Introduction
Regulatory T cells (Tregs) play a principal role in immune reactions including autoimmunity, transplantation tolerance, anti-infectious immunity and cancer. These cells represent about <5% of the peripheral CD4+ T cell population and play a crucial role in the maintenance of immune homeostasis against self- antigens [1, 2]. Numerical or functional deficit of Treg cells is linked to many autoimmune diseases such as rheumatoid arthritis [3], multiple sclerosis or type 1 diabetes. These Treg features result from their potent capacity to reduce the activation and expansion of conventional T cells [4] by suppressing their biological activities like proliferation and blocking the production of proinflammatory cytokines TNF alpha and IFNgamma, among others [5].
The two main groups of Tregs represent natural Tregs (nTregs) and induced or adaptive Tregs (iTregs) [6]. nTregs originate in thymus and express high levels of the IL-2 receptor α-chain (CD25) [7] and the forkhead-winged helix transcription factor FoxP3, which is inevitable for their development and function [8, 9]. FoxP3+ iTregs arise in periphery from conventional naive CD4+FoxP3− T cells. Under the influence of suppressive cytokines and antigen-specific activation they develop into FoxP3+ Tregs.[6].
FoxP3+ Tregs can be divided into two subpopulations based on the expression of ICOS (inducible T cell co-stimulator) [10]. These subpopulations are both anergic and suppressive, but exert different molecular mechanisms for suppression. While ICOS−FoxP3+ Tregs mediate their suppressive activity via TGF-β, ICOS+FoxP3+ Tregs moreover secrete IL-10.
Today the most widely accepted phenotype for Tregs is the coexpression of CD4, CD25 (α-chain of the IL-2 receptor), and of FoxP3 [10].
Mechanisms of Treg Function
Tregs can suppress a whole range of immune cells including B cells, NK cells, NKT cells, CD4+ or CD8+T cells, and both monocytes and dendritic cells. The most important and extensively studied is the suppression of CD4+CD25- conventional T cells (TCONV). Main function of Tregs is to suppress the activation of naive TCONV, but they can also inhibit activated effector T cells and memory CD4+ [11] and CD8+ T cells [12]. Tregs can suppress their proliferation directly [13] without the presence of antigen presenting cells (APCs). There are evidences for either contact-dependent suppression or suppression via immunosuppressive cytokines or other factors. One efï¬cient way to supppress immune responses is direct killing of CD4+ effector cells, which was described by Grossman et al.,[14]. They have shown that upon CD3/CD46 activation human nTregs express the serine protease granzyme A and kill TCONV in a perforin-dependent manner without CD95-CD95L interaction.
Nevertheless, the main mechanism of suppression of Tregs consist in influencing the activation status of APCs. In this regard the key molecule is CTLA4, which competitively inhibits the binding of CD28 to its ligands CD80 and CD86 and thus inhibits co-stimulation of effector T cells. CTLA4 together with the adhesion molecule LFA-1 also downregulates the expression of CD80 and CD86 on APCs [15, 16]. Capturing of CD80/86 ligands by CTLA4 is a process called trans-endocytosis [17]. Tregs do not only reduce antigen presenting activity of APCs, but also support an immunosuppressive cytokine milieu by reducing IL-6 while increasing IL-10 production by DCs [18].
Tregs can produce high amounts of membrane-bound and soluble TGF-ß, and blocking TGF-ß partially abrogated suppression of T cell proliferation in vitro using murine or human T cells [19, 20] by inhibiting the TCR/CD3 pathway [21]. An important role in Treg-mediated suppression plays cytokine IL-10, which exerts immunosuppressive effects on various cell types [22]. Another way of suppression offers IL-35, the cytokine involved in Treg-mediated suppression, which was shown to directly inhibit TCONV proliferation [23].
Hydrolysis of extracellular ATP to ADP or AMP by the ectoenzyme CD39, expressed by all murine Tregs and by about 50% of human Tregs, represents another Treg-mediated anti-inflammatory mechanism [24].
Treg cells express high levels of CD25 and consecutively deprive the environment of IL-2, which can also affect the survival of effector T cells.
Human and Mouse Differences
To describe all differences between human and mouse systems is not the topic of this rewiev. Shortly speaking, in mice most of CD4+CD25+ T cells are potently suppressive, whereas analogous population in humans, albeit larger, contains mainly activated effector cells and only a small percent comprising CD25high cells represents Tregs [10, 25]. Particular differences are described if needed.
Intracellular Markers
FoxP3
The transcription factor FoxP3 is regarded to be a lineage molecule for Tregs. It is neccessary for their thymic development, function and phenotype and is responsible for controlling the expression of a number of genes including suppresive cytokines and Treg surface molecules [8, 10, 26, 27]. However, FoxP3+ T cells are phenotypically and functionally heterogeneous and involve both suppressive and nonsuppressive T cells. Moreover, FoxP3 is an intracellular molecule, detection of which requires fixation and permeabilization of cells and the ï¬xed cells cannot be used in studies of Treg function. Intracellular FoxP3 staining is hardly usable on a daily clinical routine basis in large series of samples (several steps of incubation, washing, long incubation times, use of isotype control).
