Introduction
We are living in a rapidly aging world. The Centre for Disease Control and Preventions (2003) asserted that we could expect to see almost 1 billion persons aged 65 and older in 2030 worldwide. In Canada alone, Statistics Canada (2010a) estimated that the number of older persons could increase to 15 million in the next 50 years. That is to say, persons aged 65 or old would represent 28% of the projected Canadian population by 2061. Previous research estimated that 6% to 10% of individuals aged 65 or older, 5% to 15% of those aged 70 and older, and as much as 40% of those aged 85 and older suffer from dementia (Chapman et al., 2006). Together, these figures implied that we will face an unprecedented number of age-related medical adversities, including Alzheimer’s disease and other forms of dementia, in a very near future. Furthermore, the latest projections from the US Alzheimer’s Association (2010) suggested that one diagnosis of Alzheimer’s disease was made every 70 seconds presently in America. This time would decrease to every 33 seconds within the next four decades. Canada faced with a very similar situation that the number of individuals living with Alzheimer’s disease and other forms of dementia could reach 1.1 million (CIHI, 2010).
Dementia is not a single disease, but a collection of syndromes that are characterized by a plethora of cognitive, emotional and behavioural impairments (Chapman et al., 2006). To-date, only a few risk factors are identified, including family history, education, head injury, genetic predisposition and age (The Canadian Study of Health and Aging [CSHA] 1994a; Jorm 1997; Hall et al., 1998; Myhrer, 1998; Lindsay et al., 2002). However, as there is still no cure for dementia and only 9% of the cases are reversible (Chapman et al., 2006), we need to examine how we can care for these individuals and how to improve their quality of life (QOL).
Dementia Care around the World
Dementia care is a major global issue that concerns us both medically and socially (Ross-Kerr et al., 2003, Shanley, 2006). It also imposes a tremendous socioeconomic cost on our society (Zhu et al., 2006; Yaari and Corey-Bloom, 2007). The US Alzheimer’s Association (2010) stated that the country’s medicare payments for individuals with Alzheimer’s disease and other forms of dementia were estimated to be $172 billion in 2010. With our aging society, dementia care is an issue that we cannot afford to ignore.
The majority of the studies that examined dementia care were conducted in the US (Gaugler et al., 2003; Ready et al., 2004; Carroll et al., 2005), Japan (Miyamoto et al., 2002; Suwa 2002; Hosaka and Sugiyama, 2003; Fukushima et al., 2005), Korea (Kim et al., 2002), Australia and New Zealand (Shanley, 2006), UK (Upton and Reed, 2005; Hoe et al., 2005; Hoskins et al., 2005; Reilly et al., 2006) and the rest of Europe, including Italy (Balla et al., 2007), the Netherlands (Droes et al., 2004a; Droes et al., 2004b), Germany (Zank and Schacke 2002; Zank and Frank 2002), Denmark (Vogel, 2006), Sweden (Ericson et al., 2001, Andren and Elmstahl 2005; Mavall and Thorslund, 2007), France (Thomas et al., 2002; Gramain et al., 2004). In Canada, a landmark study of dementia was conducted by the Canadian Study on Health and Aging (CSHA) working group in the early 1990’s. In their reports (CSHA Working Group 1994b, 2000), the working group estimated that 1 in 13 individuals aged 65 and over had dementia, and the risk doubled for every five years of life after 65. However, only a handful of Canadian studies were conducted to determine the relationship between ADPs and QOL among older individuals and their caregivers. In a study that involved 14 ADPs in Alberta, Warren et al. (2003) examined the impact of ADPs on family caregivers of the elderly. They found that caregiver burden, QOL of caregivers and the perceived health status remained constant over time after attending ADPs. Nonetheless, the lack of improvement in outcome measures, and particularly the lack of a control group in the study, made it impossible to evaluate the findings.
Baumgarten et al. (2002) conducted a randomized trial on the satisfaction of ADPs for frail elderly in the Province of Quebec, Canada. Although the study found that clients and their family caregivers were generally satisfied with the service, there was no evidence that suggested reductions neither in the clients’ anxiety, depression and functional status nor in the family caregivers’ burden. The authors attributed the non-positive findings to the low participation of ADPs. Other Canadian research studies, Burdz et al. (1988), Chappell et al. (2001) and Bartfay and Bartfay (2013) also focused on caregivers. It is noteworthy to mention that of the aforementioned Canadian studies, only Burdz et al. (1988) was conducted specifically on individuals with dementia, and all other Canadian studies mainly concerned with the impacts on caregivers.
