Introduction
Gestational weight gain (GWG) has been thoroughly studied as a predictor of adverse pregnancy outcomes. Low GWG is associated with LFD infants and preterm birth (Sebire et al., 2001, Stotland et al., 2006, Takimoto et al., 2006, Tsukamoto et al., 2007), whereas high GWG is associated with greater risk of cesarean section (Johnson et al., 1992). In addition, Barker did a study in 1986 regarding the association between low birth weight and death from coronary heart disease, suggesting that prenatal environmental factors are involved in the pathogenesis of adult-onset diseases (Barker et al., 1986).
In the following decades, numerous epidemiological studies substantiated a close association between low birth weight and an increased risk of developing adult diseases, including cardiovascular disease (Rich-Edwards et al., 1997), stroke (Lawlor et al., 2005), type 2 diabetes mellitus (Curhan et al., 1996) and hypertension (Huxley et al., 2000). The results of these reports led to the ‘Developmental Origins of Health and Disease’ (DOHaD) hypothesis in which epigenetic changes influenced by the environment in the early life stage can alter later disease risk (Gluckman et al., 2007). In 2009, 9.6% of infants born in Japan weighed less than 2,500 grams at birth, which was almost double the level recorded in 1980, while the rate is 6.7% in Organization for Economic Co-operation and Development countries (OECD)(2012). In addition, the average birth weight of Japanese infants has gradually declined since 1985.
In 2006, the Japanese Ministry of Health, Labor and Welfare (HLWM) recommended pregnant women to eat a balanced diet and achieve adequate weight gain based on their pre-pregnancy body size (2006). In this statement, underweight and normal weight women were advised to gain 7-12kg and 9-12kg, respectively, as well as specific professional advice to prevent perinatal complicating diseases, such as gestational diabetes mellitus or PIH, for overweight and obese women. On the other hand, in 2009, the US Institute of Medicine (IOM) released a new guideline for weight gain during pregnancy that aimed to optimize outcomes each woman and infant (Rasmussen et al., 2009). The IOM recommended greater GWG than HLWM recommended: underweight women were advised to gain 12.5~18kg, normal weight women 11.5~16kg, overweight women 7~11.5kg, and obese women 5~9kg, respectively.The IOM also indicated that it was unable to find sufficient evidence to continue to support a modification of GWG guidelines for women of short stature, and they also recommended that women of short stature (<157cm) gain at the lower end of the range of their pre-pregnant BMI. In this cohort study, we investigated the independent risk factors of pre-pregnancy body mass index (BMI), GWG, and other factors, including maternal height, for pregnancy complications in Japanese women, using both recommendations released from HLWM and IOM.
Materials and Methods
Maternal and infant birth data from January 2005 to August 2013 were extracted retrospectively from hospital records at Izumiotsu Municipal Hospital on an anonymous basis. Among 6,100 Japanese pregnant women who delivered babies at this hospital, 5,602 women were investigated after excluding stillborn infants, multiple births and premature deliveries. The questionnaire elicited information about body height, pre-pregnancy bodyweight and parity. Pre-pregnancy BMI was calculated as pre-pregnancy bodyweight divided by height squared. Ethics approval for the study was obtained from institutional review boards of Izumiotu Municipal Hospital in Izumiotu, Japan.
According to pre-pregnancy BMI, we divided all patients into four groups; underweight (group U:<18.5kg/m2), normal weight (group N:18.5-24.9kg/m2), overweight (group O:25-29.5kg/m2), and obese (group OB:³30kg/m2) according to the guidelines of the World Health Organization (WHO). GWG was calculated by subtracting the maternal pre-pregnancy weight from the weight at delivery and was categorized as insufficient, adequate HLWM, adequate IOM or excessive in accordance with the HLWM guidelines of 2006 and the IOM guidelines of 2009 for each BMI category. GWG was also classified into four groups based on the recommended levels of the two guidelines as shown Table1. We divided all patients into three groups according to height; more than 158cm (average of Japanese women), 152-158cm, less than 152cm (less than 10% tile of Japanese women).Neonates were classified into three groups: light for date (LFD), appropriate for gestational age (AFD) and heavy for date (HFD) by a standard deviation score (SD score) for birth weight using the “Birth Size Standards by Gestational Age for Japanese Neonates” released by the Japan pediatric society in 2011 (Itabashi et al., 2010) LFD infant was defined as infants who had an SD score below -1.5SD and HFD infants were defined as over +1.5SD. Pregnancy-induced hypertension (PIH) was defined as gestational hypertension (blood pressure >140/90 mmHg) without proteinuria at a gestational age of >20 weeks on two or more occasions at least 6 h apart,
or pre-eclampsia (blood pressure >140/90 mmHg) in combination with proteinuria (>0.3 gm/24 h) after 20 weeks of gestation. ECS was defined as unscheduled cesarean section.The risks of LFD, HFD, ECS and PIH were evaluated using multiple logistic models (Ekuseru-Toukei2012:Social Survey Research Information Co., Ltd., Tokyo, Japan).