FoxP3 has been broadly used for characterization of thymus derived naturally occurring Tregs, although it is well known that is also expressed in peripheral induced Treg [28] and even in human activated CD4+CD25− T effector cells without suppressive activity [29-33]. Therefore, to delineate Treg subset with maximal precision, the use of quantitative analysis and a combination of other markers rather than just qualitative assessment of Foxp3 and CD25 expression is necessary. Despite these facts CD4+CD25+FoxP3+ is still generally accepted as the most reliable phenotype of Treg cells [8, 9, 34].
Because of the absence of a specific Treg cell marker, it has not been possible to distinguish between Treg cells and conventional activated T cells [35]. Later, epigenetic differences in DNA methylation within FoxP3 have been detected between Treg cells and non-regulatory T cells [36], which offered a promising option for the exact quantification of natural Treg cells. Tatura et al., [37] described the application of QAMA assay (quantitative analysis of methylated alleles), that is based on epigenetic differences within the FoxP3 Treg- specific demethylated region (TSDR) between Treg cells and all other major blood cells.
HELIOS
Helios is an Ikaros family transcription factor which was shown to play an important role in Treg function and which was at first believed to differentiate induced Treg cells from natural Treg cells [38, 39]. The aforesaid studies demonstrated the existence of two subsets of FoxP3+ Tregs which express Helios and which can discern nTregs (FoxP3+Helios+) from peripherally-induced (FoxP3+Helios−) iTregs [39]. But recently Himmel et al., found, that a lack of Helios expression does not exclusively identify human iTregs, and, their data provide the first evidence for the coexistence of Helios+ and Helios− nTregs in human peripheral blood [40]. However, it has been reported very recently that Neuropilin 1 (Nrp1, CD304), highly expressed by most of nTregs, may be used to distinguish these two subsets [41, 42].
RUNX
The role of Runt-related transcription factor (RUNX) family proteins in relation to Treg development and function has been explored during last few years. It was shown that Foxp3 expression in nTregs is dependent on RUNX proteins and correlates with the binding of RUNX to Core-binding factor β (CBF-β). Heterodimers of RUNX and CBF-β have been shown to play an important role in maintenance and development of Tregs in both mice and humans, since they controll Foxp3 expression and also the expression of target genes [43]. Complex of RUNX1 and CBF-β seems to be indispensable for the suppressive function of human nTregs [44]. Similar importance has the complex of CBF-β with RUNX1 and RUNX3 for the TGF-β mediated iTreg development and function [45]. RUNX deficient mice produce autoimmune disorders resembling those occurring in Foxp3-mutants, although the symptoms are less severe [44].
Surface Markers
CD25
Lot of studies indicate that CD25 (IL-2R —α) is a principal cell surface marker of Tregs [25, 46], but some of them have showed that only the CD4+ T cell subset expressing the high levels of CD25 (termed CD25high) exerts in vitro suppressive activity and has the greatest regulatory potential [25, 47]. However, conventional/effector T cells [10, 25, 48] and a portion of CCR7+ central memory T lymphocytes start expressing CD25 upon TCR-mediated activation [49]. Therefore, even highly pure CD4+CD25high Treg populations may contain a significant fraction of proinflammatory T effector cells [13].
CD26
CD26 is an extracellular serine protease with dipeptidyl peptidase IV (DPPIV) activity [50]. High expression of CD26 has been traditionally used as an indicator of immune activation and effector functions in T cells. Activated and memory T cells display a CD26high phenotype, and T cytokines like IL-12 raise the number of CD26 molecules on T lymphocytes. High surface levels of this protease indicate TH1 effector responses [50, 51].
Recently interesting data has been published by Salgado et al., [52] Their flow cytometry outcomes demonstrated high percentage of CD26 within CD4+CD25− or CD4+FoxP3−/low effector T lymphocytes, but negative or low levels (CD26−/low) in Treg cells, which were gated as the CD4+CD25high or the CD4+FoxP3high phenotype. While the negative marker CD127 is down modulated in CD4+ T effector lymphocytes after their activation, CD26 molecule within these activated cells is upregulated and becomes a CD4+CD25+/high CD26+ phenotype. The upregulation of CD26 within Treg cells (CD4+CD25high CD26−/low) is only slight. The differences between Treg and activated effector T cells regarding CD26 levels seem to be stable, therefore assessment of CD26 molecule, in combination with currently used markers as CD25, FoxP3, CD194 and others may be useful in distinguishing Treg cells from activated T effector cells. This approach could help to quantify or isolate Tregs from samples of patients with autoimmune or inflammatory diseases.