Adult Day Programs (ADPs) as a Form of Caring Intervention
Caring for the elderly, particularly those with dementia, is physically and emotionally arduous (Lee and Cameron, 2004; Upton and Reed, 2005; Andren and Elmstahl, 2005). In Canada, our ever escalating health care costs have prompted policy makers to put more emphasis on community care in recent health care restructuring as a way to curtail spending (Warren et al. 2003). Indeed, many older individuals prefer to live at home for as long as they can and to delay institutionalization (Zank and Schacke, 2002; Ross-Kerr et al., 2003; Warren et al., 2003). Although this stay-at-home arrangement is favoured by many, family members often assume the primary care-giving role and suffer from many physical and emotional consequences (Gramain and Malavolti 2004; Gitlin et al., 2006; Yaari and Corey-Bloom, 2007). ADPs have been projected to reduce family strain on caring for the elderly (Gaugler et al., 2003; Gramain and Malavolti, 2004). In fact, interventions are now emerging as an important aspect of dementia care (Chapman et al., 2006). Day time care was originated in Moscow in the 1930’s and further developed in the UK after the Second World War (Ross-Kerr et al., 2003; Gaugler et al., 2003). In Canada, day time services came into existence in Montreal in the 1940’s, and it became popular in the US in the 1960’s (Gaugler et al., 2003). Generally speaking, day programs provide out-of-home services during daytime where clients continue to live at home. Many programs are designed to promote cognitive functioning, facilitate independence and maintain QOL and health of their clients (Edelman et al., 2004; Debelko, 2005; Chapman et al., 2006). There is evidence that suggests rapid decline in cognitive function may decrease survival in individuals with Alzheimer’s disease (Wilson et al., 2006).
Previous research showed that mental exercises, such as reading, playing games, puzzles and musical instruments were associated with decreased risk of dementia (Wilson et al., 2002a; Wilson et al., 2002b; Verghese et al., 2003). Nonetheless, studies designed to examine the outcomes and effectiveness of ADPs remained scarce and lacked rigour (Lee and Cameron 2004; Edelman et al., 2004; Jeon et al., 2005). These studies also have mixed results due to differences in case mix, program emphases, inadequate sample size, low usage of services and the lack of control groups (Gaugler and Zarit, 2001; Ross-Kerr et al., 2003; Gaugler et al., 2003; Zarit et al., 2003). Most of the evaluations also focused on caregivers, rather than on the clients (Bartfay and Bartfay, 2013; Miyamoto et al., 2002; Zank and Schacke 2002; Gaugler et al., 2003; McCann et al., 2005; Marvall and Thorslund, 2007). If ADPs are designed to benefit both clients and caregivers, we must also examine the needs of the clients.
Quality of Life (QOL)
Health-related QOL is an important aspect in evaluating an individual’s overall health status (Schneider, 2001; Hoe et al., 2005). For many individuals with Alzheimer’s disease, improving or maintaining QOL may be the best care that we can offer. It has long been argued that “QOL should be the central goal of our professional activity” (Whitehorse and Rabins, 1992). Hoe et al. (2005) argued that measuring QOL was more important than the number of symptoms one experienced. They also stated that QOL did not decrease as cognitive functions declined.
Although the issues of QOL in individuals with dementia have received some attention since the 1990’s (Ready et al., 2004), empirical assessment has largely been ignored (Schneider 2001; Logsdon et al., 2002; Edelman et al., 2004). Fortunately, researchers are slowly recognizing the importance of QOL. For example, a Japanese qualitative study found a remarkable change in attitude in individuals with dementia after attending a daycare program (Fukushima et al., 2005).
Objectives and Hypotheses of Study
Empirical studies relating to ADPs have mainly focused on the impact of family caregivers. Little attention is paid on the relationship between QOL of individuals with Alzheimer’s disease and attending ADPs. According to Chapman et al. (2006), the majority of these individuals did not receive appropriate treatments. If we are truly committed to this vulnerable population, every effort must be made to reach out to them. As most dementia, including Alzheimer’s disease, is not reversible, timely intervention is crucial to optimize their QOL. According to many researchers (Gaugler et al., 2003; Edelman et al., 2004), the benefits of ADPs for individuals with dementia needed to be explored and the broader concerns of QOL deserved further attention. With our aging population, individuals suffer from Alzheimer’s disease will only continue to grow.