Adjusted odds ratios (ORs) and 95% confidence intervals (95% CIs) were estimated taking into account possible confounders which could have any effect on perinatal outcomes. Maternal age at delivery, parity, pre-pregnancy BMI, and maternal height were included as confounders to estimate the effect of weight gain during pregnancy on perinatal outcomes. Dunnett’s test was done to compare averages for each group with the normal weight group. The association between two categorical variables was investigated with the likelihood ratio χ2 with a Bonferroni correction. A P-value of less than 0.05 was considered significant.
Results
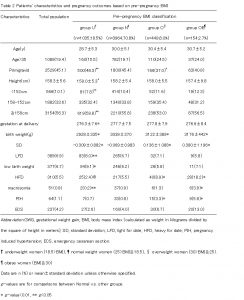
18.5% of women were in group U, 70.8% of women were in group N, 8.0% of women were in group O and 2.7% of women were in group OB based on their pre-pregnancy BMI. In the overall cohort, the infant outcome rates were significantly different in group U compared with group N (8.0% compared with 6.7% for LFD), and in group O and group OB compared with group N (8.9% and 18.2% compared with 5.5% for HFD, 3.3% and 5.8% compared with 0.8% for PIH, 6.7% and 13.0% compared with 4.0% for ECS)(Table2).
On the basis of HLWM and IOM gestational weight gain recommendations, 19.7% of women gained less weight than the HLWM recommended, 48.3% of women gained the HLWM recommended weight, and 24.5% of women gained weight as the IOM recommended, while 7.6% of women gained more than the IOM recommended (Table 3). Mean GWG weights were 10.6kg for group U, 10.3kg for group N, 8.6kg for group O, and 5.6kg for group OB.
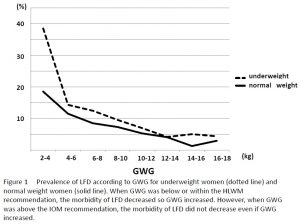
The proportions of HFD increased as pre-pregnancy BMI increased. The highest proportion of LFD infants were born to women who began pregnancy with a lower BMI. In groups U and N, the proportions of LFD continuously decreased with every 2kg of weight gain during pregnancy, and the rate of LFD was lowest when weight gain was within the IOM recommended range (Fig 1).
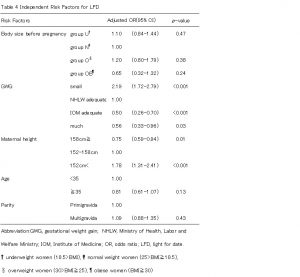
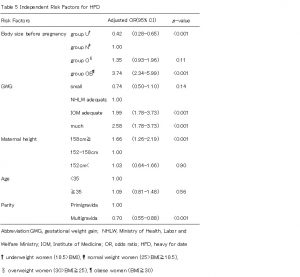
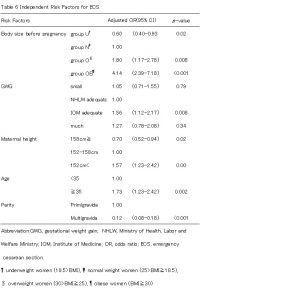
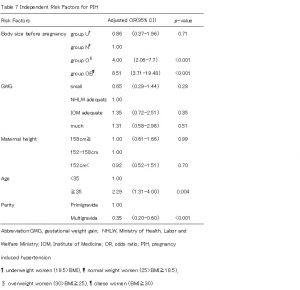
We stratified outcomes by pre-pregnancy BMI and used a multivariable model to compare women who gained less than HLWM and women who gained within IOM, as well as women who gained more than IOM with women who gained within HLWM. After adjusting for maternal age, parity, and height, women who gained less than HLWM were at increased risk of LFD (OR 2.19, 95% CI 1.72-2.79), but women who gained within IOM were at decreased risk of LFD (OR 0.50, 95% CI 0.26-0.70)(Table4). Women who gained within IOM and more than IOM were at increased risk of HFD (OR 1.99, 95% CI 1.50-2.64; OR 2.58, 95% CI 1.78-3.73) (Table5). Group O and OB were at increased risk of emergency cesarean delivery (OR 1.80, 95% CI 1.17-2.78; OR 4.14, 95% CI 2.39-7.18) and group Uwas at decreased risk (OR 0.60, 95% CI 0.40-0.93). Women who gained within IOM were also increased risk of emergency cesarean delivery (OR 1.56, 95% CI 1.12-2.17) (Table 6). On the other hand, women who were taller 158cm and multiparous were at decreased risk of ECS (OR 0.70, 95% CI 0.52-0.94; OR 0.12, 95%CI 0.08-0.18) (Table6). Group O and OB were at increased risk of PIH (OR 4.00, 95% CI 2.06-7.74; OR 8.51, 95% CI 3.71-19.48), but this was not related to weight gain during pregnancy (Table7).
