Birthweight is one of the most important determinants of perinatal, neonatal and postnatal outcomes. A poor growth during the intrauterine period increases the risk of perinatal morbidity and mortality, and during childhood according to the article by Slaughter et al (2009). Additionally, Pringsheim et al (2009) proved that the intrauterine environment affects the health of an individual not only in fetal life, but also throughout postnatal life.
Fetal weight increases with gestational age, as was shown in the articles of Pacora et al (2005) and Montoya-Restrepo and Correa-Morales (2007), adjusted to the normal pattern of growth that occurs in three stages. 95% of fetal weight gain occurs during the last 20 weeks of gestation, and Albaigés (2004) described that from 28 weeks occurs a rapid increase in cell size with a peak velocity between 33-35 weeks of gestation, is accumulated the fat, muscle and connective tissue quickly, and it is precisely when a greater increase in fetal weight shows up.
A proper fetal weight gain in the critical period of weight gain by the fetus should guarantee a good birthweight. For this to occur there must be a very low level of exposure to various factors that can negatively influence fetal weight gain and / or powerful and efficient adaptive protection mechanisms to counteract its harmful effects.
Pregnant women may be exposed regularly to various conditions that lead to a state of oxidative stress. In pregnant women, oxidative stress can be produced by an unregulated production of reactive species, by continued exposure to the same or a deficiency in antioxidant systems, as was discussed by Young-Ju et al (2005).
Monitoring redox status indicators in pregnant women are important to understand the relationship between oxidative stress and the product of pregnancy, particularly the fetal growth.
The effect of maternal oxidative stress on fetal growth has not been examined extensively, although recent evidence presented by Young-Ju et al (2005), Saker et al (2008) and Al-Gubory et al (2010) shows that maternal or fetal oxidative stress plays an important role in the pathophysiology of low birthweight. However, the studies reviewed have limitations as indicators of redox status were measured at birth, which does not necessarily reflect the exposure of pregnant women to the conditions that lead to oxidative stress throughout pregnancy, even during the critical period of weight gain by the fetus.
In a previous study done by the present authors (Corría et al (2011)) to determine the influence of maternal redox status between 30 and 36 weeks of gestation, a direct correlation between birthweight and activity of extracellular superoxide dismutase (ecSOD) and the concentration of erythrocyte reduced glutathione (eGSH) was found. Besides, it was disclosed that the association between the concentration of eGSH and Ferric Reduction Potential (FRP) and ecSOD activity were also correlated with birthweight from which, it was concluded that the antioxidant status had a positive influence on birthweight.
In order to determine if the behavior of redox status indicators and their relation to birthweight were dependent on the gestational period analyzed, or whether it was a characteristic of the third trimester, critical period of weight gain by the fetus, the present study was carried out in pregnant women between 25 and 29 and between 30 and 36 weeks of gestation.
Research Design and Methodology
Patients
The study included 65 pregnant women who received prenatal care and 40 nonpregnant women of childbearing age pertaining to the clinics’s health areas from Bayamo city, Granma, Cuba. Each patient gave a written consent, and the study was approved by the Committee for Human Research at the University of Medical Sciences. The selected pregnant women had a single fetus. Pregnant women, who had a fetus diagnosed with congenital anomalies, were not included. Nonpregnant women had no history of chronic disease; they had not toxic habits and were clinically healthy at the time of the study.
Two groups were formed according to gestational age and a third (reference) group was composed of women of childbearing age, not pregnant. Group 1 included patients with gestational age between 25 and 29 weeks, and the second group included pregnant between 30 and 36 weeks. Gestational age was estimated based on the date of the last menstrual period and confirmed by first and second trimester ultrasonography. Fetal weight was determined by ultrasound to pregnant women at the time of the study. The weight of the infants was measured immediately after birth.
Blood Sampling and Analysis
Maternal fasting blood samples were obtained from veins in the upper extremity. Blood samples were collected in test tubes and centrifuged to isolate serum. Serum antioxidant capacity was determined by the ferric reducing potential value (FRP). The serum malondialdehyde plus 4-hydroxynonenal (MDA + 4HDA), uric acid, albumin, vitamins C, E, A concentration and extracellular superoxide dismutase (ec-SOD, EC.1.15.1.1) and catalase (sCAT 1.11.1.6) activity were determined. The concentration of reduced glutathione was determined in erythrocytes (eGSH).