Accordingly, the objective of this pilot study was to explore the effect of ADPs on the QOL of individuals with Alzheimer disease. We hypothesized that individuals with Alzheimer disease who attended ADPs had higher QOL than those who did not attend ADPs. We further hypothesized that individuals with Alzheimer’s disease who attended ADPs enjoyed comparable QOL as other ADP clients without Alzheimer’s disease.
Methods
A pilot study was conducted using a cross-sectional design in Durham, Ontario, Canada. The regional municipality of Durham is located in the southern part of Ontario, with an approximate area of 2,500 square kilometres and a population of just over 560,000 persons (Statistics Canada, 2010b). Participants were recruited at five adult day programs and at six caregiver support groups in the region, involving primary data collection using assisted self-report questionnaires and a 13-item quality-of-life scale.
Sample 1 — Adult day program (ADP) clients with Alzheimer’s disease and
sample 2 — Adult day program (ADP) clients without Alzheimer’s disease
ADPs within the area of Durham region were contacted and meetings were arranged with the managers and staff members at each site. All day programs had similar program emphases and case mix. The programs provided social, recreational and educational activities to their clients. The clients consisted of frail elderly, individuals with needs relating to physical disability, aging and mental problems as well as individuals with cognitive impairment. All ADP clients were eligible to participate if they attended the programs at the time of data collection. The only ineligibility was when the client was unable to speak, write or comprehend English, where they would be excluded from the study. No one was excluded in our pilot study.
Subject Recruitment and Data Collection Procedure for Sample 1 and Sample 2
To recruit day program clients to participate in our study, we visited the day program meetings and explained the study to the clients with the presence of day program staff members. Letters of invitation were then circulated. For clients who were interested in the study, members of the research team subsequently went over the consent form with these clients individually. For clients who were not able to complete the consent form, we approached the family member for assent when the clients were picked up at the end of the day program session. Upon completion of the consent form, the clients were given a copy of the questionnaire to complete with one-on-one assistance provided by the research team. When any participants felt uncomfortable in answering questions in public, a separate room was offered to conduct the interview. The questionnaire consists of client’s basic demographic information and a 13-item QOL scale developed by Logsdon (Logsdon et al., 1999; Logsdon et al., 2002). Participants rated each item as “poor” (score=1), “fair” (score=2), “good” (score=3) or “excellent” (score=4). In this study, we decided to adopt only one scale to measure QOL. According to Logsdon, this scale could be used on individuals with or without dementia. Thus, the same scale was used for all participants for consistency. Furthermore, the main reason for this choice was the simplicity of this scale, such that we did not impose too much burden on our subjects. Unlike many other scales (e.g., the dementia QOL scale (DQoL) is a 29-item scale (Ready et al., 2004)), Logsdon’s QOL scale consists of only 13 items and has demonstrated good validity, internal and test-retest reliability (Logsdon et al., 1999). Hoe et al. (2005) adopted the Logsdon scale and found that it was valid and reliable for use in individuals with dementia with Mini-Mental State Examination (MMSE) scores of 3-11.
Sample 3 — Alzheimer’s Disease Caregiver Support Group Clients
Due to the privacy issue at the Alzheimer’s Society of Durham Region, we were not able to contact the clients directly. As an alternative, we decided to approach caregivers as a proxy to obtain patient information. All caregiver support groups in the Durham region were organized by the Alzheimer’s Society of Durham Region. These support groups were designed to encourage caregivers to talk with others who understood or had gone through similar experiences about the issues and decisions they were facing regarding dementia care. They also provided attendees with information, emotional support and opportunities for socialization to cope with the changes in their lives. Attendees of the support groups were typically adult children and spouses of individuals with Alzheimer’s disease. Our sample included all family caregivers who attended the support groups at the time of data collection and the person they were caring for (who had been diagnosed with Alzheimer’s disease).
Subject Recruitment and Data Collection Procedure for Sample 3
With the permission of the Alzheimer Society of Durham Region, we attended the support group meetings and invited the attendees to participate in our study. The same procedure described above was followed. At the meeting, we explained the study to the attendees with the presence of a support group facilitator. A letter of invitation and a consent form were then given to the support group attendees. For individuals who agreed to participate, they were given the same questionnaire to complete with regard to the information of the person they were caring for.
