Introduction
While the city is facing a power blackout, a hospital switches on to its emergency backup generator. Large engines, up to 15 metres tall, power ships and liners along the high seas. At the same time, shovels and drills excavate natural resources like precious metals, iron ore, oil and coal, and load them onto enormous mining trucks or conveyer belts. What is common to these three scenarios is the source of energy that drives these activities, diesel. This fuel is derived from petroleum, one of the most ubiquitously used fossil fuels, the others being coal and oil.
With increasing industrialization and urbanization, the demand for such resources, which take millions of years to form, is only going to increase. However, their supply is limited. Coal, oil and natural gas have only 119, 45.7 and 62.8 years of production left respectively, even with the most optimistic proved reserve estimates [1]. In addition to this, there is the emerging problem of pollution and global warming caused by greenhouse gases released on using these fuels. Every year, around 21.3 billion tonnes of carbon dioxide is produced because of the burning of fossil fuels. But, only about half of this amount can be absorbed and done away with by natural processes [2]. In a world so dependent on fossil fuels, the combined effect of the two problems stated poses a threat to the very survival of the civilization. The development of alternate sources of energy is a goal of paramount importance for scientists in the worldwide.
The widespread use of diesel stems from the fact that it is highly cost effective. Diesel has low volatility, thus is safer to use, and also provides more energy per unit consumption, making it a fuel of choice in most industrial sectors [3]. While petroleum diesel is being used generally, other sources of diesel can be exploited and developed, and that could tackle the dual problem of shortage and environmental damage. Biodiesel provides a biofriendly and easy-to-extract alternative to petrodiesel. It is a diesel fuel made up of long-chain alkyl esters and is produced from vegetable oil or animal fat [4]. What makes it special is the fact that its source is present everywhere, farms, fields, or even your kitchen.
For achieving combustion properties similar to petroleum diesel, so as to make it an effective replacement to diesel, vegetable oil or animal fat needs to be processed to convert it into mono-alkyl esters of long chain fatty acids [5]. Biodiesel is typically made by chemically reacting lipids ,which are present in vegetable oil or animal fat, with an alcohol ,generally methanol, and thus producing fatty acid esters (called FAME- Fatty Acid Methyl Esters) , in a process called transesterification. In the industry, it is usually done in huge plants using alkali or acid catalysts [6].
Even though widely used, this operation is beset with many problems. The transesterification reaction produces crude glycerol as a by-product which is of a little market value. During alkali catalysed transesterification, the production of soap is a major cause of contamination. Wastewater release and treatment is also a major issue [7]. Such conditions require scientists to look beyond conventional methods of production of unconventional fuels.
Enzymatic transesterification is an inspiring method, since the enzyme to carry out this reaction is present in all animals and most micro-organisms for the breakdown of excess lipids, and is therefore called the lipase. Lipases under physiological conditions catalyse the hydrolysis of the ester bonds in triglycerides, and liberate free fatty acids and diglycerides, monoglycerides and glycerol. In low water content environment, they catalyse the reverse reaction of transesterification and accept a wide range of acyl donors and nucleophiles besides their natural substrates [8]. Scientists worldwide have used lipases from various sources to produce biodiesel with efficiency as high as 98% .However, in spite of the amazing results achieved, enzymatic transesterification with crude lipases is not being used industrially owing to high cost and inefficiency. Immobilizing or trapping lipases onto a suitable medium provides a simple solution to overcome these inefficiencies [9]. In fact, it makes it possible for a molecule, so crucial in an organism’s life to work on a large scale as a device to overcome fuel shortage in the world.
This article begins with a description of the structure of the lipase and the mechanism of transesterification employed by it. From this premise, the article builds a discussion on the means by which the lipase is immobilized in different techniques. How these means affect the stability, enzyme activity and efficiency of immobilization is also discussed at length. Thus, this article adds to the literature by consolidating and detailing the various immobilization methods from the perspective of molecule-level interaction between the support and the enzyme.
Need for Immobilization
Using immobilized lipases has multi-fold advantages over using them in their powdered form. Because the powdered enzyme forms an emulsion, the extraction and reuse of the enzyme is difficult, and hence a lot of potential enzyme is wasted [10]. Crude enzyme extract has proteins other than the lipase, which increase the reaction volume and reduce the efficiency. By immobilizing enzyme with a suitable method, it can be separated from contaminating proteins [11]. Immobilization is associated with a change in conformation, and by clever use of technique, the lipase can be stabilized in an active conformation, which increases its thermal and ionic stability over the course of the reaction, while also increasing its efficiency [12]. It is also easier to control reaction parameters like flow rate and accessibility of substrates when the enzyme is immobilized [13]. With a suitable technique, the cost-effectiveness of the process can be increased, as wastage is significantly reduced. By creating reactors made up of immobilized enzyme, biodiesel production can be carried out at a large scale at the industry level.