FRP Quantification
Ferric reducing potential (FRP) was determined by the modified colorimetric assay presented by Bahr and Basulto (2004). This method measures the total reducing power attributable to antioxidant species which are present in serum or plasma, based on the final concentration of Fe2+ (ferrous) ions formed from Fe3+ (ferric) reduction, using potassium ferricyanide as chromogen substance. Fe2+ reacts with potassiun ferricyanide reagent to form a blue compound that absorbs light at 720 nm. FRP was expressed in μmol Fe2+/L.
Uric Acid Quantification
Uric acid concentrations were measured by the enzymatic method described by Gochman and Schmitz (1971), which uses urate oxidase-peroxidase system. The chromogenic product of the system action was quantified spectrophotometrically at 550 nm. The Urate-Monotest Kits (HELFA® DIAGNÓSTICO, Quimefa, Cuba) were used. Values were expressed in μmol/L.
Albumin Quantification
Serum albumin concentrations were measured by bromocresol green method described by Thomas (1998). The chromogenic product of the reaction was quantified spectrophotometrically at 628 nm. The Albumin Kits (HELFA® DIAGNÓSTICO, Quimefa, Cuba) were used. Values were expressed in g/L.
Determination of Serum Levels of Antioxidants Vitamins
Vitamin C levels were measured by dinitrophenylhydrazine method described by Nino and Shaw (1982). Serum α-tocopherol (vitamin E) and retinol (vitamin A) were determined by reverse phase HPLC, and detected by an UV detector at 292 nm for vitamin E and 325 nm for vitamin A according to the method of Thurham et al (1988). Values were expressed in μmol/L.
Determinations of Serum Antioxidant Enzyme Activities
Total superoxide dismutase SOD activity was determined by the indirect method presented by Marklund and Marklund (1974), which is based on the ability of this enzyme to inhibit auto-oxidation reactions of pyrogallol, auto-oxidation reactions that spread by superoxide radicals formed. Pyrogallol oxidized forms absorb light at 420 nm. One unit (U) of SOD activity was taken as the amount of enzyme capable of inhibiting by 50%, the pyrogallol auto-oxidation reaction at 25º C and pH 8.20. The activity was expressed in U.mL-1 min-1. Catalase (CAT EC 1.11.1.6) activity was measured by spectrophotometric analysis of the rate of hydrogen peroxide decomposition at 230 nm, following the method used by Taysi et al (2002). The activity was expressed in UI.mL-1 min-1.
Serum MDA + Alkenals Quantification
Malondialdehyde plus 4- hidroxynonenals (MDA + 4HDA) were measured by the espectrofotometric method presented in the article by Esterbauer and Cheeseman (1990), which is based on the reaction of aldehydes with N-methyl-2-phenylindole at 45ºC to form an intensely colored carbocyanine dye that absorbs at 586 nm. Values were expressed in μmol/L.
GSH Quantification
Erythrocyte reduced glutathione (eGSH) concentrations were measured by the Beutler colorimetric method. This method is based on the reaction of the reduced glutathione with DTNB (5,5-dithiobis (nitrobenzoic acid)) reagent. Absorbance readings were done at 412nm the first 5min, as was recommended in the article by Beutler et al (1963). Values were expressed in μmol/gHb.
Statistical Analysis
Statistical analysis of the data was carried out using STATISTICA (version 4.1, Statsoft, Tulsa, OK) and GraphPad Prism (version 5.03, COMP, PAIS). Shapiro-Wilk test was used to assess the normality of continuous data. Results are given as means ± SEM, or median (25-75 percentile intervals) when the data were not normally distributed. Student t test was used for data that passes the normality, and Mann—Whitney U test was used for data that failed the normality. Dunn’s test was used for multiple comparisons. Spearman’s correlation was applied to study the correlation between quantitative variables. Statistical significance was accepted at p < .05 for all comparisons and in all correlations.
Results
Demographic Characteristics
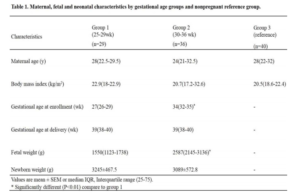
Maternal, fetal and neonate characteristics by gestational age groups and nonpregnant group are shown in Table 1. Three groups were then selected and studied: Group 1 consisted of 29 mothers with gestational age at enrollment between 25 and 29 weeks; group 2 consisted of 36 mothers with gestational age between 30 and 36 weeks; group 3 included 40 nonpregnant women. The three groups were similar with respect to age and body mass index (BMI) before conception. Then, the groups 1 and 2 were similar respect to newborn weight and duration of pregnancy. There were no significant differences among the groups.