CD39
Two studies highlighted the importance of CD39/NTPDase1 (ecto-nucleoside triphosphate diphosphohydrolase 1) as a useful sign for the identification of Tregs [24, 53]. Mandapathil et al., [54] have further focused on CD39 as a positive selection marker of Tregs. Their paper pointed out that CD39 molecule can be successfully used for routine characterization and isolation of functionally unharmed human T regulatory cells from the peripheral blood of patients or healthy donors. The ectoenzyme NTPDase1 catalyzes the generation of AMP from ATP, which is necessary to produce adenosine, an important mediator of active suppression [24, 55]. Some papers have also been published about the frequencies of CD39+CD4+ T cells of patients infected with HTLV-1 [56] or in HIV patients [57], regarding their correlation to Tregs.
It was found, that 50% to 90% of CD4+CD39+ T lymphocytes are FoxP3+ and express low levels of CD127 [54]. But further studies have shown that CD39 expression could serve as a marker to identify not only suppressive CD4+T cells, but also a CD4+ T-cell subpopulation with immunostimulatory properties [58]. Ndhlovu et al., [59] reported that the CD39 could identify new subset of “inducer” CD4+ T cells that significantly increases the proliferation and cytokine production of responder T cells. This unique cell subset produces a distinct repertoire of cytokines in comparison to the other CD4+ T cell subsets. It was proposed that this novel CD4+ T cell population counterbalances the suppressive activity of Tregs in periphery and serves as a calibrator of immunoregulation.
So the CD39 molecule expressed on CD4+ T cells together with CD25 co-expression, is the marker of T cells with distinct effector (CD39+CD25−) or regulatory (CD39+CD25+) function.
Moreover, further studies have shown, that activated T cells upregulate CD39 [60], and a new subset of human CD4+CD39+FoxP3− T cells that produce IFN-gamma and IL-17 has been found [61].
CD45RA
The naïve T cell marker CD45RA and FoxP3 have been identified on the population of CD4+CD25+ T cells from peripheral blood and these cells showed potent suppressive functions [62]. Miyara et al., [27] described CD45RA+FoxP3low T cells as a resting Tregs and Tregs with a CD45RA−FoxP3high phenotype as activated Treg cells. While activated Tregs are terminally differentiated and rapidly die, resting Tregs show a high proliferative capacity and convert into their activated descendants. The subpopulation of CD45RA+ Tregs can be used for cellular therapies, because it retains its suppressive activity also after in vitro expansion [63].
CD49d
Markus Kleinewietfeld et al., [64] have reported that the application of depleting antibodies for CD49d (alpha chain of VLA-4 integrin), a marker of effector T cells producing proinflammatory cytokines, will eliminate conventional T cells from Tregs and in combination with the CD127 antibody can give highly pure population of FoxP3+ Tregs.
CD49d molecule is expressed on most of IFN-gamma or IL-17-producing proinflammatory T cells and is reduced on Tregs, even though later data from the same group [24] as well as other researchers [65] admitted, that some degree of CD49d is expressed also in some subsets of Treg cells.
CD101
CD101 molecule, previously described as V7, is a type I transmembrane glycoprotein, which is expressed on monocytes, granulocytes, dendritic cells and activated T cells [66]. Binding of anti-CD101 monoclonal antibody on CD101 molecule on T cells blocks TCR/CD3 —induced proliferation by inhibiting calcium flux and activation of tyrosine kinase, leading to the suppression of IL-2 transcription.
In the study of Fernandez et al., cell surface expression of CD101 well correlated with functional suppressor activity of CD4+ CD25+ Treg cells in mice [67]. Tregs with high expression of CD101 are supposed to be activated Tregs.
CD127
In 2006 Liu et al., [47] discovered important hallmark for human Treg delineation: the surface molecule CD127, interleukin 7 receptor- α (IL7R- α) chain. They showed that the expression of CD127 inversely correlates with FoxP3 expression and suppressive activity of Tregs. The combination of CD127 with CD25 then allowed the isolation of highly purified Treg population [68].
The comparisons of intracellular FoxP3 staining with CD4+CD25+CD127− phenotype initially reported good correlation. The experiments were made in healthy subjects [68], in septic patients [69], in viremic HIV patients [57, 70] and in patients with systemic lupus erythematosus [71]. Isolated CD4+CD25highCD127− were used several time for clinical applications [72]. CD4+CD25hiCD127− isolated Tregs showed the best reached Treg population regarding purity, function, stability and in vitro expansion capacity [73], promising isolation of pure Treg populations with high suppressive functionality [68, 74]. Later on, most studies of Tregs started to use CD127 marker not only to isolate Tregs, but also to directly characterize them in human peripheral blood [75]. Yu et al., [71] found that CD4+CD25+CD127low/− T cells expressed the highest level of FoxP3 and had the strongest correlation with CD4+CD25+FoxP3+ T cells, the accepted identifying characteristics for “real” nTreg cells. Moreover, functional data showed that CD4+CD25+CD127low/− T cells could effectively suppress the proliferation of CD4+CD25− T cells, suggesting that these T cells best ï¬t the deï¬nition of naturally occurring regulatory T cells in human peripheral blood.