How the Lipase Works
Understanding the structure and functioning of lipase is essential if we wish to process it to a more usable form. Lipase breaks down fats and lipids in the presence of water in a reaction called hydrolysis. But in a microenvironment with low water content and presence of alcohol, it accepts alcohol as a substrate and produces biodiesel in a bi-bi ping pong reaction [9].
A fantastic thing about this enzyme is that while its substrates are hydrophobic lipids, it is itself soluble in water. This means that the actual catalytic site is hidden within the structure of this monomeric protein which is exposed at the lipid-water interface. This feature is called interfacial activation. This probable mechanism also hints at the presence of a cap or a lid which when closed covers the catalytic site (the triad of serine, histidine and aspartic acid) making the enzyme inactive but in the presence of the interface, opens to reveal the active site, a hypothesis that has been proven [14]. This type of structure can be imagined to be like the character of the popular arcade game Pacman, and is in fact often depicted in a similar fashion.
With this knowledge, we can move ahead and explore the various options available for immobilization of this enzyme, the prominent among them being physical adsorption, covalent attachment, entrapment, or forming cross-linked enzyme aggregates or whole cell biocatalysts.
Physical Adsorption
The easiest way to link the lipase is to let it adsorb to a solid support via the non-permanent joins like hydrogen bonds, salt linkages, or Van der Waal’s forces. Because of the nature of these joins, this process is carried out in mild conditions. The chemical nature of the support affects how well the lipase is loaded onto the support [15] and whether it is in its active state or not. Enzyme loading also depends on the particle size, porosity of support and its surface area [11].
Active lipase has been achieved by adsorption on Accurel (polypropylene), polypropylene-silica, activated carbon, polymethylacrylate, organosillicates, etc [8].
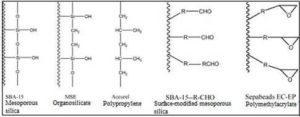
Figure 1: Functional Groups Present on the Surface of Various Supports for Physical Adsorption
Usually a hydrophobic support is used. The logic behind this is that hydrophobic supports resemble the surface of the drops of the natural substrates as they have a low ionic strength, allowing the lipase to attach easily on these supports. Lipase adsorption involves hydrophobic areas surrounding the active centre, and the internal face of the lid. This way, the lipase is fixed in its active form and the active centre is made easily accessible to small substrates [12]. The role of hydrophobic interactions was proven in a study in which, the enzyme loading onto a hydrophobic support at different pH was recorded, and similar results were achieved at different pH conditions [11].
Adsorption isotherms of lipase adsorption are graphs depicting enzyme loading with respect to the concentration of lipase residual in solution. Adsorption isotherms describe the support affinity, while the isotherm shape illustrates the lipase distribution on the support surface [15]. Hence, these isotherms are an important tool in understanding the suitability of a particular lipase to a particular support.
This approach was used to gauge the adsorption characteristics of different lipases on hydrophobic and hydrophobic-hydrophilic supports. On hydrophobic supports, like Accurel, the adsorption isotherms suggest a monolayer formation and a homogeneous mode of adsorption. However, hydrophobic-hydrophilic supports like 45SAA (a polypropylene-silica composite) show a 2-step adsorption isotherm. Earlier studies have suggested that a 2-step isotherm indicates different modes of adsorption, one in the native configuration and the other in an unfolded conformation. The study suggested that because of the hydrophobic part of 45SAA, the lipase is loaded in an unfolded, active conformation but because of the hydrophilic part, lipase clusters by means of hydrophilic residues on the surface of the lipase, are formed as an inactive complex. Thus, 45SAA showed greater enzyme loading. However, its activity and efficiency were less than that on Accurel. The burying of enzyme inside the inactive clusters is understood to be the reason behind this [15].

Figure 2: (A) Adsorption Isotherm for Lipase Adsorption on Accurel EP 100,
(B) Adsorption Isotherm for Lipase Adsorption on 45SAA
The amount of lipase adsorbed depends on the porosity. Usually mesoporous and macroporous supports are used, as larger diameter of pores leads to more diffusion of substrates. It’s commonly accepted that pore diameter should be greater than 350Šis suitable for penetration of lipase [34]. In a study [16], Candida antarctica lipase B (CALB) was adsorbed onto active carbon, which is microporous and has a high surface area. Despite the high surface area of the support, little of the enzyme was available for lipase immobilization since the mean pore diameter of the support was less than the size of the CALB molecule. This indicates that, the enzyme was immobilized on the superficial area and was susceptible to desorption, which is also a major drawback of adsorption.
In adsorption, the enzyme is spread on the surface of the support and no aggregates are formed, so there is an increased substrate access [10]. An improvement in thermal stability and efficiency is observed in most studies [8].