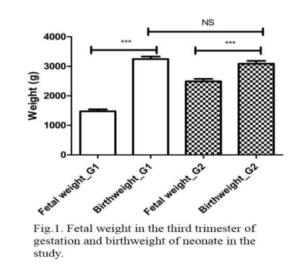
It has been shown that was greater increase in fetal weight in the third trimester of pregnancy. In the works by Gairdner and Pearson (1971), Fernández et al (2008) and Montoya-Restrepo and Correa-Morales (2007) was observed in weight gain curves according to the weeks of gestation an increase from week 25, which becomes more marked from week 30.
In this study we have tried to show whether the influence of maternal redox status on birth weight reported in a previous study by us in pregnant women between 30 and 36 weeks (Corría et al (2011)) was a characteristic only of this period, or if it was similar in the initial period of the third trimester.
These results show that maternal redox status does not change significantly with gestational age groups between 25 and 29 and the 30 to 36 weeks. The lower statistical difference in the levels of FRP and albumin concentrations in the groups of pregnant women compared with nonpregnant found in this study are consistent with the results reported by Hung et al (2010). Such results may be due to differences in diet. Serum antioxidant capacity is mainly attributable to uric acid, the protein thiol groups, ascorbic acid, vitamin E and bilirubin, as it was stated in the book edited by Halliwell and Gutteridge (1999). The amount of uric acid and vitamins C and E is greatly influenced by diet, as was discussed by Pus et al (2013). Likewise, serum albumin concentration has been associated with the consumption of protein and is an indicator of protein status, according to Seres (2005).
The statistically significant increase in the activity of serum ecSOD and erythrocyte GSH concentration found in both groups of pregnant women compared to nonpregnant group is further evidence to the a study previously done on the increased activity of this enzyme and erythrocyte GSH concentration in pregnant women between 30 and 36 weeks of gestation (Cruz et al (2010)). The results for the ecSOD differ from those found by Kharb (2012). The studies of Hung et al (2010) and Leal et al (2011) have reported an increase in SOD activity whole blood measured in pregnant women in the third trimester compared to nonpregnant women.
Taking into account that there was no significant difference in the levels of lipid peroxidation indicators with respect to non pregnant; results were interpreted as a consequence of the regulatory mechanisms of redox state, as was discussed by the present author in a previous paper (Cruz et al (2010)). The advantages for the gestation of an increase in activity ecSOD and GSH levels are extensively described by Biondi et al (2005), Valko et al (2007), Filomena et al (2008), and others.
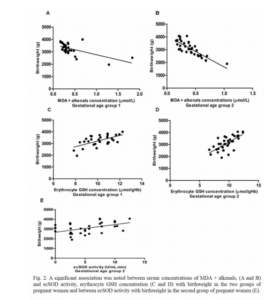
The results of the study concerning the influence of maternal redox status on birthweight between 25 and 29 weeks gestation compared with the period between 30 and 36 are very similar; however, the activity of the ecSOD in the group of pregnant women between 25 and 29 weeks did not correlate with birthweight. The significant direct correlation of ecSOD activity and erythrocyte GSH concentration with birthweight in pregnant group between 30 and 36 weeks of gestation confirm our previous results (Corría et al (2011)).
The significant inverse correlation between the levels of lipoperoxidation indicators and birthweight in the group of pregnant women between 30 and 36 weeks is a new result that has not been found in previous study with the determination of the concentration of MDA, and it was also observed in this study in the group of pregnant women between 25 and 29 weeks. Young et al (2005) also found significant inverse correlation of MDA concentrations with birth weight.
The authors of this research strongly believe that these results are consistent with the evidence reported on the regulation of maternal redox status and its influence on birthweight, which has been discussed in our previous papers (Corría et al (2011), Cruz et al (2012)). The results are also supported by the findings of Saker et al (2008) and Hsieh et al (2012) who have shown that the oxidant and antioxidant status of the mother is related to the small neonates and appropriate for gestational age.
A new evidence was brought in to light that oxidant and antioxidant status in the third trimester of pregnancy is related to birthweight. A good antioxidant state ensures a good birthweight; however, an increase of lipid peroxidation may affect birthweight.
Conflict of Interest Statement
The authors declare that there are no conflicts of interest regarding this work.