Results
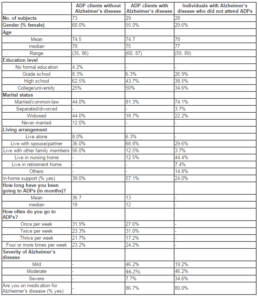
We collected information on a total of 130 individuals. In reference to AD diagnosis and disease severity, all participants were asked two simple questions: (i) ‘have you ever been told by your doctor that you have Alzheimer’s disease’ and (ii) ‘if so, what is the severity of the disease?’ Based on the answer of either ‘yes’ or ‘no’ to the first question, participants were classified into one of three groups: (1) disease-free ADP clients (n=73), (2) individuals with Alzheimer’s disease who attended ADP (n=28), and (3) individuals with Alzheimer’s disease who did not attend ADP (n=29). For the latter question, participants were asked to select his or her disease severity from mild, moderate or severe. Table 1 summarizes the basic demographic characteristics of our participants. We first compared only participants who attended ADPs. The mean age (74.5 vs. 74.7) was similar regardless of the disease status. There were slightly more females who did not have a diagnosis of Alzheimer’s disease. Furthermore, ADP clients with a diagnosis of Alzheimer’s disease were more likely to have post-secondary education (50.0% vs. 25.0%), to be married (81.3% vs. 44.0%) and living with a spouse/partner (68.8% vs. 36.0%), as well as to have in-home support (57.1% vs. 39.0%). Next, we compared only participants who had a diagnosis of Alzheimer’s disease. We found that individuals who attended ADPs were slightly younger (74.7 vs. 78.0). They were also more likely to have a post-secondary education (50.0% vs. 34.6%), to be married (81.3% vs. 74.1%), and be living with a spouse/partner (68.8% vs. 29.6%), as well as to have in-home support (57.1% vs. 24.0%). In terms of disease severity, there were equal numbers of moderate stage but ADP clients were likely to have a mild stage (46.2% vs. 19.2%), and less likely to have a severe stage of Alzheimer’s disease (7.7% vs. 34.6%).
Table 1: Demographic characteristics of participants
In accordance with our hypotheses stated earlier, we performed separate 2-sample t-tests to compare the three groups. First, we involved only individuals who attended ADPs. Our results showed that the overall QOL scores for individuals who attended ADP were similar to those of non-disease ADP clients (2.7 vs. 2.9, p=0.1). We also observed that all specific QOL scores were similar among these individuals, with the exception of the memory category (see Figure 1). In the memory category, approximately 60% of ADP clients without Alzheimer’s disease rated themselves as either good or excellent, whereas only 17% of ADP clients with Alzheimer’s disease rated themselves as either good or excellent.
Second, we involved only individuals with a diagnosis of Alzheimer’s disease. The overall QOL scores were significantly lower for individuals who did not attend ADP than those who attended ADP (1.9 vs. 2.7, p=0.0001). In addition, we noticed that individuals with Alzheimer’s disease who attended ADPs were more likely to rate each item as either good or excellent, as compared to those who did not attend ADPs. This observation held true for all QOL categories.
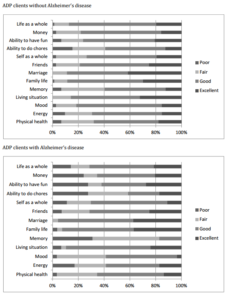
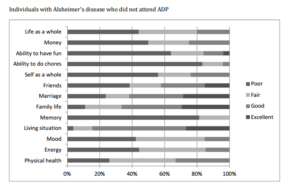
Figure 1: Study participants’ QOL scores
Discussion
Due to its subjective nature, assessment of QOL is a highly complex matter. To make matters worse, individuals with severe cognitive impairments may be unable to evaluate their own health status and/or issues relating to their QOL. To compensate this predicament, many researchers either used proxy measures (e.g., ask a family member or professional caregiver to rate the patient’s QOL), or abandoned the idea of measuring QOL altogether and concentrated on caregivers only. Our research team, however, believed it was of paramount importance to understand the needs and concerns of individuals with Alzheimer’s disease as well.
In this study, our first task was to compare the QOL of all ADP clients with or without Alzheimer’s disease. Our results showed that all ADP clients had similarly favourable QOL ratings, regardless of whether they have the disease or not. The lack of a difference in QOL ratings among individuals with or without Alzheimer’s disease may provide support for future studies to further examine the benefits of ADPs.