But later outcomes implicated, that the negative correlation with FoxP3+ T cells is not absolute.
CD4+CD25highCD127low and CD4+CD25highFoxP3+ were used at once to determine the frequency of Tregs in two different groups of human immunodeficiency virus (HIV) infected persons, one viremic and the other aviremic. As expected, a strong correlation between both Treg phenotypes was observed in the aviremic group, but surprisingly, in the viremic group this correlation was completely absent. These findings on T cell activation levels suggested that the CD4+CD25highCD127low population corresponds with the elevated numbers of activated non-regulatory T cells in the viremic HIV group, suggesting that CD127 molecule is greatly influenced by mere T cell activation [76]. Very similar outcomes reported Rios et al., [77]. In HIV patients detection of Tregs as CD4+CD25+CD127Low/− cells resulted in a significantly lower percentage of cells in comparison with FoxP3+ population.
These results were confirmed by measurement of DNA-methylation (QAMA) in samples of CD4+ T cells isolated from blood of septic patients and healthy donors. Data were compared with measurement of CD4+CD25highCD127low population by flow cytometry.
In healthy subjects, the results obtained by both methods were clearly positively correlated, while the correlation between both methods in septic patients was only poor [37]. This finding revealed that quantification of Treg cells by QAMA detects CD4+ T cells with unmethylated FoxP3−TSDR, hidden in the CD25med/low fraction of flow cytometry.
So that although CD127 was for a long period an efficient tool to determine the phenotype and functional activity of Treg [47, 78], there were increasing controversy in comparisons with CD4+CD25+FoxP3+ cells, particularly in the context of chronic infections [77]. It became obvious, that the mere existence of an underlying disease (e.g., HIV infection) [76] or the in vitro activation [79] cause an intense CD127 down modulation on formerly CD127+ T effector cells.
FR4
The folate receptor (FR), is also known as the folic acid binding protein. There are four isoforms of the human FR: FR-α,-ß,-γ and—δ. They are expressed on epithelial cells, macrophages and malignant cells. Since FRs are overexpressed on approximately 40% of human cancers, folate conjugates have been used to deliver attached imaging and therapeutic agents selectively to malignant cells. Recently, FR- δ has been described also on human Tregs [80].
In mice has been described highly homologous molecule to human FR-δ, called FR4 [81].
Studies in mice determined that FR4 is expressed at particularly high amounts in nTregs and plays an important role in the maintenance of the Treg phenotype [82].
Other surface molecules
Besides CD25, FoxP3 and above mentioned molecules, Treg cells constitutively express surface proteins like CD45RO, cytotoxic T-lymphocyte-associated protein (CTLA-4) [83, 84], activation marker HLA-DR, or glucocorticoid-induced tumour necrosis factor receptor family-related protein (GITR) [10, 25], CD45RB, CD62L [85], neuropilin 1 (NRP1, CD304), CD103, CD195 (CCR5), ICOS [57] or lymphocyte activation gene LAG-3, that show enriched expression in Treg cell populations [86]. Unfortunately, all these markers exhibit lack of specifity, because they are neither present in 100% of Tregs or exclusive of this cell lineage and this phenotype is also shared by memory TH cells or activated Th cells. Tregs also epress high amount of chemokine receptor CCR4 (CD194), which is the reason why they can migrate to several different types of chemotactic ligand CCL17- and CCL22-secreting tumors to facilitate tumor cell evasion from immune surveillance [87].
Conclusion
Finding a speciï¬c cell surface marker of Treg cells which would allow their identification and isolation was the topic of a past decade. Presumably no other Treg-specific positive surface marker shall be identified in the future, but revealing the molecule CD26 as another negative marker in combination with other listed molecules could slowly amend our possibilities in distinguishing this important population from effector T cells and improve our diagnostic possibilities.
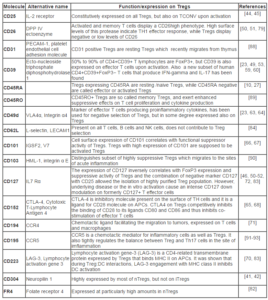
Table 1: The most important surface molecules on Tregs and their role.
References
1. Sakaguchi, S., Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol, 2004. 22: p. 531-62.
Publisher
2. Jonuleit, H., et al., Identification and functional characterization of human CD4(+)CD25(+) T cells with regulatory properties isolated from peripheral blood. J Exp Med, 2001. 193(11): p. 1285-94.