Acknowledgments
We would like to acknowledge Dr.Sc. Consuelo Macías Matos, Dra. Yeneisy Lanyau Domínguez, Technical Dania Herrera Javier and Technical María Eugenia Quintero Alejo from Nutrition and Food Hygiene Institute for their contribution to the determination of vitamins A, E and C. We would also like to thank Angel Vega at University of Granma, Orlando Luciano Rojas, Professor at Bayamo Medical School and David Del Llano Sosa, EFL Associate Professor at Bayamo Medical School for helping us in the translation of this article.
References
1. Albaigés, G. (2004) “Frontera entre feto pequeño para edad gestacional y restricción de crecimiento intrauterino”, Ginecología y Obstetricia Clínica, 5 (1) 8-21.
2. Al-Gubory, K., Fowler P. and Garrel, C. (2010) “The roles of cellular reactive oxygen species, oxidative stress and antioxidants in pregnancy outcomes”, Intenational Journal of Biochemestry and Cell Biology, 42 (10) 1634—50.
Publisher – Google Scholar
3. Bahr, P. and Basulto, Y. (2004) “El Potencial Reductor Férrico (FRAP). Un ensayo para evaluar la capacidad antioxidante en suero”, Revista Correo Científico Médico Holguín, 8 (4) 12-16.
4. Beutler, E., Duron, O. and Nelly, B. (1963) “Improved methods for determination of blood glutathione”, Journal of Laboratory Clinical Medicine, 61 882—90.
5. Biondi, C., Pavan, B., Lunghi, L., Fiorini, S. and Vesce, F. (2005) “The role and modulation of the oxidative balance in pregnancy”, Current Pharmaceutical Design, 11 2075-89.
Publisher – Google Scholar
6. Corría, J.; Cruz, E.; Milanés, M.; Ramírez, Y.; Sierra, M.; Cruz, M. and Sanfiel, L. (2011) “Influence of maternal redox status on birth weight”, Reproductive Toxicology, 31 (1) 35—40.
Publisher – Google Scholar
7. Cruz, E., Corría, J., Milanés, M., Ramírez, Y., Sierra, M. and Cruz M. (2010) “Evidencias de la regulación del estado redox materno entre las 30 y 36 semanas de gestación”, Revista Cubana de Obstetricia y Ginecología, 36 (4) 519-531.
Google Scholar
8. Esterbauer, H. and Cheeseman, K. H. (1990) “Determination of aldehydic lípid peroxidation products: malonaldehyde and 4-hydroxynonenal”, Methods in Enzymology, 186 407-421.
9. Fernández-Massó, J. R., Silva-Leal, N., Santana-Porbén, S., Bustamante-Frandenthaler, L. and Pérez García, J. (2008) “Peso en la captación del embarazo y ganancia de peso durante la gestación: impacto sobre el peso del recién nacido”, Revista Cubana de Alimentación y Nutrición, 18 (2) 186-203.
10.Filomena, G., Ottaviano, B. S., Handy, D. E. and Loscalzo, J. (2008) “Redox Regulation in the Extracellular Environment”, Circulation Journal, 72 (January) 1-16.
Google Scholar
11. Gairdner, D. and Pearson, J. (1971) “A growth chart for premature and other infants”, Archivements in Dissease of Childrens, 46 783-7.
Publisher – Google Scholar
12. Gochman, N. and Schmitz, J. M. (1971)) “Automated determination of uric acid with use of a uricase-peroxidase system”, Clinical Chemestry, 17 1154.
Google Scholar
13. Halliwell, B. and Gutteridge, M. C. (1999) “Antioxidant defences”, Free Radicals in Biology and Medicine, Halliwell, B. and Gutteridge, M. C. (eds), Oxford University Press, Oxford, USA.
Google Scholar
14. Hsieh, T. T., Chen, S. F., Lo, L. M., Li, M. J., Yeh, Y. L. and Hung, T. H. (2012) “The association between maternal oxidative stress at mid-gestation and subsequent pregnancy complications”, Reproductive Sciences, 19 (5) 505-12.
Publisher – Google Scholar
15. Hung, T. H., Liang-Ming, L., Tsung-Hong, C., Meng-Jen, L., Yi-Lin, Y., Szu-Fu, C. and T’sang-T’ang, H. (2010) “A Longitudinal Study of Oxidative Stress and Antioxidant Status in Women With Uncomplicated Pregnancies Throughout Gestation”, Reproductive Sciences, 17 (4) 401-409.