Our second task was to compare the QOL of individuals with Alzheimer’s disease according to whether they attended ADPs or not. There were two issues relating to this task. First, information was obtained differently for each group. For the ADP attendees who also had Alzheimer’s disease, they filled out the questionnaires themselves with a one-on-one assistance from the research team. For individuals who did not attend ADP, information was obtained by proxy (caregivers). There has been much debate over the use of patient ratings versus proxy ratings. In particular, proxy ratings may sometimes be mixed-in or affected by the proxy’s own feelings. Second, there were slightly more individuals with severe disease stage in our non-ADP groups. Although disease stage may explain some of the differences in QOL, we believed that this difference only represented a portion of the QOL. As pointed out by Hoe et al. (2005), QOL did not decrease as cognitive function decreased. Thus, our study result provided evidence that suggests that ADPs may be partially responsible for the observed differences in QOL ratings among individuals with Alzheimer’s disease who attended or did not attend ADPs, and the merits of ADP should be further examined. Given the prevailing shift of the Canadian health care system from institutional- to community-based care, future studies are desperately needed to examine all community-based interventions, including ADPs, and their effects on the QOL of individuals living with Alzheimer’s disease and other forms of dementia. As there is still no cure for Alzheimer’s disease, improving and maintaining QOL may be the best course of action that we can provide to this group of vulnerable individuals. In addition, longitudinal studies, that follow participants over time, may provide further evidence that community-based interventions can provide sustainable improvement. As dementia is a condition that also greatly affects family caregivers, studies that examine the impact of these interventions on caregivers such as caregiver strain and burden are warranted. Indeed, an understanding of how community-based services and resources for client’s with Alzheimer’s disease and their families are critical in order to provide safe and cost-effective services to this growing population.
References
1.Alzheimer’s Association. (2010) “Alzheimer’s disease facts and figures,”Alzheimer’s and Dementia, 6(2) 158-94.
Publisher – Google Scholar
2.Andren, S. and Elmstahl, S. (2005) “Family caregivers’ subjective experiences of satisfaction in dementia care: Aspects of burden, subjective health and sense of coherence,” The Scandinavian Journal of Caring Sciences, 19 157-168.
Publisher – Google Scholar
3. Balla, S, Simoncini, M, Giacometti, I, Magnano, A, Leotta, D. and Pernigotti, L. M. (2007) “The daily center care on impact of family burden,” Archives of Gerontology and Geriatrics, 44 Suppl(1) 55-59.
Google Scholar
4.Bartfay, E. and Bartfay, W. J. (2013) “Quality of life outcomes among Alzheimer’s’ disease family caregivers following community-based intervention,” Western Journal of Nursing Research, 35(1) 98-116.
Publisher – Google Scholar
5.Baumgarten, M, Lebel, P, Laprise, H, Leclerc, C. and Quinn, C. (2002) “Adult day care for the frail elderly: Outcomes, satisfaction and cost,” Journal of Aging and Health, 14(2) 237-259.
Publisher – Google Scholar
6.Burdz, M. P, Eaton, W. O. and Bond Jr., J. B. (1988) “Effect of respite care on dementia and nondementia patients and their caregivers,” Psychology and Aging, 3(1) 38-42
Publisher – Google Scholar
7.Canadian Institute for Health Information (2010). Caring for seniors with Alzheimer’s disease and other forms of dementia, CIHI: Ottawa, Ontario [Online], http://www.cihi.ca.
8.Canadian Study of Health and Aging. (1994a) “Risk factors for Alzheimer’s disease in Canada,” Neurology, 44 2073-2080.
Publisher – Google Scholar
9.Canadian Study of Health and Aging [CSHA] Working Group. (1994b) “Canadian study on health and aging: Study methods and prevalence of dementia,” Canadian Medical Association Journal, 150 899-913.
Google Scholar
10. Canadian Study of Health and Aging [CSHA] Working Group. (2000) “The incidence of dementia in Canada,” Neurology, 55 66-73.
11.Carroll, A. M, Vetor, K, Holmes, S. and Supiano, K. P. (2005) “Ask the consumer: An innovative approach to dementia-related adult day service evaluation,” American Journal of Alzheimers Disease and Other Dementia, 20(5) 290-294.