During the process of adsorption, slower and more progressive adsorption of lipase takes place by gradually displacing other proteins from the crude extract, which have lower affinity for the support [34]. Contaminating proteins or salts like ammonium sulphate have been shown to increase the enzyme activity. In presence of inert proteins, the high affinity to the support is blocked, preventing it from unfolding and hence stabilizing it [16]. However, the need for such proteins defeats the primary aim of reducing reaction volume and hence increasing the efficiency, thus making it a significant disadvantage.
In spite of certain drawbacks, physical adsorption is a very popular technique given the fact that it is easy, cheap and requires only mild chemical conditions. However, finding the right combination of support and lipase remains a challenge, usually tackled using hit-and-trial methods, and thus keeping the innovation of immobilization in the research stage only. This issue can be dealt with emerging technologies, which would be discussed later in this paper.
Covalent Attachment
Covalent binding is a more permanent and a stronger linkage between the lipase and the immobilization support. Successful covalent attachment of the lipase has been achieved on high internal phase emulsion styrene-divinylbenzene copolymer (polyHIPE), poly (acrylonitrile-co-2-methylacryloxyethyl) or PANCHEMA, polysiloxane-polyvinyl alcohol (POS-PVA), silica nanoparticles coated with glycidyl methyl acrylate (GMA), etc [8].
Covalent attachment of the enzyme with the support takes place as a result of a chemical reaction between the active amino acid residues outside the active catalytic and binding site of the enzyme, and the active functionalities of the carrier [8]. This requires treatment of the support with coupling agents required for activation of support. Support activation generates high concentration of aldehyde groups on the support surface, which can easily link with amine groups present on lysine residues of the enzyme [17].
For activation for the covalent attachment of lipase, the support is treated with coupling agents. The commonly used coupling agents are (i) glutaraldehyde, which has 2 aldehyde groups to bind with amine groups, and thus there is a need to minify the support before activation [18], (ii) epichlorohydrin, which can bind with amino, hydroxyl and sulfhydryl groups present on the surface of the lipase [13].
A multi-point covalent attachment takes place, rigidifying the enzyme [19]. This reduces spontaneous conformational change and increases the pH stability, thermal stability and reusability of the enzyme as seen by broader pH and temperature profiles. This method is the most effective one with the view of operational stabilization [20].
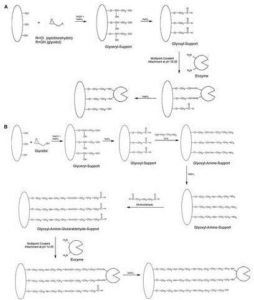
Figure 3: Mechanism of Multipoint Covalent Attachment of Enzyme on (A) Glyoxyl and (B) Glyoxyl-Amine-Glutaraldehyde Supports
Source: http://dx.doi.org/10.1016/j.molcatb.2010.10.002
With covalent attachment, the length of the spacer arm of the coupling molecule between the support and the enzyme plays a crucial role. The length can be manipulated by means of a series of chemical reactions. A long spacer arm length is desired, for the following advantages it provides (i) it prevents reaction between the active site of lipase and the support itself, (ii) it prevents the denaturation of lipase, (iii) it enhances substrate accessibility [13] and (iv) it reduces steric hindrance, and hence increases the loading capacity of the support [17].
Careful manipulation of the support as well as the enzyme can lead to a better efficiency of loading. Svendsen et al. identified lysine residues on the surface of various lipases. They then utilized genetic engineering to remove all residues close to the active site. For the Thermomyces lanuginosa lipase, only one residue diametrically opposite to the active site was left. This residue was used for selective covalent immobilization. There was greater enzyme loading as multiple enzymes surrounded the support particle, and all the active sites were oriented towards the reaction mixture, leading to a higher activity [21]. This suggests that, the knowledge of the lipase structure can lead to a better judgment of immobilization parameters, and tweaking around with it can give wondrous results.
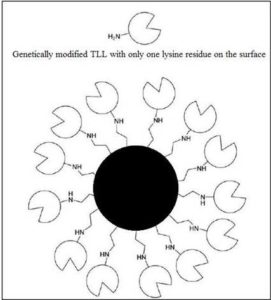
Figure 4: Modified TLL with Just One Amino Group on its Surface is Ideally Oriented When Covalently Immobilized
Although, multi-point attachment of enzyme to the support enhances the stability and reusability of lipase, this technique comes with its own flaws. Enzyme activity is reduced as for multipoint attachment to take place via a covalent bond, there is bound to be some structural distortion. Support activation is done with chemicals which make a harsh environment, and keeping the structural integrity of the enzyme is a challenge in such a condition. Also, Al-Khatib et al. found that the amount of loading was less because of the alteration of membrane porous structure [22]. This indicates that the use of covalent attachment is basically a trade-off between greater strength and greater efficiency.
Entrapment/Encapsulation
When we discuss immobilizing an enzyme, the technique which comes to mind as a first instinct is trapping the enzyme in a mesh of some sort. Indeed, it is a popular method, albeit one which still needs to be perfected because of the tricky processes involved. Lipase has been entrapped in agarose, alginate, chitosan, cellulose, polyethersulfone, κ-carrageenan, etc. [8] Lipase entrapped in beads can be used in fixed bed reactors to produce biodiesel on a large scale [22].