Publisher – Google Scholar
16. Kharb, S. (2012) “Extracellular superoxide dysmutase levels in preclampsia”, Mediterranial Journal of Nutrition and Metabolism, 5 (1) 53-55.
Publisher – Google Scholar
17. Leal, C., Schetinger, M., Leal, D., Morsch, V., Schafer-da-Silva, A., Rezer, J., Valle-de-Bairros, A. and Jaques, J. (2011) “Oxidative stress and antioxidant defenses in pregnant women”. Redox Report, 16 (6) 230-236.
Publisher – Google Scholar
18. Marklund, S. and Marklund, G. (1974) “Involvement of the superoxide anion radical in the autoxidation of pyrogallol as a convenient assay for superoxide dismutase”, European Journal of Biochemestry, 47 469 74.
Google Scholar
19. Montoya-Restrepo, N. and Correa-Morales, J. (2007) “Curvas de Peso al Nacer”, Revista de Salud Pública, 9 (1) 1-10.
Publisher – Google Scholar
20. Nino, H. V. and Shaw, W. (1982) “Vitamins”, Fundamental of clinical chemistry, Tietz, N. W. (ed), WB Saunders Co., Philadelphia, USA.
21. Pacora, P., Buzzio, Y. and Ingar, W. (2005) “El peso del recién nacido sano según edad gestacional en una población de Lima”, Annales de la Facultad de Medicina, 66 (3) 212-217.
Google Scholar
22. Pringsheim, T., Sandor, P., Lang, A., Shah, P. and O’Connor, P. (2009) “Prenatal and perinatal porbidity in children with Tourette Syndrome and Attention-Deficit Hyperactivity Disorder”, Journal of Development Behavior Pediatry, 30 (2) 115-21.
Publisher – Google Scholar
23. Pus, T., Podgrajsek, K., Fidlermis, N., Benedik, E., Rogelj, I. and Simcic, M. (2013) “Vitamin and mineral Nutritional status of healthy pregnant women improved by nutrient suplement”, Acta Argiculturae Slovenica, 102 (1) 47-54.
24. Saker, M., Mokhtari, N. S., Merzouk, S. A., Merzouk, H., Belarbi, B. and Narce, M. (2008) “Oxidant and antioxidant status in mothers and their newborns according to birth weight”, European Journal of Obstetrics, Gynecology and Reproductive Biology, 141 95—9.
Publisher – Google Scholar
25. Seres, D. S. (2005) “Surrogate nutrition markers, malnutrition and adequacy of nutrition suports”, Nutrition in Clinical Practice, 20 308-313.
Publisher – Google Scholar
26. Slaughter, J. C., Herring, A. H. and Thorp, J. M. (2009) “A Bayesian Latent Variable Mixture Model for Longitudinal Fetal Growth”, Biometrics, 65 1233—1242.
Publisher – Google Scholar
27. Taysi, S., Polat, F., Gul, M., Sari, R. A. and Bakan, E. (2002) “Lipid peroxidation, some extracellular antioxidants, and antioxidants enzymes in serum of patients with rheumatoid arthritis”, Rheumatology International, 21 200-204.
Publisher – Google Scholar
28. Thomas, L. (1998) Albumine. Clinical Laboratory Diagnostics. Use and assessment of Clinical Laboratory Results, TH-Books, Main, Frankfurt, Germany.
29. Thurham, D. I., Smith, E., Flora and S. P. (1998) “Concurrent Liquid-Chromatographic Assay of Retinol, alpha-Tocopherol, beta-Carotene, alfa-Carotene, Lycopene, and beta-Cryptoxantin in Plasma with Tocopherol Acetate as Internal Standart”, Clinical Chemestry, 34 377-381.
30. Valko, M., Leibfritz, D., Moncol, J., Cronin, M. T. D., Mazur, M. and Telser, J. (2007) “Free radicals and antioxidants in normal physiological functions and human disease”, The International Journal of Biochemistry & Cell Biology, 39 44-84.
Publisher – Google Scholar
31. Young-Ju, K., Yun-Chul, H., Kwan-Hee, L., Heon Joo, P., Eun Ae, P., Hye- Sung, M. and Eun-Hee, H. (2005) “Oxidative stress in pregnant women and birth weight reduction”, Reproductive Toxicology, 19 487—492.
Publisher – Google Scholar