Publisher – Google Scholar
12.Centre for Disease Control and Prevention. (2003) “Trends in aging — United States and Worldwide,” MMWR Morbidity and Mortality Weekly Report, 52 101-106.
13.Chapman, D. P, Williams, S. M, Strine, T. W, Anda, R. F. and Moore, M. J. (2006) “Dementia and its implications for public health,” Preventing Chronic Disease [serial online].¬ [Retrieved April 2010], http://www.cdc.gov/pcd/issues/2006/apr/05_0167.htm.
Google Scholar
14.Chappell, N. L, Reid, R. C. and Dow, E. (2001) “Respite reconsidered: A typology of meanings based on the caregiver’s point of view,” Journal of Aging Studies, 15(2) 201-216.
Publisher – Google Scholar
15.Debelko, H. (2005) “A comparative analysis of short stays versus long stays in adult day health care programs,” Social Work in Health Care, 42(1) 57-71.
16.Droes, R. M, Breebaart, E, Meiland, F. J. M, Van Tilburg, W. and Mellenbergh, G. J. (2004a) “Effect of meeting centres support program on feelings of competence of family carers and delay institutionalization of people with dementia,” Aging and Mental Health, 8(3) 201-211.
Publisher – Google Schola
17.Droes, R. M, Meiland, F. J. M, Schmitz, M. J, Boerem, I, Derksen, E, De Lange, J, Vernooij-Dassen, M. J. F. J. and Van Tilburg, W. (2004b) “Variations in meeting centres for people with dementia and their carers, results of a multi-center implementation study,” Archives of Gerontology and Geriatrics, 9 127-147.
18.Edelman, P, Fulton, B. R. and Kuhn, D. (2004) “Comparison of dementia-specific quality of life measures in adult day centres,” Home Health Care Services Quarterly, 23(1) 25-42.
Publisher – Google Scholar
19.Ericson, I, Hellstrom, I, Lundh, U. and Nolan M. (2001) “What constitutes good care for people with dementia?” British Journal of Nursing, 10(11) 710-714.
Google Scholar
20.Fukushima, T, Nagahata, K, Ishibashi, N, Takahashi, Y. and Moriyama, M. (2005) “Quality of life from the viewpoint of patients with dementia in Japan: Nurturing through an acceptance of dementia by patients, their families and care professionals,” Health and Social Care in the Community, 13(1) 30-37.
Publisher – Google Scholar
21.Gaugler, J. E, Jarrott, S. E, Zarit, S. H, Stephens, M. A. P, Townsend, A. and Greene, R. (2003) “Respite for dementia caregivers: The effects of adult day service use on caregiving hours and care demands,” International Psychogeriatrics, 15(1) 37-58.
Publisher – Google Scholar
22.Gaugler, J. E, Zarit, S. H, Townsend, A, Stephens, M. A. P. and Greene, R. (2003) “Evaluating community-based programs for dementia caregivers: The cost implications of adult day services,” Journal of Applied Gerontology, 22(1) 118-133.
Publisher – Google Scholar
23.Gaugler, J. E. and Zarit, S. H. (2001) “The effectiveness of adult day services for disabled older people,” Journal of Aging and Social Policy, 12 23-47.
Publisher – Google Scholar
24.Gitlin, L. N, Reever, K, Dennis, M. P, Mathieu, E. and Hauck, W. W. (2006) “Enhancing quality of life of families who adult day services: Short- and long-term effects of the adult day services plus program,” The Gerontologist, 46(5) 630-639.
Publisher – Google Scholar
25.Gramain, A. and Malavolti, L. (2004) “Evaluating the effects of care programs for elderly persons with dementia on caregiver’s well being,” The European Journal of Health Economics, 5 6-14.
Publisher – Google Scholar
26.Hall, K, Gureje, O, Gao, S, Ogunniyi, A, Hui, S. L. and Baiyewu, O. (1998) “Risk factors and Alzheimer’s disease: A comparative study of two communities,” Australian and New Zealand Journal of Psychiatry, 32 698-706.
Publisher – Google Scholar
27.Hoe, J, Katona, C, Roch, B. and Livingston, G. (2005) “Use of the QOL-AD for measuring quality of life in people with severe dementia — the LASER-AD study,” Age and Ageing, 34 130-135.
Publisher – Google Scholar
28.Hosaka, T. and Sugiyama, Y. (2003) “Structured intervention in family caregivers of the demented elderly and changes in their immune function,” Psychiatry and Clinical Neurosciences, 57(2) 147-151.
