Introduction
Hydrogen storage is one of the biggest issues associated with the transformation of fossils fuel scenario to hydrogen economy (Schlapbach & Züttel, 2001). The available conventional hydrogen storage technologies, i.e. gaseous storage and liquid state storage, have challenges of size, safety and high energy involvement(Manna, Roy, Vashistha, & Sharma, 2013; Zhou, 2005). Hydrogen storage in solid materials, such as sorbents and hydrides, is the safest and effective method(Jain, Jain, & Jain, 2010; Manna, Vashistha, & Sharma, 2010; Orimo, Nakamori, Eliseo, Zu, & Jensen, 2007).Ammonia Borane (AB) is one of the most investigated chemical hydrides due to its enormous hydrogen content (19.6 wt%) (Gangal, Edla, et al., 2012; Stowe, Shaw, Linehan, Schmid, & Autrey, 2007). The low decomposition temperature, stability in atmospheric condition, hydrogen release by hydrolysis and thermolysis makes ammonia borane an interesting candidate for hydrogen storage application(U B Demirci, Bernard, Chiriac, Toche, & Miele, 2011; Umit B Demirci & Miele, 2010; Rakap & Ozkar, 2010). It has been reported that the decomposition of ammonia borane occurs in three steps at the temperatures of about 100°C, 140°C and above 500°C, respectively, releasing one mole of hydrogen in each step (Baitalow, Baumann, Wolf, Jaenicke-Rößler, & Leitner, 2002; Gangal, Kale, Edla, Manna, & Sharma, 2012). Along with the aforementioned advantages, certain critical issues are also consociated with ammonia borane thermal decomposition, such as, long induction period and slow kinetics of hydrogen evolution, foaming and expansion of the material on heating, release of ammonia and borazine along with hydrogen(Gangal, Edla, et al., 2012).
There are several reports available with addressing and abolishing some of the issues by partially or completely using different heating rates, processes and/or materials(Cheng, Ma, Li, & Chen, 2007; Gangal, Edla, et al., 2012; Gutowska et al., 2005; He et al., 2009). Wolf et al. analyzed the influence of the heating rate and mentioned that with low heating rates and isothermal conditions, ammonia borane decomposes below its melting point(Wolf, Baumann, Baitalow, & Hoffmann, 2000). It has been observed that at high pressure, AB decomposes completely in two steps as compared to classical three step decomposition (NyleÌn et al., 2009). Milling and doping effect on the thermal decomposition of AB is studied by Benedetto et al. (Benedetto et al., 2006). It has been found that the activation energy of the reaction decreases for doped samples (doping material: hydrogen hexachloroplatinate hydrate) and increases for milled samples. Transition metals, like cobalt and nickel, has catalytic ability on the thermal decomposition of AB (He et al., 2009). Our previous reports showed that, zeolites (Gangal, Edla, et al., 2012) and silicon nanoparticles (Gangal, Kale, et al., 2012) have potential catalytic effect on the ammonia borane thermal decomposition. The current paper deals with misch metal nanoparticles (MmNi4.5Al0.5) as a catalyst and its effect on the thermal decomposition of ammonia borane.
Experimental
Samples Preparation
The micron size misch metal with a composition of La 9.17%, Ce 17.43%, Pr 1.86%, Nd 5.34%, Ni 62.92%, Al 3.28% having LaNi5 structure was used as received and was mechanically milled in a FRITSCH Pulverette-6 ball mill to prepare the nanosized catalyst material. Misch metal was ball milled in the presence of acetone with a speed of 300 rpm for 30 h in an 80 ml stainless steel (SS) bowl and balls using a FRITSCH Pulverette-6 machine. The diameter of the SS balls used was 10 mm. The sample to ball ratio was used as 1:10. The entire milling cycle consisted of 10 min milling followed by 10 min pause to dissipate the generated heat. After ball milling, the bowl was placed inside an inert atmosphere and allowed to evaporate the solvent. The dried powder was then collected and stored in a glove box for further use.
The prepared Mm nanoparticles were mixed with ammonia borane (Sigma-Aldrich) in the MM:AB mass ratio of 1:10 and were milled for 1 h to obtain homogenous mixture of AB and catalyst. The ball to sample ratio was taken as 10:1. All the sample handling was done inside a glove box to avoid atmospheric moisture and oxygen. The milled powder of AB and Mm will be mentioned as ABMm, throughout the paper.
