetes Care. 2005 Apr; 28(4):956-62.
Publisher – Google Scholar
3- Vinik, A. I., Maser, R. E., Mitchell, B. D. & Freeman, R. (2003). “Diabetic Autonomic Neuropathy,” Diabetes Care.2003 May; 26(5):1553-79.
Publisher – Google Scholar
4- Consensus Statement: Report and Recommendations of the San Antonio Conference on Diabetic Neuropathy. (1988). American Diabetes Association American Academy of Neurology, Diabetes Care. 1988 Jul-Aug; 11(7):592-7.
Publisher
5- Vinik, A. I. & Ziegler, D. (2007). “Diabetic Cardiovascular Autonomic Neuropathy,” Circulation. 2007 Jan; 115(3):387-97.
Publisher – Google Scholar
6- Pfeifer, M. A., Weinberg, C. R., Cook, D. L., Reenan, A., Halter, J. B., Ensinck, J. W. & Porte, D. (1984). “Autonomic Neural Dysfunction in Recently Diagnosed Diabetic Subjects,” Diabetes Care. 1984 Sep-Oct; 7(5):447-53.
Publisher – Google Scholar
7- Mackay, J. D., Page, M. M., Cambridge, J. & Watkins, P. J. (1980). “Diabetic Autonomic Neuropathy: The Diagnostic Value of Heart Rate Monitoring,” Diabetologia. 1980 Jun; 18(6):471-8.
Publisher – Google Scholar
8- Kahn, R. (1992). “Proceedings of a Consensus Development Conference on Standardized Measures in Diabetic Neuropathy,” Diabetes Care .1992 15(Suppl. 3):1080—107.
Publisher – Google Scholar
9- Young, M. J., Boulton, A. J. M., Macleod, A. F., Williams, D. R. R. & Sonksen, P. H. (1993). “A Multicenter Study of the Prevalence of Diabetic Peripheral Neuropathy in the United Kingdom Hospital Clinic Population,” Diabetologia. 1993 Feb; 36(2):150-4.
Publisher – Google Scholar
10- Ziegler, D., Dannehl, K., Muhlen, H., Spuler, M. & Gries, F. A. (1992). “Prevalence of Cardiovascular Autonomic Dysfunction Assessed by Spectral Analysis, Vector Analysis, and Standard Tests of Heart Rate Variation and Blood Pressure Responses at Various Stages of Diabetic Neuropathy,” Diabetic Medicine. 1992 Nov; 9(9):806-14.
Publisher – Google Scholar
11- May, O. & Arildsen, H. (2000). “Assessing Cardiovascular Autonomic Neuropathy in Diabetes Mellitus: How Many Tests to Use?,” Journal of Diabetes and its Complications. 2000 Jan-Feb; 14(1):7-12.
Publisher – Google Scholar
12- Hussein, A., Fatah, T. A., Sidig, A., Hamad, A., Gadour, M. O., Yassien, F., Ali, A. B. M., Saad, A. & Aldar, M. M. (2011). “Frequency and Clinical Pattern of Autonomic Neuropathy in Adult Diabetic Sudanese Patients,” International Journal of the Physical Sciences. 2011 Jan; 6(2): 308—312.
Publisher
13- Basu, A. K., Bandyopadhyay, R., Chakrabarti, S., Paul, R. & Santra, S. (2010). “A Study on the Prevalence of Cardiac Autonomic Neuropathy in Type-2 Diabetes in Eastern India,” Journal, Indian Academy of Clinical Medicine. 2010; 11(3): 190-4.
Publisher – Google Scholar
14- Mehta, S., Mathur, D., Chaturvedi, M. & Verma, K. (2002). “Incidence of Cardiac Autonomic Neuropathy and Its Correlation with Retinopathy, Micro-Albuminuria and Glycated Haemoglobin in Non-Insulin Dependent Diabetes Mellitus,”Journal of the Indian Medical Association. 2002; 100:141-3.
Publisher – Google Scholar
15- May, O. & Arildsen, H. (2011). “Long-Term Predictive Power of Simple Function Tests for Cardiovascular Autonomic Neuropathy in Diabetes: A Population-Based Study,” Acta Diabetologica. 2011 Dec; 48(4):311-6.
Publisher – Google Scholar
16- Chen, H.- T., Lin, H.- D., Won, J. G. S., Lee, C.- H., Wu, S.- C., Lin, J.- D., Juan, L.- Y., Ho, L.- T. & Tang, K.- T. (2008). “Cardiovascular Autonomic Neuropathy, Autonomic Symptoms and Diabetic Complications in 674 Type 2 Diabetes,” Diabetes Research and Clinical Practice. 2008 Nov; 82(2):282-90.
Publisher – Google Scholar
17- Lacigová, S., Safránek, P., Cechurová, D., Krcma, M., Vísek, J., Jankovec, Z., Zourek, M., Haladová, I. & Rusavý, Z. (2007). “Could We Predict Asymptomatic Cardiovascular Autonomic Neuropathy in Type 1 Diabetic Patients Attending Out-Patients Clinics?,” Wiener Klinische Wochenschrift. 2007; 119(9-10):303-8.
Publisher – Google Scholar
18- Witte, D. R., Tesfaye, S., Chaturvedi, N., Eaton, S. E. M., Kempler, P. & Fuller, J. H. (2005). “Risk Factors for Cardiac Autonomic Neuropathy in Type 1 Diabets Mellitus,” Diabetologia. 2005 Jan; 48(1):164-71.
Publisher – Google Scholar





