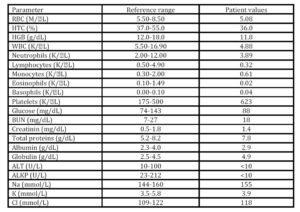
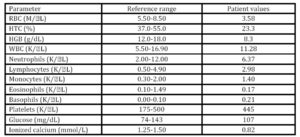
The animal was premedicated with subcutaneous morphine (0.4 mg/kg), induction was performed with propofol (4 mg/kg) and diazepam (0.5 mg/kg) and maintained with isofluorane in oxygen. Before surgery, vaginoscopy using a 30º 2.7 mm telescope was performed in order to detect the probable source of haemorrhage. Blood clots were observed and, due to the open cervix, suture dehiscence of the uterine surgical wound was appreciated. Remnants of foetal membranes were not observed. Ventral midline laparotomy was performed and strong adherences of the uterus to the muscular layer and the omentum were found (Fig.1) as well as free peritoneal fluid. After careful dissection of adherences, rupture of part of the surgical wound of the C-section was observed as well as suture dehiscence and abundant granulation tissue (Fig. 2). Ovariohysterectomy was performed conventionally, peritoneal cavity was washed abundantly with 1,5 litres of warm sterile saline solution and after devitalized tissue of the muscular, subcutaneous and skin was removed, laparotomy was routinely closed. Tissue samples from the uterus were submitted for culture and histological evaluation.
The animal recovered uneventfully and was discharged 24h after surgery with analgesic and antibiotic therapy consisting on tramadol 2 mg/kg q8h PO and cephalexin 20mg/kg q12h PO, respectively.
Culture of the uterine secretions obtained after surgery yielded growth of Proteus spp. and Escherichia coli, both of which were resistant and sensible, respectively, to cephalexin. Antibiogram results were obtained 6 days after last surgery and, due to the clinical improvement of the dog during this time period, the fact that she was still lactating, and the difficulty to administer another antibiotic by other route different from the oral one, cephalexin was maintained until skin staples’ removal.
Histology revealed a severe purulent and necrotizing metritis with intense accumulation of degenerated neutrophils in the mucosa that caused an evident endometrial disruption. The mesentery adjacent to the uterus showed an intense and severe purulent necrotizing inflammation associated with zones of septal fibrosis and presence of granulation tissue on serosal surface.
Discussion and Conclusions
Metritis and infection of the surgical incision in women account for nearly three fourths of post-caesarean infections, most of them are found to be surgical site related, with 55% classified as uterine in origin and 19% classified as arising from the surgical incision (Horan et al., 1993). The latter complicates approximately 5% to 8% of caesarean deliveries (Normand et al., 2001). This case report describes a severe diffuse fibrino-purulent necrotizing metritis with subsequent uterine rupture following a caesarean section in a bitch. The prolonged labour and dystocia, probably associated to a failed delivery of a puppy with foetal anasarca, could cause that presence of normal vaginal bacteria (Escherichia coli and Proteus spp.) colonizing the uterus. Indeed, post-caesarean metritis, as well as the majority of wound infections, are ascending in nature and caused by normal vaginal microorganisms entering the uterus (Normand et al., 2001; Orfanou et al., 2010). Also, a highly distended uterus caused by a pregnancy of 11 foetuses in a French Bulldog may provide an ideal culture medium susceptible to bacterial colonization. Additionally, a possible suture material rejection, maybe enhanced by the presence of infection, as a promoting cause of suture dehiscence could not be discarded either, as the same suture material was used for the closure of the uterus, skin and subcutaneous tissue, but not for the muscular layer, which appeared intact. Nevertheless, during surgery, bacteria can be transported by foetal fluids not only to the uterine incisions, but also to the abdominal incisions (Normand et al., 2001). This could also explain why dehiscence of the skin and subcutaneous tissue occurred. Although samples for bacterial culture from these tissues were not collected, one may expect that surgical contamination of skin and subcutaneous tissue could easily happen during foetal delivery by c-section.
Hypocalcemia in bitches can occur prior to parturition, as it occurs in cows, but it is much more common during the first weeks of the post-partum (Kaufman, 1986), being more frequent in the bitches of small breeds, (Burke, 1977; Martin and Capen, 1980; Mosier, 1980; Johnston, 1983; Feldman, 1995). Both clinical and subclinical hypocalcemia has been associated to metritis in cows (Curtis et al., 1983; Martinez et al., 2012). The hypothetical mechanism is based on the fact that hypocalcemia decreases smooth muscle tone which in turn would increase the risk of metritis (Goff and Horst, 1997; Martínez et al., 2012) and placenta retention (Curtis et al., 1983). According to the reviewed literature, hypocalcemia has not been determined as causing metritis in bitches, although this possibility should be taken into account. However, in the present clinical case a more feasible reason for metritis would be the primary uterine inertia induced by an elevated number of foetuses that would overstretches the uterus. The lack of uterine contractions prevented the delivery of the foetuses and allowed the entrance of vaginal microorganisms into the uterus that would induce metritis.
Puerperal haemorrhage can occur in animals with coagulation disorders. Rarely, it can be the result of erosion of large blood vessel(s) in case of subinvolution of placental sites. It may also be due to uterine rupture, to venereal neoplasms or to vaginal injuries (Orfanou et al., 2010). Initial treatment of cases of mild haemorrhage includes administration of oxytocin (5 to 20 IU/kg, intramuscularly) and the general support of the animal (Orfanou et al., 2010). In cases of severe haemorrhage or when after oxytocin administration haemorrhage persists, ovariohysterectomy is indicated after the animal’s condition is stabilized.