Characterization and Thermal Decomposition
The prepared ABMm samples and their decompositions products were characterized by X-ray diffractometry (XRD) using Philips X-ray Diffractometer using Cu Kα radiation having wavelength 1.54 Å. The bond connectivity within the samples was observed by Fourier Transform Infrared Spectroscopy (FTIR) using Jasco FTIR system in the range of 400 cm-1 to 4000 cm-1. Non-Isothermal decomposition of the sample was studied in a Perkin Elmer TG-DTA system from room temperature to 200 °C with heating rate of 3°C/min. Isothermal decomposition was studied using a standard Sievert’s apparatus (Kale, Gangal, Edla, & Sharma, 2011) at 90, 110, 135 and 150°C. Gas evolved during the isothermal decomposition at 150°C was transferred from the reactor to a gas sampler by water displacement method. Subsequently, gas was injected from the sampler into the GC instrument to quantize the gas evolved from the samples.
Results and discussion
Characterization
X-ray diffraction pattern of AB and ABMm, before and after decomposition is compared in the Fig. 1. The sample ABMm shows distinct peaks for ammonia borane as well as Mm, which clearly indicates their uniform distribution and physical mixing during the course of ball milling. The absence of any chemical changes and ionic substitution is also furnished from the obtained X-ray diffraction data. The diffraction pattern of decomposed product of ABMm has only one broad peak in the range 20-30Ëš, which may be formed due to the formation of amorphous product (NHxBHx) after heat treatment.
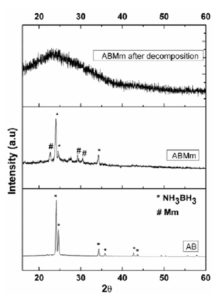
Figure 1: XRD pattern of AB ABMm and ABMmafter decomposition
FTIR spectrum of the ABMm and the decomposed product after heating at different temperature are shown in Fig 2. The spectrum of AB has the characteristic frequency bands corresponding to B-N near 500 cm-1, B-H in the range of 1000 to 1500 cm-1, N-H near 1600 cm-1, B-H near 2300 cm-1 and N-H near 3300 cm-1(Gangal, Kale, et al., 2012; Hu, Geanangel, & Wendlandt, 1978). As the decomposition temperature increases, N-H and B-H bands are broadened and finally disappear. Disappearance of these two bonds is the prime evidence of hydrogen formation from AB molecules. It can also be seen from the figure that B-N band near 500 cm-1 is not disrupted even after decomposition at 150 °C which rules out the breaking of ammonia borane molecule (H3N-BH3) at the middle and therefore there is no possibility of ammonia or diborane gas formation.
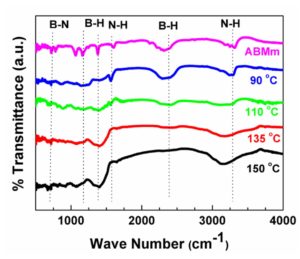
Figure 2: FTIR Spectrum of AB Mm before and after decomposition at different temperature
Thermal Decomposition of AB and ABMm
TG-DTA
The mass loss and DTA profiles of AB and ABMm recorded by TG-DTA analyser are shown in Fig 3. Both of the samples were heated upto 300ËšC with a heating rate of 3ËšC/min in nitrogen atmosphere. For pristine ammonia borane (AB), there is an onset temperature at 108.5ËšC and a second onset is at about 130ËšC. Other two humps around 150 and 173.5ËšC are also observed. Around 195-200ËšC, mass loss stabilizes at 31.5wt %. For ABMm sample, mass loss started as soon as heating started. Mass loss can be seen after 50ËšC. The melting event for ABMm sample gets delayed due to the addition of Mm nanoparticles. The decrease in the exothermicity of the decomposition reaction may be due to the thermal runway after addition of misch metal. On the first event for ABMm, total mass loss is around 3 wt%. Afterwards, gas evolution started around 100ËšC and completed at 250ËšC, with a total mass loss of 25.4 wt%. The mass loss during the heating and hydrogen evolution from the samples were closely related. NH3BH3 contains 19.6% hydrogen by mass; the excess mass loss may be due to the evolution of other gases, c.a. diborane, ammonia etc.