Entrapment involves capture of the enzyme within a matrix of a polymer. The matrix is usually formed during immobilization itself in a sol-gel process [23].This process is generally used to fabricate ceramic and glassy materials. It is a chemical technique, which involves the progressive evolution of the sol, or the solution, to a gel-like network which has both solid and liquid phases. The process requires curing, i.e., hardening of a polymer material by cross-linking of polymer chains, with the enzyme trapped between them.
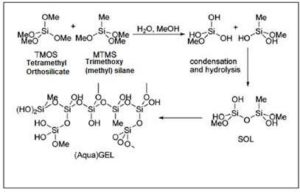
Figure 5: Synthesis of Sol Gels
The method of entrapment and the kind of support material are very crucial to successful entrapment of the enzyme. The choice of a good support material includes its strong stable gel binding ability, compatibility with the enzyme and high gel loading efficiency. A base-catalyzed entrapment causes heat generation which could lead to the denaturation of the enzyme. A non-shrinkable sol-gel is required since shrinking damages the enzyme molecular structure [23]. When trapping lipase in agarose, undesirable swelling and disintegration of the gel takes place, damaging the enzyme. Such a combination is not suitable for entrapment [24].
One would think that the enzyme will be secure in the network of the polymers. However, the case is not so. There is a great possibility for the loss of the enzyme in the various processes involving entrapment. The immobilized enzyme, when stored without any medium of suspension shows higher stability than when stored in distilled water, indicating that the enzyme slowly leaches out from the support into the aqueous medium [22]. During freeze-drying of the gel, freezing of water might lead to lipase remaining on the surface of beads, and thus being more susceptible to loss. Freeze drying also makes the beads more friable [25].
Increasing the polymer content of the gel led to increased entrapment but lowered activity. Because of the increase in polymer density, there is a reduced diffusivity of substrate and product. Enzyme aggregation results in poor dispersion [25]. A good dispersion of the gel within the support is important to preserve the accessibility of lipase molecules, even if we assume that it is in its active state. In most entrapment techniques, the lipase is entrapped with low efficiency. They frequently require the use of volatile organic solvents, which may not only disrupt the enzyme structure, but also be potentially hazardous to the environment.
While, entrapment shows a better immobilization yield and also facilitates a large scale production of biodiesel, if the gel beads are used in a fixed bed reactor, it has manifold drawbacks. There is a great chance of leaching away of the enzyme and the loss of structure due to the harsh microenvironment. This technique doesn’t allow multiple reuses [25]. However, it is suggested that this can be improved by coating the surface of the beads by other polymeric materials.
Cross-Linked Enzyme Aggregates
The aforementioned techniques involved a lot of complex processes involving multiple steps. Cross-linked Enzyme Aggregates are the result of a highly simplified single step method to immobilize the lipase. CLEAs are formed by chemically cross-linking physical enzyme aggregates, by precipitating them with inorganic salts or organic solvents [26]. The enzyme is precipitated with the help of inorganic salts like ammonium sulphate. They compress the solvation layer and increase protein-protein interaction. These precipitated aggregates are then cross-linked with the help of agents like glutaraldehyde, which is a bifunctional reactive agent, capable of reacting with the surface amino groups of enzymes [27, 28].
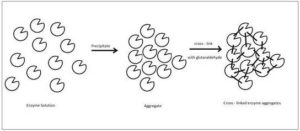
Figure 6: Aggregation and Cross-Linking of an Enzyme to Prepare a CLEA
The fraction of ammonium sulphate affects the particle size of the aggregate. Also, increased amounts of enzyme and glutaraldehyde increase particle size. The particle size is important, as it influences the accessibility of the inner enzymes to the substrates in the reaction mixture [27]. Thus, greater particle size leads to wasted lipase activity.
The activation of lipases by additives like surfactants (SDS, Triton-X, Tween 80), crown ethers or amines show better activity. This has been attributed to the lipase adopting a more active conformation under these conditions [28].
CLEAs are novel in the sense that a carrier-free enzyme immobilization takes place. This leads to a highly concentrated enzyme activity, high stability and low production cost due to the lack of additional carrier [27]. It is a one step purification and immobilization process, making it responsive to rapid optimization, which translates to low cost and short time-to-market process [28]. These benefits of cross-linking, however, do not overshadow the fact that when the particle size is large, a significant amount of lipase is buried inside, showing low activity and efficiency. Enzymatic activity is especially less when the substrate size is close to the particle size [26]. These features render it quite a wasteful technique.