Publisher – Google Scholar
29.Hoskins, S, Coleman, M. and McNeely, D. (2005) “Stress in carers of individuals with dementia and community mental health teams: An uncontrolled evaluation study,” Journal of Advanced Nursing, 50(3) 325-333.
Publisher – Google Scholar
30.Jeon, Y. H, Brodaty, H. and Chesterson, J. (2005) “Respite care for caregivers and people with severe mental illness: Literature review,” Journal of Advanced Nursing, 49(3) 297-306.
Publisher – Google Scholar
31.Jorm, A. F. (1997) “Alzheimer’s disease: risk and protection,” The Medical Journal of Australia, 167 443-446.
Google Scholar
32.Kim, N. C, Kim, H. S, Yoo, Y. S, Hahn, S. W. and Yeom, H. A. (2002) “Outcomes of day care: A pilot study on changes in cognitive function and agitated behaviors of demented elderly in Korea,” Nursing and Health Sciences, 4(1-2) 3-7.
Google Scholar
33.Lee, H. and Cameron, M. (2004) “Respite care people with dementia and their carers,” Cochrane Database Systematic Reviews, 2, CD004396.
34.Lindsay, J, Laurin, D, Verreault, R, Hebert, R, Helliwell, B. and Hill, G. B. (2002) “Risk factors for Alzheimer’s disease: A prospective analysis from the Canadian Study of Health and Aging.” American Journal of Epidemiology, 156 445-453.
Publisher – Google Scholar
35.Logsdon, R. G, Gibbons, L. E, McCurry, S. M. and Teri, L. (2002) “Assessing quality of life in older adults with cognitive impairment,” Psychosomatic Medicine, 64 510-519.
Google Scholar
36.Logsdon, R. G, Gibbons, L. E, McCurry, S. M. and Teri, L. (1999) “Quality of life in Alzheimer’s disease: patients and caregiver reports,” Journal of Mental Health and Aging, 5 21-32.
37.Mavall, L. and Thorslund, M. (2007) “Does day care also provide care for the caregivers?” Archives of Gerontology and Geriatrics, 45(2) 137-150.
Publisher – Google Scholar
38.McCann, J. J. Hebert, L. E, Li, Y, Wolinsky, F. D, Gilley, D. W, Aggarwal, N. T, Miller, J. M. and Evans, D. A. (2005) “The effect of adult day care services on time to nursing home placement in older adults with Alzheimer’s disease,” The Gerontologist, 45(6) 754-763.
Publisher – Google Scholar
39.Miyamoto, Y, Ito, H, Otsuka, T. and Kurita, H. (2002) “Caregiver burden in mobile and non-mobile demented patients: A comparative study,” International Journal of Geriatric Psychiatry, 17(8) 765-773.
Publisher – Google Scholar
40.Myhrer T. (1998) “Adverse psychological impact, glutamatergic dysfunction, and risk factors for Alzheimer’s disease,” Neuroscience and Biobehavioral Reviews, 23 131-139.
Publisher – Google Scholar
41.Ready, R. E, Ott, B. R. and Grace, J. (2004) “Patients versus informant perspectives of quality of life in mild cognitive impairment and Alzheimer’s disease,” International Journal of Geriatric Psychiatry, 19 256-265.
Publisher – Google Scholar
42.Reilly, S, Venables, D, Hughes, J, Challis, D. and Abendstern, M. (2006) “Standards of care in day hospitals and day centres: A comparison of services for older people with dementia,” International Journal of Geriatric Psychiatry, 21(5) 460-468.
Publisher – Google Scholar
43.Ross-Kerr, J. C, Warren, S, Schalm, C, Smith, D. L. and Godkin, M. D. (2003) “Adult day programs: Who needs them?” Journal of Gerontological Nursing, 29(12) 11-17.
Google Scholar
44.Schneider, L.S. (2001) “Assessing outcomes in Alzheimer disease,” Alzheimers Disease and Associated Disorders, 15(suppl 1) S8-S18.
Publisher – Google Scholar
45.Shanley, C. (2006) “Developing more flexible approaches to respite for people living with dementia and their carers,”American Journal of Alzheimers Disease and Other Dementias, 21(4) 234-241.