Discussion
Since there has been no standard guideline for GWG according to pre-pregnancy BMI for pregnant Japanese women, we investigated the differences in perinatal prognosis when pregnant women gained weight during pregnancy within the recommended levels from the Japanese HLWM or the US IOM.In our cohort of 5,602 pregnant women, approximately half of them gained weight during pregnancy within the HLWM recommendation and a quarter of women within the IOM recommendation.
We found that lower GWG than the HLWM recommendation was significantly associated with a higher risk of LFD as others have reported (Watanabe ea al., 2010). On the other hand, GWG within the IOM recommendation resulted in significantly lower risk of LFD. In our analysis, there was no statistical significance for the relationship between body size and LFD, which differed from the observation Murakami et al., reported (Murakami et al., 2005). GWG within the IOM recommendation was significantly associated with increased risk of HFD (OR:1.99), and a high prevalence of HFD is linked with increased risk of emergent cesarean section (OR:1.56). Women who were taller than 158cm (average height of Japanese women) or multigravida women were associated with significantly lower risk of ECS. In this regard, it is possible that women taller than 158cm height or multigravida women should gain weight during pregnancy within the IOM recommendation rather than the HLWM recommendation in order to reduce the prevalence of LFD. Meanwhile, the ORs for PIH were not higher regardless of GWG, although it was higher in groups O (OR:4.00 95%CI 2.06-7.74) and group OB (OR:8.51 95%CI 3.71-19.48). In U.S. studies, a significant relationship between GWG and the prevalence of PIH was reported (DeVader et al., 2007, Kiel et al., 2007). Murakami et al., reported that GWG did not show any significant influence on the onset of PIH, although being obese before pregnancy increased the risk of PIH onset compared with normal weight women, which was similar to the result of our study19. Other perinatal factors other than GWG such as hypertension or diabetes mellitus may be involved in the onset of PIH. Further investigation will be necessary to confirm these factors because we did not exclude these complicating cases.
The study population was from a single hospital with the same protocol. This is very important to strengthen the statistical power. We could directly manage all the medical records of the participants, which provided highly reliable data. Nevertheless, our study has certain limitations. In this study, we did not consider gestational diabetes because the diagnostic criteria of gestational diabetes were changed during the cohort. There is some concern that higher GWG results in increased risk of gestational diabetes. Consequently, further research will be needed to investigate the relationship between women who gain weight during pregnancy within the IOM recommendation and maternal glucose in tolerance.
It is necessary to reconsider adequate weight gain during pregnancy because birth weight in Japanese infants has continuously declined since the 1980s and the prevalence of low-birth-weight infants has steadily increased, reaching almost 10% of live births in recent years. In our investigation, adequate weight gain during pregnancy should be within the IOM recommendation to reduce the number of LFD infants, but we need to pay attention to adverse events when pregnant women gain more weight. Women whose BMI is over 25 should gain weight during pregnancy, but not over the HLWM recommendation because they are at higher risk for emergency cesarean section and PIH. Furthermore, women who are shorter than 152cm should not gain weight over the HLWM recommendation because they are at higher risk of HFD and ECS. Our investigation suggests that women of normal weight or lower should gain weight during pregnancy within the IOM recommendation; this will not increase perinatal or maternal adverse events and will reduce the number of LFD infants.