Whole-Cell Biocatalyst
So far, the immobilization of lipase extracted from different organisms has been considered. But, scientists are also exploring the possibility of getting complete, immobilized cells to produce biodiesel. Whole cell biocatalysts are formed when the cells of organisms are immobilized, instead of the enzyme itself. The technological and economic advantages of immobilized cells over immobilized enzymes are well known: higher operational stability, higher yields of enzyme activity after immobilization, greater resistance to environmental perturbations, greater potential for multistep processes, and lower effective enzyme cost (enzyme purification and extraction are avoided) [8].
Fukuda et al. compared Novozym, a commercially available lipase, with Rhizopus oryzae cells immobilized on polyurethane foam [29]. They concluded that ROL was better than Novozym and no significant differences in reusability were observed. ROL was more susceptible to be poisoned by methanol. It is suggested that the support for ROL adsorbs more methanol than acrylic resin, so activity decreases. However, the choice of support can significantly alter the results.
The presence of water improved the activity of ROL but not Novozym, but only up to a point. Since acyl migration occurs with the intracellular lipase of immobilized cells, water may improve the cell permeability. Thus, the rate of methanolysis catalysed by ROL increases in the presence of added water. However, excess water reduces methanolysis as it also acts as a competitive inhibitor for transesterification. However, ROL is easier to separate from reaction mixture because of its larger size, and thus has leverage.
In another study, fungi were screened from soil for carrying out methanolysis of palm oil to form biodiesel. It was found that PDA-6, identified as Aspergillus nomius was the best isolate and showed maximum WCL methanolysis activity when waste cooking oil was used as carbon source. This is possibly the first report on wild-type Aspergillus species, which grew on waste cooking oil. The production of the whole cell biomass was highly favoured by polypeptone. [31]
While whole-cell biocatalysts seem to be a lucrative option given the reduced number of processes involved and its versatility in presence of water, the challenge of aseptic handling and immobilization of the cells looms as a big threat. Even so, research in this arena is in its budding stage, and it is hoped that this technique will provide a viable solution in the near future.
Table 1: A Comparison of Different Immobilization Techniques
|
|
Adsorption
|
Covalent Attachment
|
Entrapment/
Encapsulation
|
Cross-Linked
Enzyme
Aggregates
|
Whole Cell Biocatalyst
|
|
Enzyme Activity
|
Increased activity on hydrophobic supports
|
Reduced activity because of shape distortion
|
Activity reduced because of the use of harsh chemicals or because of increased polymer density
|
Increased activity with low particle size
|
Higher enzyme activity
|
|
Advantages
|
Easy and cheap
Requires mild conditions, so enzyme not damaged
|
Enhanced stability and reusability because of multipoint attachment
Little or no desorption
|
Increased immobilization yield
Gel beads can be used in reactors for industrial use
|
One step process of purification and immobilization
Carrier-free, hence low cost process
|
Less number of procedures involved
Active even in presence of water, thus more versatile
|
|
Disadvantages
|
Easy desorption
Requires inert proteins that reduce loading efficiency
|
Reduced enzyme activity
Support activation requires a series of chemical reactions with harsh chemicals
|
Possibility of leaching away of enzyme
Disruption of enzyme structure because of harsh microenvironment
|
Low activity on high enzyme loading
Low activity if substrate size is close to particle size
|
Susceptibility to methanol poisoning
Aseptic immobilization will be a challenge
|
|
Viability for Industrial Use
|
The reuse efficiency is low because of desorptive properties. The savings from using this cheaper method must outweigh the cost of replenishing with new enzyme.
|
Best method for operational stabilization, but poor enzyme activity can reduce efficiency of production
|
Suitable for industrial use after special treatment with chemicals, which increases the cost of production
|
Allows for rapid optimization and hence, suitable for scaling up
|
Higher operational stability and activity make it a preferable industrial option
|
Looking Ahead
While numerous ways of lipase immobilization are being studied, many such lipases have become commercially available. The most commonly used lipases are of bacterial or fungal origin. Candida antarctica lipase is available as Novozym 435, Candida rugosa as Lipase AY, Thermomyces lanuginose as Lipozime TL, and so on [8]. The biggest contributor to the cost of enzymatic biodiesel production is the cost of the lipase. The carrier or support material used for immobilizing the lipase greatly enhances this cost. The ideal support will be low cost and have optimal reaction and mass transfer efficiency. In 2012, Piedmont Biofuels announced the successful scaling up of the enzymatic biodiesel production in a commercial product setting. This indicates that immobilized enzymes have finally arrived at the industrial scene [35].
Even though, a lot has been achieved in the field of enzyme immobilization for biodiesel production, there is a great scope for refining these techniques to make lipase catalyzed biodiesel production suitable for an industrial scale.
Different reactions setups, and reactor designs for enzymatic biodiesel production have been used. The most commonly used bioreactors type for enzymatic biodiesel production are batch-stirred tank reactor and packed bed reactors (PBR) [32]. Fixed bed bioreactors consist of enzyme immobilized on fibrous membranes or gel beads. In such bioreactors, the flow rate of substrates can be easily altered, and a constant reaction environment can be maintained, thus optimizing the process and improving the efficiency [13, 22].