Publisher – Google Scholar
3. Sakaguchi, S., et al., Foxp3+ CD25+ CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol Rev, 2006. 212: p. 8-27.
Publisher
4. Fehervari, Z. and S. Sakaguchi, CD4+ Tregs and immune control. J Clin Invest, 2004. 114(9): p. 1209-17.
Publisher – Google Scholar
5. Prado, C., et al., Relationship between FOXP3 positive populations and cytokine production in systemic lupus erythematosus. Cytokine, 2012. 61(1): p. 90-6.
Publisher
6. Curotto de Lafaille, M.A. and J.J. Lafaille, Natural and adaptive foxp3+ regulatory T cells: more of the same or a division of labor? Immunity, 2009. 30(5): p. 626-35.
Publisher – Google Scholar
7. Sakaguchi, S., et al., Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol, 1995. 155(3): p. 1151-64.
8. Fontenot, J.D., M.A. Gavin, and A.Y. Rudensky, Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol, 2003. 4(4): p. 330-6.
Publisher – Google Scholar
9. Hori, S., T. Nomura, and S. Sakaguchi, Control of regulatory T cell development by the transcription factor Foxp3. Science, 2003. 299(5609): p. 1057-61.
Publisher
10. Sakaguchi, S., et al., FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol, 2010. 10(7): p. 490-500.
Publisher
11. Levings, M.K., R. Sangregorio, and M.G. Roncarolo, Human cd25(+)cd4(+) t regulatory cells suppress naive and memory T cell proliferation and can be expanded in vitro without loss of function. J Exp Med, 2001. 193(11): p. 1295-302.
Publisher
12. Suvas, S., et al., CD4+CD25+ T cells regulate virus-specific primary and memory CD8+ T cell responses. J Exp Med, 2003. 198(6): p. 889-901.
Publisher – Google Scholar
13. Dieckmann, D., et al., Ex vivo isolation and characterization of CD4(+)CD25(+) T cells with regulatory properties from human blood. J Exp Med, 2001. 193(11): p. 1303-10.
Publisher
14. Grossman, W.J., et al., Human T regulatory cells can use the perforin pathway to cause autologous target cell death.Immunity, 2004. 21(4): p. 589-601.
Publisher – Google Scholar
15. Onishi, Y., et al., Foxp3+ natural regulatory T cells preferentially form aggregates on dendritic cells in vitro and actively inhibit their maturation. Proc Natl Acad Sci U S A, 2008. 105(29): p. 10113-8.
Publisher – Google Scholar
16. Wing, K., et al., CTLA-4 control over Foxp3+ regulatory T cell function. Science, 2008. 322(5899): p. 271-5.
Publisher
17. Qureshi, O.S., et al., Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science, 2011. 332(6029): p. 600-3.
Publisher – Google Scholar
18. Veldhoen, M., et al., Modulation of dendritic cell function by naive and regulatory CD4+ T cells. J Immunol, 2006. 176(10): p. 6202-10.
19. Nakamura, K., A. Kitani, and W. Strober, Cell contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med, 2001. 194(5): p. 629-44.
Publisher – Google Scholar
20. Levings, M.K., et al., Human CD25+CD4+ T suppressor cell clones produce transforming growth factor beta, but not interleukin 10, and are distinct from type 1 T regulatory cells. J Exp Med, 2002. 196(10): p. 1335-46.
Publisher – Google Scholar
21. Jutel, M. and C.A. Akdis, T-cell subset regulation in atopy. Curr Allergy Asthma Rep, 2011. 11(2): p. 139-45.
Publisher – Google Scholar
22. Moore, K.W., et al., Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol, 2001. 19: p. 683-765.
Publisher
23. Collison, L.W., et al., The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature, 2007. 450(7169): p. 566-9.
Publisher – Google Scholar
24. Borsellino, G., et al., Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood, 2007. 110(4): p. 1225-32.
Publisher – Google Scholar
25. Baecher-Allan, C., et al., CD4+CD25high regulatory cells in human peripheral blood. J Immunol, 2001. 167(3): p. 1245-53.
26. Sakaguchi, S., Naturally arising Foxp3-expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat Immunol, 2005. 6(4): p. 345-52.
Publisher – Google Scholar
27. Miyara, M., et al., Functional delineation and differentiation dynamics of human CD4+ T cells expressing the FoxP3 transcription factor. Immunity, 2009. 30(6): p. 899-911.
Publisher – Google Scholar
28. Buckner, J.H. and S.F. Ziegler, Regulating the immune system: the induction of regulatory T cells in the periphery. Arthritis Res Ther, 2004. 6(5): p. 215-22.