Publisher – Google Scholar
46.Statistics Canada. (2010a) “Population Projections for Canada, Provinces and Territories,” Retrieved from http://www.statcan.gc.ca/pub/91-520-x/2010001/aftertoc-aprestdm1-eng.htm.
47.Statistics Canada. (2010b) “2006 Community Profile,” Retrieved from http://www12.statcan.ca/census-recensement/2006/dp-pd/prof/92-591/details/Page.cfm?Lang=EandGeo1=CDandCode1=
3518andGeo2=PRandCode2=35andData=CountandSearchText=durhamandSearchType=BeginsandSearchPR=01andB1=
AllandCustom=Suwa,S. (2002) “Assessment scale for caregiver experience with dementia: Development of an instrument in Japan,” Journal of Gerontological Nursing, 28(12) 5-12.
48.homas, P, Chantoin-Merlet, S, Hazif-Thomas, C, Belmin, J, Montagne, B, Clement, J. P, Lebruchec, M. and Billon, R. (2002) “Complaints of informal caregivers providing home care for dementia patients: The Pixel study,”International Journal of Geriatric Psychiatry, 17(11) 1034-1047.
Publisher – Google Scholar
49.Upton, N. and Reed, V. (2005) “Caregiver coping in dementing illness — implications for short-term respite care,” International Journal of Psychiatry Nursing Research, 10(3) 1180-1196.
Google Scholar
50.Verghese, J, Lipton, R. B, Katz, M. J, Hall, C. B, Derby, C. A. and Kuslansky, G. (2003) “Leisure activities and the risk of dementia in the elderly,” New England Journal of Medicine, 348 2508-2516.
Publisher – Google Scholar
51.Vogel, A, Mortensen, E. L, Hasselbalch, S. G, Andersen, B. B. and Waldemar, G. (2006) “Patients versus informant reported quality of life in the earliest phases of Alzheimer’s disease,” International Journal of Geriatric Psychiatry,” 21 1132-1138.
Publisher – Google Scholar
52.Warren, S, Kerr, J. R, Smith, D, Godkin, D. and Schalm, C. (2003) “The impact of adult day programs on family caregivers of elderly relatives,” Journal of Community Health Nursing, 20(4) 209-221.
Publisher – Google Scholar
53.Wilson, R. S, Li, Y, Aggarwal, N. T, McCann, J. J, Gilley, D. W, Bienias, J. L, Barnes, L. L. and Evans, D.A. (2006)“Cognitive decline and survival in Alzheimer’s disease,” International Journal of Geriatric Psychiatry, 21 356-362.
Publisher – Google Scholar
54.Wilson, R. S, Bennett, D. A, Bienias, J. L, Aggarwal, N. T, Mendes De Leon, C. F. and Morris, M.C. (2002a)“Cognitive activity and incident AD in a population-based sample of older persons, ” Neurology, 59 1910-1914.
Publisher – Google Scholar
55.Wilson, R. S, Mendes De Leon, C. F, Barnes, L. L, Schneider, J. A, Bienias, J. L. and Evans D. A. (2002b) “Participation in cognitively stimulating activities and risk of incident Alzheimer’s disease, ” Journal of American Medical Association, 287 742-748.
Publisher – Google Scholar
56.Yaari, R. and Corey-Bloom, J. (2007) “Alzheimer’s disease, ” Seminars in Neurology, 27(1) 32-41.
57.Zank, S. and Frank, S. (2002) “Family and professional caregivers’ ratings of dementia symptoms and activities of daily living of day care patients: do differences change over time? ” Aging and Mental Health, 6(2) 161-165.
Publisher – Google Scholar
58.Zank, S. and Schacke, C. (2002) “Evaluation of geriatric day care units: Effects on patients and Caregivers, ”Journal of Gerontology, 57B(4) 348-357.
Publisher – Google Scholar
59.Zarit, S. H, Stephens, M. A. P, Townsend, A, Greene, R. and Femia, E. E. (2003) “Commentary on “Evaluation of geriatric day care units: Effects on patients and caregivers” and authors’ reply, ” Journal of Gerontology, 58B(3) 195-196.
60.Zhu, C. W, Scarmeas, N, Torgan, R, Albert, M, Brandt, J, Blacker, D, Sano, M. and Stern, Y. (2006) “Clinical features associated with costs in early AD: baseline data from the predictors study, ” Neurology, 66(7) 1021-1028.
Publisher – Google Scholar