When scaling up production using immobilized enzymes, a balance between cost and effectiveness needs to be created. Solvent-free systems for packed bed reactors decrease production cost while retaining enzyme activity, but increase the pressure drop in the reactor. For optimal working of PBRs, the pressure drop needs to be minimized, for which an appropriate particle diameter for immobilized enzyme has to be chosen. This, however, may lower the mass transfer rate and the overall reaction rate. Thus, these factors need to be taken care of when evaluating systems and reactor choices. Reactors using static mixers can be used instead of the PBRs [8]. Thus, the system and reactor must be chosen with a great care, keeping in mind the characteristics of the immobilized enzyme in use.
In order to reduce operational costs, enzymatic biodiesel must be produced in continuously operated plants. Several studies reported the successful application of PBRs for enzymatic biodiesel production using different setups: single PBR used with stepwise addition of methanol, single recirculating PBR, three PBRs in series with intermediate glycerol removal and methanol addition, nine PBRs in series with a hydrocyclone set after PBR to separate glycerol[32].
Using PBRs has the advantage of lengthened temporal enzyme stability. PBRs are very applicable for continuous biodiesel production, but a big drawback is that the resulted glycerol remains at the bottom of the reactor and might deposit on the surface of the immobilized lipase, thus decreasing the accessibility, and hence, catalytic efficiency. So, glycerol must be eliminated in a periodic manner during the process [32].
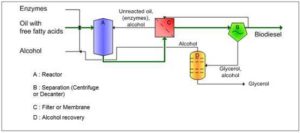
Figure 7: Enzymatic Biodiesel Production
Excess methanol, trapped within the immobilization support, tends to strip away water molecules surrounding the enzyme. These water molecules help maintain the three-dimensional structure of the enzyme. Thus, the lipase may not be stable in presence of excess methanol. A solution for the same problem is stepwise addition of methanol. Another way is to immerse the immobilized lipase in tert-butanol. It has been seen that this increases the activity of the lipase. Washing with tert-butanol will effectively regenerate the deactivated lipase and also remove contaminating glycerol, thus killing two birds with one stone [19].
While selecting appropriate support for adsorption, Atomic Force Microscopy (AFM) can be utilized. In AFM, a probe at the end of a cantilever is used to examine the surface of the specimen. When the tip or the probe is brought close to the sample surface, the cantilever deflects according to the Hooke’s Law, as a result of the force between the probe and the surface [30]. Thus, the force of interaction between sample and the tip can be measured [30].This technique can be utilized to measure the force of interaction between the enzyme, which can be coated on the tip of the probe, and the possible supports. This way, a better judgment can be made, and one can avoid the less economic hit-and-trial methods.
Co-immobilization of lipases is another option. Co-immobilization of the necessary enzymes for the pathway results in a rapid conversion through the pathway due to the localized high concentrations of the intermediates. The reduction in the apparent lag phase is most noticeable when there are more enzymes in the pathway. In a recent study, biodiesel production from Jatropha and waste oil was investigated in a packed-bed reactor using co-immobilized lipases fromRhyzopus oryzae and Candida rugosa. When the continuous process was operated for 72 h under the optimized conditions for biodiesel production, the normal conversion yield was about 90 % [33].
Understanding enzyme’s primary, secondary and tertiary structure and modifying it to carry out easy immobilization, while still retaining activity is necessary and can lead to amazing results as was seen in the case of genetically modified TLL with just one lysine residue covalently binding with aldehyde groups on the support surface [21].
Conclusion
The study of different modes of lipase immobilization shows that lipases from different sources are different in structure and activity, and the interaction between one enzyme and one support is unique. Altering the microenvironment of the lipase can significantly change its immobilization efficiency and activity. A variety of immobilization techniques like adsorption, covalent attachment, entrapment, cross-linked enzymes and whole-cell biocatalysts are available at hand. A clever judgment and manipulation of these techniques are required to carry out a successful immobilization which can make the lipase suitable for biodiesel production at an industrial scale. While immobilization is a well studied science, it will take the genius of an artiste to find the right combination of support, enzyme and process that will make large scale lipase catalysed biodiesel production a reality.