Publisher – Google Scholar
29. Wang, J., et al., Transient expression of FOXP3 in human activated nonregulatory CD4+ T cells. Eur J Immunol, 2007. 37(1): p. 129-38.
Publisher – Google Scholar
30. Gavin, M.A., et al., Single-cell analysis of normal and FOXP3-mutant human T cells: FOXP3 expression without regulatory T cell development. Proc Natl Acad Sci U S A, 2006. 103(17): p. 6659-64.
Publisher – Google Scholar
31. Ziegler, S.F., FOXP3: not just for regulatory T cells anymore. Eur J Immunol, 2007. 37(1): p. 21-3.
Publisher – Google Scholar
32. Ziegler, S.F., FOXP3: of mice and men. Annu Rev Immunol, 2006. 24: p. 209-26.
Publisher – Google Scholar
33. Tran, D.Q., H. Ramsey, and E.M. Shevach, Induction of FOXP3 expression in naive human CD4+FOXP3 T cells by T-cell receptor stimulation is transforming growth factor-beta dependent but does not confer a regulatory phenotype. Blood, 2007. 110(8): p. 2983-90.
Publisher – Google Scholar
34. Zheng, Y. and A.Y. Rudensky, Foxp3 in control of the regulatory T cell lineage. Nat Immunol, 2007. 8(5): p. 457-62.
Publisher – Google Scholar
35. Wieczorek, G., et al., Quantitative DNA methylation analysis of FOXP3 as a new method for counting regulatory T cells in peripheral blood and solid tissue. Cancer Res, 2009. 69(2): p. 599-608.
Publisher – Google Scholar
36. Baron, U., et al., DNA demethylation in the human FOXP3 locus discriminates regulatory T cells from activated FOXP3(+) conventional T cells. Eur J Immunol, 2007. 37(9): p. 2378-89.
37. Tatura, R., et al., Quantification of regulatory T cells in septic patients by real-time PCR-based methylation assay and flow cytometry. PLoS One, 2012. 7(11): p. e49962.
Publisher – Google Scholar
38. Getnet, D., et al., A role for the transcription factor Helios in human CD4(+)CD25(+) regulatory T cells. Mol Immunol, 2010. 47(7-8): p. 1595-600.
Publisher – Google Scholar
39. Thornton, A.M., et al., Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol, 2010. 184(7): p. 3433-41.
Publisher – Google Scholar
40. Himmel, M.E., et al., Helios+ and Helios- Cells Coexist within the Natural FOXP3+ T Regulatory Cell Subset in Humans. J Immunol, 2013. 190(5): p. 2001-8.
Publisher
41. Weiss, J.M., et al., Neuropilin 1 is expressed on thymus-derived natural regulatory T cells, but not mucosa-generated induced Foxp3+ T reg cells. J Exp Med, 2012. 209(10): p. 1723-42, S1.
42. Yadav, M., et al., Neuropilin-1 distinguishes natural and inducible regulatory T cells among regulatory T cell subsets in vivo. J Exp Med, 2012. 209(10): p. 1713-22, S1-19.
43. Bruno, L., et al., Runx proteins regulate Foxp3 expression. J Exp Med, 2009. 206(11): p. 2329-37.
Publisher – Google Scholar
44. Kitoh, A., et al., Indispensable role of the Runx1-Cbfbeta transcription complex for in vivo-suppressive function of FoxP3+ regulatory T cells. Immunity, 2009. 31(4): p. 609-20.
Publisher – Google Scholar
45. Klunker, S., et al., Transcription factors RUNX1 and RUNX3 in the induction and suppressive function of Foxp3+ inducible regulatory T cells. J Exp Med, 2009. 206(12): p. 2701-15.
Publisher – Google Scholar
46. Takahashi, T., et al., Immunologic self-tolerance maintained by CD25+CD4+ naturally anergic and suppressive T cells: induction of autoimmune disease by breaking their anergic/suppressive state. Int Immunol, 1998. 10(12): p. 1969-80.
Publisher – Google Scholar
47. Liu, W., et al., CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J Exp Med, 2006. 203(7): p. 1701-11.
Publisher – Google Scholar
48. Aerts, N.E., et al., Activated T cells complicate the identification of regulatory T cells in rheumatoid arthritis. Cell Immunol, 2008. 251(2): p. 109-15.
Publisher – Google Scholar
49. Jourdan, P., et al., Cytokines and cell surface molecules independently induce CXCR4 expression on CD4+ CCR7+ human memory T cells. J Immunol, 2000. 165(2): p. 716-24.
50. Ohnuma, K., N.H. Dang, and C. Morimoto, Revisiting an old acquaintance: CD26 and its molecular mechanisms in T cell function. Trends Immunol, 2008. 29(6): p. 295-301.