Acknowledgments
We thank all medical clerks of Izumiotsu Municipal Hospital for a great deal of assistance in managing the participants’ data. Extending appreciation for their cooperation to all study participants, we wish them and their children future good health.
Disclosure
The authors have no potential conflicts of interest to disclose.


(adsbygoogle = window.adsbygoogle || []).push({});
References
- Barker DJ and Osmond C. (1986) “Infant mortality, childhood nutrition, and ischaemic heart disease in England and Wales.”Lancet, 327 (8489) 1077-81.
- Curhan GC, Willett WC andRimm EB (1996) “Birth weight andadulthypertension, diabetes mellitus, and obesity in US men.”Circulation, 94 (12) 3246-50.
- DeVader SR, Neeley HL and Myles TD.(2007)“Evaluation of gestational weight gain guidelines forwomen with normal pre-pregnancy body mass index.”Obstetrics and Gynecology, 110 (4) 745-51.
- Gluckman PD, Hanson MA andBeedle AS. (2007)“Early life events and their consequences for later disease:a life history and evolutionary perspective.”American Journal of Human Biology, 19 (1) 1-19.
- Huxley RR, Shiell AWand Law CM. (2000)“The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature.”Journal of Hypertension, 18 (7) 815-31.
- Itabashi K, Fujimura M and Kusuda S (2010)“Introduction of new gestational age-specific standards for birth size.”The Journal of the Japan Pediatric Society, 114 (8) 1271-93.
- Johnson JW, Longmate JA and Frentzen B. (1992)“Excessive maternal weight and pregnancy outcome.”American Journal of Obstetrics and Gynecology, 167 (2) 353-70.
- Kiel DW, Dodson EA andArtal R (2007)“Gestational weight gain and pregnancy outcomes in obese women: how much is enough?”Obstetrics and Gynecology, 110 (4) 752-8.
- Lawlor DA, Ronalds G and Clark H. (2005)“Birth weight is inversely associated with incident coronary heart disease and stroke among individuals born in the 1950s: findings from the Aberdeen Children of the 1950s prospective cohort study.”Circulation, 112 (10) 1414-8.
- Ministry of Health, Labour and Welfare.(2006) “Indicator of Eating Habits for Pregnant and Postpartum Women, Promotion of Healthy Parents and Children 21 Report.” Tokyo: Mothers’ & Children’s Health Organization (in Japanese).
- Murakami M, Ohmichi M and Takahashi T. (2005) “Pre-pregnancy body mass index as an important predictor of perinatal outcomes in Japanese.” Archives of Gynecology and Obstetrics, 271 (4) 311-5.
- OECD Health data (2012) “Organisation for Economic Co-operation and Development.” [online][Retrieved19August 2014.] http://www.oecd.org/health/ healthdata
- Rasmussen KM andYaktine AL.(2009) “Weight Gain during Pregnancy: Reexamining the Guidelines.” National Academies Press, Washington, DC, USA.
- Rich-Edwards JW, Stampfer MJ and Manson JE.(1976) “Birth weight and risk of cardiovascular disease in a cohort of women followed up since 1976.”British Journal of Medicine,315 (7105) 396-400.
- Sebire NJ, Jolly M and Harris J. (2001) “Is maternal underweight really a risk factor for adverse pregnancy outcome? A population-based study in London.”British Journal of Obstetrics and Gynaecology, 108 (1) 61-6.
- Stotland NE, Cheng YW and Hopkins LM.(2006) “Gestational weight gain and adverse neonatal outcome among term infants.”Obstetrics and Gynecology, 108 (3) 635-43.
- Takimoto H, Sugiyama T and Fukuoka H. (2006) “Maternal weight gain ranges for optimal fetal growth in Japanese women.”International Journal of Obstetrics and Gynecology, 92 (3) 272-8.
- Tsukamoto H, Fukuoka H andKoyasu M. (2007) “Risk factors for small for gestational age.”Pediatrics International, 49 (6) 985-90.
- Watanabe H, Inoue K andDoi M. (2010) “Risk factors for term small for gestational age infants in women with low pre-pregnancy body mass index.”Journal of Obstetrics and Gynaecology Research, 36 (3) 506-12.