References
1.Finfacts Team (2010). “BP Statistical Review of World Energy 2010: Recession Drove 2009 Energy Demand Lower; Oil Giant Says Global Reserves Sufficient to Meet 2009 Production for 45.7 Years,” EnergyInsights.net, [Retrieved March 1, 2014] http://www.energyinsights.net/cgi-script/csArticles/articles/000062/006243.htm
Publisher
2. Energy Information Administration, USA. (2004). “Greenhouse Gases, Climate Change, and Energy,” EIA brochures, [Retrieved March 1, 2014] http://www.eia.gov/oiaf/1605/ggccebro/chapter1.html
Publisher – Google Scholar
3. Diesel Service and Supply. “Why Should I Choose a Diesel Powered Generator?,” [Retrieved January 10, 2013],www.dieselserviceandsupply.com/why_use_diesel.aspx
Publisher
4. Wikipedia, Biodiesel, [Retrieved January 10, 2013], http://en.wikipedia.org/wiki/Biodiesel
Publisher
5. Knothe, G. & Van Gerpen, J. (2010). The Biodiesel Handbook, second ed., AOCS Publishing
Publisher
6. Akoh, C. C., Chang, S.- W., Lee, G.- C. & Shaw, J.- F. (2007). “Enzymatic Approach to Biodiesel Production,”Journal of Agricultural and Food Chemistry, 55 (22): 8995—9005
Publisher – Google Scholar
7. Freedman, B., Pryde, E. H. & Mounts, T. L. (1984). ‘Variables Affecting the Yield of Fatty Esters from Transesterified Vegetable Oils,’ Journal of Oil and Fat Industries, 61 (10)1638—1643
8. Stoytcheva, M., Montero, G., Toscano, L., Gochev, V. & Valdez, B. (2011). “Biodiesel — Feedstocks and Processing Technology,” ISBN: 978-953-307-713-0, Intech
Publisher
9. Fjerbaek, L., Christensen, K. V. & Norddahl, B. (2009). “A Review of the Current State of Biodiesel Production Using Enzymatic Transesterification,” Biotechnology and Bioengineering, 102(5)1298—1315.
Publisher – Google Scholar
10. Shah, S., Solanki, K. & Gupta, M. N. (2007). “Enhancement of Lipase Activity in Non-Aqueous Media upon Immobilization on Multi-Walled Carbon Nanotubes,” Chemistry Central Journal, 1:30-36.
Publisher – Google Scholar
11. Gitlesen, T., Bauer, M. & Adlercreutz, B. (1997). “Adsorption of Lipase on Polypropylene Powder,” Biochimica et Biophysica Acta (BBA)- Lipids and Lipid Metabolism, 1345(2)188—196
Publisher – Google Scholar
12. Palomo, J. M., Muñoz, G., Fernández-Lorente, G., Mateo, C., Fernández-Lafuente, R. & Guisán, J. M. (2002). “Interfacial Adsorption of Lipases on Very Hydrophobic support (Octadecyl—Sepabeads): Immobilization, Hyperactivation and Stabilization of the Open Form of Lipases,” Journal of Molecular Catalysis B: Enzymatic 19-20:279—286
Publisher – Google Scholar
13. Huang, X.- J., Yu, A.- G. & Xu, Z.- K. (2008). “Covalent Immobilization of Lipase from Candida Rugosa onto Poly (Acrylonitrile-Co-2-Hydroxyethyl Methacrylate) Electrospun Fibrous Membranes for Potential Bioreactor Application,”Bioresource Technology, 99(13)5459—5465
Publisher – Google Scholar
14. Eggert, T., van Pouderoyen, G., Pencreac’h, G., Douchet, I., Verger, R., Dijkstra, B. W. & Jaeger, K.- E. (2002). “Biochemical Properties and Three-Dimensional Structures of Two Extracellular Lipolytic Enzymes from Bacillus Subtilis,” Colloids and Surfaces B: Biointerfaces, 26(1-2)37—46
Publisher – Google Scholar
15. Al-Duri, B. & Yong, Y. P. (2000). “Lipase Immobilisation: An Equilibrium Study of Lipases Immobilised on Hydrophobic and Hydrophilic/Hydrophobic Supports,” Biochemical Engineering Journal, 4(3)207—215.
Publisher – Google Scholar
16. Rodrigues, D. S., Cavalcante, G. P., Ferreira, A. L. O. & Goncalves, L. R. B. (2008). “Immobilization of Candida Antarctica Lipase Type B by Adsorption on Activated Carbon,” Chemical and Biochemical Engineering Quarterly, .22(1)125-133
Publisher
17. Mendes, A. A., Giordano, R. C., Giordano, R. L. C. & de Castro, H. F. (2011). “Immobilization and Stabilization of Microbial Lipases by Multipoint Covalent Attachment on Aldehyde-Resin Affinity: Application of the Biocatalysts in Biodiesel Synthesis,” Journal of Molecular Catalysis B: Enzymatic, 68(1)109—115
Publisher – Google Scholar
18. Kim, M. I., Ham, H. O., Oh, S.- D., Park, H. G., Chang, H. N. & Choi, S. H. (2006). “Immobilization of Mucor Javanicus Lipase on Effectively Functionalized Silica Nanoparticles,” Journal of Molecular Catalysis B: Enzymatic, 39: 62—68.