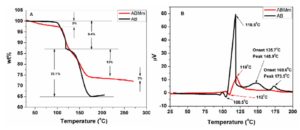
Figure 3: TGA (left) and DTA (right) plots for AB and ABMm
Isothermal Decomposition
Fig 4 (a,b,c,d) shows the isothermal decomposition profiles for AB and ABMm samples. It has been evident from the figure that at 90°C, hydrogen evolution is started very fast for ABMm sample than that of AB sample. Absence of induction period is also observed for ABMm sample, which is in well agreement with the obtained TG-DTA result. The amount of hydrogen gas evolved from AB, and ABMm samples are almost comparable for all the temperature. With the increase in temperature, the amount of hydrogen released increases and the induction period reduces. The amount of gas released in case of isothermal experiments is in the theoretical limits of hydrogen release from AB, thus it can be inferred that only hydrogen is released when AB is decomposed isothermally in a constant volume apparatus. GC of evolved gases of isothermal decomposition reaction on Fig. 5 also confirms the release of only hydrogen and the unwanted gaseous species get suppressed. At 110, 135 and 150°C, hydrogen evolution plots are almost comparable for both samples. The effect of misch metal is not prominent in those cases. This has been occurred possibly due to the destabilization of ammonia borane after its melting at higher temperature.
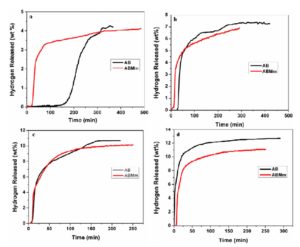
Figure-4 Isothermal decomposition plots of AB and ABMm at (a)90°C, (b)100°C,(c)110°C and (d)150°C
Gas chromatograph was recorded for the gases evolved from decomposition of AB and ABMm samples after decomposition at 150°C. Gaseous products were transferred to the GC instrument from Sieverts apparatus using a gas sampler by water displacement method. Fig. 5 shows the gas chromatograph for ABMm isothermal decomposition at 150°C. Two peaks can be observed from the figure. The broad and highest peak is attributed to the formed hydrogen gas whereas the small peak appeared due to the air or nitrogen, which was possibly trapped during the gas transfer to the sampler. Thus, it can be inferred that the gas released during the isothermal decomposition is only hydrogen. Similar plots were also obtained for AB sample.
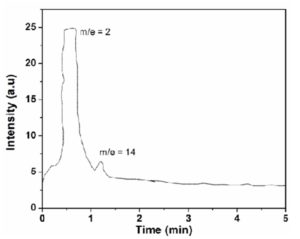
Figure 5 : Chromatograph of gas Evolved of isothermal decomposition of ABMm
Conclusions
Misch metal nanoparticles are found to be a good catalyst for ammonia borane thermal decomposition by reducing the induction period for AB decomposition. Reduction in induction period can be attributed to the breaking of the dihydrogen bond and assisting formation of diammoniate of diborane (DADB) Gangal et al., 2012. In the isothermal decomposition of AB and ABMm, the unwanted gaseous species were suppressed completely and only hydrogen was evolved. The presence of the B-N frequencies in the FTIR spectra after decomposition also confirms the formation of pure hydrogen in case of isothermal decomposition.
Acknowledgements
Authors are thankful to the Ministry of New and Renewable Energy (MNRE), Govt. of India for funding the research work. Authors are also thankful to Dr. I. P. Jain, Rajasthan University for providing catalyst material.
References
1. Baitalow, F., Baumann, J., Wolf, G., Jaenicke-Rößler, K., & Leitner, G. (2002). “Thermal decomposition of B—N—H compounds investigated by using combined thermoanalytical methods” Thermochimica Acta, 391(1-2), 159—168.
Publisher – Google Scholar
2. Benedetto, S. De, Carewska, M., Cento, C., Gislon, P., Pasquali, M., Scaccia, S., & Prosini, P. P. (2006).
“Effect of milling and doping on decomposition of NH3BH3 complex” Thermochimica Acta, 441(2), 184—190.
Publisher – Google Scholar
3. Cheng, F., Ma, H., Li, Y., & Chen, J. (2007). Ni1-xPtx (x 0−0.12) “Hollow Spheres as Catalysts for Hydrogen generation from ammonia borane” Inorganic Chemistry, 46, 788—794.
Publisher – Google Scholar
4. Demirci, U. B., Bernard, S., Chiriac, R., Toche, F., & Miele, P. (2011). “Hydrogen release by thermolysis of ammonia borane NH3BH3 and then hydrolysis of its by-product [BNHx]”. Journal of Power Sources, 196(1), 279—286.