Publisher
51. Cordero, O.J., et al., Interleukin-12 enhances CD26 expression and dipeptidyl peptidase IV function on human activated lymphocytes. Immunobiology, 1997. 197(5): p. 522-33.
Publisher
52. Salgado, F.J., et al., CD26: a negative selection marker for human Treg cells. Cytometry A, 2012. 81(10): p. 843-55.
Publisher – Google Scholar
53. Deaglio, S., et al., Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med, 2007. 204(6): p. 1257-65.
Publisher – Google Scholar
54. Mandapathil, M., et al., Isolation of functional human regulatory T cells (Treg) from the peripheral blood based on the CD39 expression. J Immunol Methods, 2009. 346(1-2): p. 55-63.
Publisher – Google Scholar
55. Maliszewski, C.R., et al., The CD39 lymphoid cell activation antigen. Molecular cloning and structural characterization. J Immunol, 1994. 153(8): p. 3574-83.
56. Leal, F.E., et al., Expansion in CD39(+) CD4(+) Immunoregulatory T Cells and Rarity of Th17 Cells in HTLV-1 Infected Patients Is Associated with Neurological Complications. PLoS Negl Trop Dis, 2013. 7(2): p. e2028.
Publisher – Google Scholar
57. Schulze Zur Wiesch, J., et al., Comprehensive analysis of frequency and phenotype of T regulatory cells in HIV infection: CD39 expression of FoxP3+ T regulatory cells correlates with progressive disease. J Virol, 2010. 85(3): p. 1287-97.
Publisher – Google Scholar
58. Fletcher, J.M., et al., CD39+Foxp3+ regulatory T Cells suppress pathogenic Th17 cells and are impaired in multiple sclerosis. J Immunol, 2009. 183(11): p. 7602-10.
Publisher – Google Scholar
59. Ndhlovu, L.C., et al., A novel human CD4+ T-cell inducer subset with potent immunostimulatory properties. Eur J Immunol, 2009. 40(1): p. 134-41.
Publisher – Google Scholar
60. Alam, M.S., et al., CD73 is expressed by human regulatory T helper cells and suppresses proinflammatory cytokine production and Helicobacter felis-induced gastritis in mice. J Infect Dis, 2009. 199(4): p. 494-504.
Publisher – Google Scholar
61. Moncrieffe, H., et al., High expression of the ectonucleotidase CD39 on T cells from the inflamed site identifies two distinct populations, one regulatory and one memory T cell population. J Immunol, 2010. 185(1): p. 134-43.
Publisher – Google Scholar
62. Seddiki, N., et al., Persistence of naive CD45RA+ regulatory T cells in adult life. Blood, 2006. 107(7): p. 2830-8.
Publisher – Google Scholar
63. Hoffmann, P., et al., Only the CD45RA+ subpopulation of CD4+CD25high T cells gives rise to homogeneous regulatory T-cell lines upon in vitro expansion. Blood, 2006. 108(13): p. 4260-7.
Publisher – Google Scholar
64. Kleinewietfeld, M., et al., CD49d provides access to “untouched” human Foxp3+ Treg free of contaminating effector cells. Blood, 2009. 113(4): p. 827-36.
Publisher – Google Scholar
65. Venken, K., et al., Compromised CD4+ CD25(high) regulatory T-cell function in patients with relapsing-remitting multiple sclerosis is correlated with a reduced frequency of FOXP3-positive cells and reduced FOXP3 expression at the single-cell level. Immunology, 2008. 123(1): p. 79-89.
Publisher – Google Scholar
66. Jovanovic, D.V., et al., CD101 expression and function in normal and rheumatoid arthritis-affected human T cells and monocytes/macrophages. J Rheumatol, 2011. 38(3): p. 419-28.
Publisher
67. Fernandez, I., et al., CD101 surface expression discriminates potency among murine FoxP3+ regulatory T cells. J Immunol, 2007. 179(5): p. 2808-14.
68. Seddiki, N., et al., Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J Exp Med, 2006. 203(7): p. 1693-700.
Publisher – Google Scholar
69. Venet, F., et al., Increased circulating regulatory T cells (CD4(+)CD25 (+)CD127 (-)) contribute to lymphocyte anergy in septic shock patients. Intensive Care Med, 2009. 35(4): p. 678-86.
Publisher – Google Scholar
70. Montes, M., et al., Normalization of FoxP3(+) regulatory T cells in response to effective antiretroviral therapy. J Infect Dis, 2010. 203(4): p. 496-9.
Publisher – Google Scholar
71. Yu, N., et al., CD4(+)CD25 (+)CD127 (low/-) T cells: a more specific Treg population in human peripheral blood. Inflammation, 2012. 35(6): p. 1773-80.
Publisher – Google Scholar
72. Ukena, S.N., et al., Human regulatory T cells of G-CSF mobilized allogeneic stem cell donors qualify for clinical application. PLoS One, 2012. 7(12): p. e51644.