Publisher – Google Scholar
19. Aybastier, O. & Demir, C. (2010). “Optimization of Immobilization Conditions of Thermomyces Lanuginosus Lipase on Styrene—Divinylbenzene Copolymer Using Response Surface Methodology,” Journal of Molecular Catalysis B: Enzymatic, 63: 170—178
Publisher – Google Scholar
20. Bruno, L. M., Coelho, J. S., Melo, E. H. M. & Lima-Filho, J. L. (2005). “Characterization of Mucor Miehei Lipase Immobilized on Polysiloxane-Polyvinyl Alcohol Magnetic Particles,” World Journal of Microbiology and Biotechnology, 21: 189—192.
Publisher – Google Scholar
21. Hanefeld, U., Gardossi, L. & Magner, E. (2009). “Understanding Enzyme Immobilisation,” Chemical Society Reviews, 38:453-468
Publisher – Google Scholar
22. Hilal, N., Kochkodan, V., Nigmatullin, R., Goncharuk, V. & Al-Khatib, L. (2006). “Lipase-Immobilized Biocatalytic Membranes for Enzymatic Esteriï¬cation: Comparison of Various Approaches to Membrane Preparation,” Journal of Membrane Science, 268:198-207.
Publisher – Google Scholar
23. Chen, J.- P. & Lin, W.- S. (2003). “Sol-Gel Powders and Supported Sol-Gel Polymers for Immobilization of Lipase in Ester Synthesis,” Enzyme and Microbial Technology, 32: 801—811.
Publisher – Google Scholar
24. Betigeri, S. S. & Neau, S. H. (2002). “Immobilization of Lipase Using Hydrophilic Polymers in the Form of Hydrogel Beads,” Biomaterials, 23:3627-3636
Publisher – Google Scholar
25. Alloue, W. A. M., Destain, J., Medjoub, T. E., Ghalfi, H., Kabran, P. & Thonart, P. (2008). “Comparison of Yarrowia Lipolytica Lipase Immobilization Yield of Entrapment, Adsorption, and Covalent Bond Techniques,” Applied Biochemistry and Biotechnology, 150:51-63
Publisher – Google Scholar
26. Cao, L. (2005). “Immobilized Enzymes: Science or Art?,” Current Opinion in Chemical Biology, 9:217—226
Publisher – Google Scholar
27. Yu, H. W., Chen, H., Wang, X., Yang, Y. Y. & Ching, C. B. (2006). “Cross-Linked Enzyme Aggregates (CLEAs) with Controlled Particles: Application to Candida Rugosa Lipase,” Journal of Molecular Catalysis B: Enzymatic, 43:124—127
Publisher – Google Scholar
28. Gupta, P., Dutt, K., Misra, S., Raghuwanshi, S. & Saxena, R. K. (2009). “Characterization of Cross-Linked Immobilized Lipase from Thermophilic Mould Thermomyces Lanuginosa Using Glutaraldehyde,” Bioresource Technology, 100:4074—4076
Publisher – Google Scholar
29. Tamalampudi, S., Talukder, M. R., Hama, S., Numata, T., Kondo, A. & Fukuda, H. (2008). “Enzymatic Production of Biodiesel from Jatropha Oil: A Comparative Study of Immobilized-Whole Cell and Commercial Lipases as a Biocatalyst,”Biochemical Engineering Journal, 39:185—189
Publisher – Google Scholar
30. Wikipedia, Atomic Force Microscopy, [Retrieved on January 10, 2013],http://en.wikipedia.org/wiki/Atomic_force_microscopy
Publisher
31. Talukder, M. M. R., Lee, H. Z. S., Low, R. F., Pei-Lyn, L. C., Waszecha, D. & Wu, J. (2013). “Potential Use of Whole Cell Lipase from a Newly Isolated Aspergillus Nomius for Methanolysis of Palm Oil to Biodiesel,” Journal of Molecular Catalysis B: Enzymatic, 89:108-113.
Publisher – Google Scholar
32. Gog, A., Roman, M., Tosa, M., Paizs, C. & Irimie, F. D. (2012). “Biodiesel Production Using Enzymatic Transesterification — Current State and Perspectives,” Renewable Energy, 39:10-16
Publisher – Google Scholar
33. Lee, J. H., Yoo, H.- Y., Suh, Y. J., Han, S. O., Park, C. & Kim, S. W. (2012). “Process Development Using Co-Immobilized Lipases for Biodiesel Production,” International Proceedings of Chemical, Biological and Environmental Engineering. 28 (2012) 171-174
Publisher – Google Scholar
34. Salis, A., Sanjust, E., Solinas, V. & Monduzzi, M. (2003). “Characterisation of Accurel MP1004 Polypropylene Powder and its Use as a Support for Lipase Immobilisation,” Journal of Molecular Catalysis B: Enzymatic, 24-25:75-82
Publisher – Google Scholar
35. Kotrba, R. (2012). “Piedmont Proves Liquid Enzyme Catalysis Works at Commercial Scale,” Biodiesel Magazine[Online], [Retrieved February 28, 2014], http://www.biodieselmagazine.com/articles/8328/piedmont-proves-liquid-enzyme-catalysis-works-at-commercial-scale
Publisher