Publisher – Google Scholar
5. Demirci, U. B., & Miele, P. (2010). “Hydrolysis of solid ammonia borane”. Journal of Power Sources, 195(13), 4030—4035.
Publisher – Google Scholar
6. Gangal, A. C., Edla, R., Iyer, K., Biniwale, R., Vashistha, M., & Sharma, P. (2012). “Effect of zeolites on thermal decomposition of ammonia borane” International Journal of Hydrogen Energy, 37(4), 3712—3718.
Publisher – Google Scholar
7. Gangal, A. C., Kale, P., Edla, R., Manna, J., & Sharma, P. (2012). “Study of kinetics and thermal decomposition of ammonia borane in presence of silicon nanoparticles” International Journal of Hydrogen Energy, 37(8), 6741—6748.
Publisher – Google Scholar
8. Gutowska, A., Li, L., Shin, Y., Wang, C. M., Li, X. S., Linehan, J. C., Autrey, T. (2005). “Nanoscaffold mediates hydrogen release and the reactivity of ammonia borane” Angewandte Chemie (Int Ed.), 44, 3578—82.
Publisher – Google Scholar
9. He, T., Xiong, Z., Wu, G., Chu, H., Wu, C., Zhang, T., & Chen, P. (2009). “Nanosized Co- and Ni-Catalyzed Ammonia Borane for Hydrogen Storage” Chemistry of Materials, 21(11), 2315—2318.
Publisher – Google Scholar
10. Hu, M. G., Geanangel, R. A., & Wendlandt, W. W. (1978). “The thermal decomposition of ammonia borane” Thermochimica Acta, 23, 249—255.
11. Jain, I. P., Jain, P., & Jain, A. (2010). “Novel hydrogen storage materials: A review of lightweight complex hydrides”Journal of Alloys and Compounds, 503(2), 303—339.
Publisher – Google Scholar
12. Kale, P., Gangal, A. C., Edla, R., & Sharma, P. (2011). “Investigation of hydrogen storage behavior of silicon nanoparticles” International Journal of Hydrogen Energy, 37(4), 3741—3747.
13. Manna, J., Roy, B., Vashistha, M., & Sharma, P. (2013). “Effect of Co+2/BH4− ratio in the synthesis of Co—B catalysts on sodium borohydride hydrolysis” International Journal of Hydrogen Energy, 39 (1), 406—413
14. Manna, J., Vashistha, M., & Sharma, P. (2010). “Lithium Borohydride as a Hydrogen Storage Material”¯: A Review”. International Journal of Energy for a Clean Environment, 11(1-4).
Publisher – Google Scholar
15. NyleÌn, J., Sato, T., Soignard, E., Yarger, J. L., Stoyanov, E., & Häussermann, U. (2009). “Thermal decomposition of ammonia borane at high pressures” The Journal of Chemical Physics, 131(10),
Google Scholar
16. Orimo, S., Nakamori, Y., Eliseo, J. R., Zu, A., & Jensen, C. M. (2007). “Complex Hydrides for Hydrogen Storage”Chemical Review, 107, 4111—4132.
Publisher – Google Scholar
17. Rakap, M., & Ozkar, S. (2010). “Zeolite confined palladium ( 0 ) nanoclusters as effective and reusable catalyst for hydrogen generation from the hydrolysis of ammonia-borane”. International Journal of Hydrogen Energy, 35, 1305—1312.
Publisher – Google Scholar
18. Schlapbach, L., & Züttel, A. (2001). “Hydrogen storage materials for mobile applications” Nature, 414, 353—358.
Publisher – Google Scholar
19. Stowe, A. C., Shaw, W. J., Linehan, J. C., Schmid, B., & Autrey, T. (2007). “In situ solid state 11B MAS-NMR studies of the thermal decomposition of ammonia borane: mechanistic studies of the hydrogen release pathways from a solid state hydrogen storage material” Physical Chemistry Chemical Physics, 9(15), 1831—6.
Publisher – Google Scholar
20. Wolf, G., Baumann, J., Baitalow, F., & Hoffmann, F. P. (2000). “Calorimetric process monitoring of thermal decomposition” Thermochimica Acta, 343, 19—25.
Publisher – Google Scholar
21. Zhou, L. (2005). “Progress and problems in hydrogen storage methods” Renewable and Sustainable Energy Reviews, 9(4), 395—408.
Publisher – Google Scholar