Because of the owners’ willingness to preserve the reproductive function of the animal and in order to identify and detect the possible source of haemorrhage, a vaginoscopy was performed. However, during the procedure, an active haemorrhage was not identified, but loose suture material in the uterine incision was observed and an exploratory laparotomy was decided followed by an ovariohysterectomy. Pneumoperitoneum was not induced probably due to the seal provided by the presence of adherences between the uterine incision and the muscular wall.
Amoxicillin is a safe antibiotic for lactating puppies and is a good choice when C-section shows no complications. In the present case, C-section surgery was completely routine and amoxicillin therapy for 10 days was decided. However, when metritis was diagnosed, amoxicillin was replaced by cephalexin which is a wider broad antibiotic and also safe for lactating puppies. Sensitivity test showed that cephalexin was effective against Escherichia coli but not Proteus spp, both obtained from uterine secretions. Of course, lactation could be suspended and feed the puppy with artificial feeding and administrate a more sensitive antibiotic. However, the good recovery of the bitch indicated that the therapy was effective and the antibiotic was not changed.
Thus, although in bitches, metritis is usually induced by foetal membranes or foetuses retention (Burke,1977; Boscos et al.,1996; Linde-Forsberg, 2005), in this case report, we could conclude that metritis was induced by a prolonged delivery that facilitated the ascending colonization of bacteria from the vagina. The metritis promoted the dehiscence of the uterine surgical incision to occur. The infection probably spread to the subcutaneous tissue and abdominal cavity during the surgery and following uterine rupture, respectively. Peritonitis caused by uterine rupture was kept circumscribed due to omental and peritoneal adherences.
References
1. Boscos, C., Smartzi, F. (1996). “Post-partum disorders in the bitch”. Bull Hell Vet Med Soc 46, 342-351.
2. Burke, T.J. (1977). “Post parturient problems in the bitch”. Vet Clin N Am 7, 342-351.
3. Casey, B.M., Cox, S.M. (1997). “Chorioamnionitis and endometritis”. Infectious Disease Clinics of North America 11(1), 203-222.
4. Curtis, C.R., Erb, H.N., Sniffen, C.J., Smith, S.D., Powers, P.A., Smith, M.C., White, M.E., Hillman, R.B., Pearson, E.J. (1983). Association of parturient hypocalcemia with eight periparturient disorders in Holstein cows. J. Am. Vet. Med. Assoc., 183, pp. 559—561.
Google Scholar
5. Goff, J.P., Horst, R.L. (1997). Physiological changes at parturition and their relationship to metabolic disorders. J. Dairy Sci., 80, pp. 1260—1268.
Publisher – Google Scholar
6. Haas, D.M., Morgan, S., Contreras, K. (2013). Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev., 31;1:CD007892.
Google Scholar
7. Hofmeyr, G.J., Smaill F.M. (2002). Antibiotic prophylaxis for cesarean section. Cochrane Database Syst Rev. Issue 3:CD000933.
Google Scholar
8. Horan, T.C., Culver, D.H., Gaynes, R.P., Jarvis, W.R., Edwards, M.S., Redi, C.R.(1993). “Nosocomial infection in surgical patients in the United States, January 1986-June 1992”. Infection Control and Hospital Epidemiology 14(2), 73-80.
Publisher – Google Scholar
9. Linde-Forsberg, C. (2005).”Abnormalities in pregnancy, parturition, and the periparturient period”. In: S.J. Ettinger, E.C. Feldman, (Eds) Textbook of Veterinary Internal Medicine; 6th edition, 1664-1667.
10. Magne, M.L. (1986) “Acute metritis in the bitch”. In: Morrow. A.D. (Ed) Current Therapy in Theriogenology; 2nd Edition 505-506.
11. Martinez, N., Risco, C.A., Lima, F.S., Bisinotto, R.S., Greco, L.F., Ribeiro, E.S., Maunsell, F., Galvão, K., Santos, J.E.P. (2012). Evaluation of peripartal calcium status, energetic profile, and neutrophil function in dairy cows at low or high risk of developing uterine disease J. Dairy Sci., 95, pp. 7158—7172.
Publisher – Google Scholar
12. Normand, M.C., Damato, E.G. (2001). “Postcesarean Infection”. Journal of Obstetric, Gynecologic, & Neonatal Nursing 30(6), 642-648.
Publisher – Google Scholar
13. Orfanou, D.C., Ververidis, H.N., Boscos, C.M., Fthenakis, G.C.(2010). “Post-partum pathological condition in the bitch — Part II”. European Journal of Companion Animal Practice 20(2), 119-126.
Google Scholar
14. Pastorekured, J.G. (1995).”Post-cesarean endometritis”. Comprehensive Therapy 21(5): 249-253.
15. Spandorfer, S.D., Graham, E., Forouzan, I. (1996). ”Postcesarean endometritis: Clincial risk factors predictive of positive blood cultures”. Journal of Reproductive Medicine 41(11), 797-800.
Google Scholar
16. Wheeler, S.L., Magne, M.L., Kaufman, J., Husted, P.A., Allen, T.A., Olson, P.N. (1984). “Postpartum disorders in the bitch”. Comp. Cont. Educ. Pract. 6, 493-500.
17. Wykes, P.M., Olson, P.N. (1993) “Diseases of the uterus”. In: Bojrab, M.M., Bloomberg, M.S., Smeak, D.D. (Eds) Disease Mechanisms in Small Animal Surgery; 2nd Edition 570-573.