Publisher – Google Scholar
73. Ukena, S.N., et al., Isolation strategies of regulatory T cells for clinical trials: phenotype, function, stability, and expansion capacity. Exp Hematol, 2011. 39(12): p. 1152-60.
Publisher
74. Hartigan-O’Connor, D.J., et al., Human CD4+ regulatory T cells express lower levels of the IL-7 receptor alpha chain (CD127), allowing consistent identification and sorting of live cells. J Immunol Methods, 2007. 319(1-2): p. 41-52.
Publisher
75. Tenorio, A.R., et al., The relationship of T-regulatory cell subsets to disease stage, immune activation, and pathogen-specific immunity in HIV infection. J Acquir Immune Defic Syndr, 2008. 48(5): p. 577-80.
Publisher
76. Del Pozo-Balado Mdel, M., et al., CD4(+)CD25(+/hi)CD127(lo) phenotype does not accurately identify regulatory T cells in all populations of HIV-infected persons. J Infect Dis, 2009. 201(3): p. 331-5.
Publisher
77. Rios, C.M., P.A. Velilla, and M.T. Rugeles, Chronically HIV-1 Infected Patients Exhibit Low Frequencies of CD25+ Regulatory T Cells. Open Virol J, 2012. 6: p. 49-58.
Publisher – Google Scholar
78. Roncador, G., et al., Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level. Eur J Immunol, 2005. 35(6): p. 1681-91.
Publisher – Google Scholar
79. Alves, N.L., et al., Differential regulation of human IL-7 receptor alpha expression by IL-7 and TCR signaling. J Immunol, 2008. 180(8): p. 5201-10.
80. Low, P.S. and S.A. Kularatne, Folate-targeted therapeutic and imaging agents for cancer. Curr Opin Chem Biol, 2009. 13(3): p. 256-62.
Publisher
81. Elnakat, H. and M. Ratnam, Distribution, functionality and gene regulation of folate receptor isoforms: implications in targeted therapy. Adv Drug Deliv Rev, 2004. 56(8): p. 1067-84.
Publisher
82. Tian, Y., et al., A novel splice variant of folate receptor 4 predominantly expressed in regulatory T cells. BMC Immunol, 2012. 13: p. 30.
Publisher – Google Scholar
83. Corthay, A., How do regulatory T cells work? Scand J Immunol, 2009. 70(4): p. 326-36.
Publisher – Google Scholar
84. Oderup, C., et al., Cytotoxic T lymphocyte antigen-4-dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory T-cell-mediated suppression. Immunology, 2006. 118(2): p. 240-9.
Publisher – Google Scholar
85. Fazekas de St Groth, B. and A.L. Landay, Regulatory T cells in HIV infection: pathogenic or protective participants in the immune response? AIDS, 2008. 22(6): p. 671-83.
Publisher
86. Banham, A.H., Cell-surface IL-7 receptor expression facilitates the purification of FOXP3(+) regulatory T cells. Trends Immunol, 2006. 27(12): p. 541-4.
Publisher – Google Scholar
87. Chang, D.K., et al., Humanization of an anti-CCR4 antibody that kills cutaneous T-cell lymphoma cells and abrogates suppression by T-regulatory cells. Mol Cancer Ther, 2012. 11(11): p. 2451-61.
Publisher
88. Marson, A., et al., Foxp3 occupancy and regulation of key target genes during T-cell stimulation. Nature, 2007. 445(7130): p. 931-5.
Publisher – Google Scholar
89. Walter, G.J., et al., Interaction with activated monocytes enhances cytokine expression and suppressive activity of human CD4+CD45ro+CD25+CD127(low) regulatory T cells. Arthritis Rheum, 2013. 65(3): p. 627-38.
Publisher
90. Huehn, J., et al., Developmental stage, phenotype, and migration distinguish naive- and effector/memory-like CD4+ regulatory T cells. J Exp Med, 2004. 199(3): p. 303-13.
Publisher – Google Scholar
91. Dobaczewski, M., et al., CCR5 signaling suppresses inflammation and reduces adverse remodeling of the infarcted heart, mediating recruitment of regulatory T cells. Am J Pathol, 2010. 176(5): p. 2177-87.
Publisher – Google Scholar
92. Kroetz, D.N. and G.S. Deepe, Jr., CCR5 dictates the equilibrium of proinflammatory IL-17+ and regulatory Foxp3+ T cells in fungal infection. J Immunol, 2010. 184(9): p. 5224-31.
Publisher – Google Scholar
93. Zhang, N., et al., Regulatory T cells sequentially migrate from inflamed tissues to draining lymph nodes to suppress the alloimmune response. Immunity, 2009. 30(3): p. 458-69.
Publisher – Google Scholar