Introduction
In a range of clinical and forensic situations evaluation of depression and anxiety is important but problematic. For instance, aphasic patients are commonly not able to report their emotional complains. The capacity to verbalize emotional state is limited in patients with cognitive deficits as well. In forensic settings or during professional skills evaluations, simulation or dissimulation of depression and anxiety is also possible. Finally, experienced psychiatrists are usually not available in surgical centers at least in Russia, whereas the diagnostics of depression by non-psychiatric medical professionals is problematic (Mitchell et al., 2009).
Depression and anxiety are important risk factors of postoperative complications and poor outcome of cardiac surgery. Depression was shown to be an independent predictor of postoperative delirium (Leung et al., 2005; Smith et al., 2009), infectious complications (Doering et al., 2008), and overall increased morbidity and mortality at postoperative period (Goyal et al., 2005; Connerney et al., 2010). Increased anxiety in the early postoperative period was demonstrated to be a predictor of postoperative atrial fibrillation (Tully et al., 2011a), whereas baseline anxiety disorder was shown to be significantly associated with increased cardiac morbidity and mortality after coronary artery and valve surgery (Szekely et al., 2007; Tully et al., 2011b). In the study of Williams and colleagues (2013) excessive preoperative anxiety was registered only in 7% of cardiac surgery patients, however, these patients demonstrated 5 times higher mortality and major morbidity after surgery during the index hospitalization (after adjusting for potential confounders). Importantly,depression and anxiety are potentially curable conditions, and treatment was shown to be effective in improving outcomes and mortality in depressive and anxious cardiologic patients (Lie et al., 2007; Milani & Lavie, 2007). Hence, screening for depression and anxiety in cardiac surgery patients is an important clinical and research issue.
In contemporary research of neuropsychological outcomes of cardiac surgery the screening diagnostics of depression and anxiety is most commonly based on the self-report or clinician-rated scales. Both approaches were consistently shown to be reliable screening tools in different clinical settings, however, not ideal tools (Wagle et al., 2000; Zimmerman et al., 2012; Uher et al., in press). In the context of the screening for depression in patients awaiting cardiac surgery, the main disadvantage of questionnaires and psychiatric interview is a need for considering highly negative emotional statements, which increase nervousness related to operation. For instance, the Beck Depression Inventory requires from the patient to consider the following statements: “I feel the future is hopeless, and that things cannot improve”, and “I feel I am being punished” and “I would like to kill myself”. Many patients reported psychological discomfort while completing the Beck Depression Inventory 2—3 days before on-pump operations in our previous study (Golukhova et al., in press). Therefore, we undertook an attempt to develop a non-verbal instrument for screening diagnostics of depression in patients awaiting cardiac surgery.
It was consistently shown that depression is associated with an increased tendency to perceive negative emotional states in others when judging facial expressions (Gur et al., 1992; Hale, 1998; Leppanen et al., 2004). The most typical characteristic of depression is the bias towards false sad responses (Leppanen et al., 2004) or angry responses (Crews & Harrison, 1994) while processing emotionally neutral faces. Anxious individuals also tend to interpret ambiguous facial stimuli in a negative fashion demonstrating the bias towards interpreting the neutral faces as threatening (Yoon, & Zinbarg, 2007). Neuroimaging studies confirmed the abnormal brain activation in patients with mood disorders during both explicit and implicit facial affect recognition (Chen et al., 2006; Price & Drevets, 2010).
Here we present the results of two pilot studies, which evaluated a screening non-verbal tool for diagnostics of depression based on the abnormal perception of facial emotions in depression patients. In both studies we used photographed portraits from the Szondi test (Sobchik, 1995), because the latter one was widely available in Russia.
The original projective psychoanalytical tool was developed by Leopold Szondi in 1947 and included photographed faces of psychiatric patients and criminals. The author instructed examinees to choose the two most appealing and unappealing photos. We changed the procedure and asked patients to select one of two responses concerning mood of a person at the portrait: good versus bad mood. Then we asked subjects to select one of three responses concerning friendliness of a person at the same portrait: friendly, unfriendly or indifferent. We did not use psychoanalytical interpretations of our findings and analyzed number of ‘bad mood’ and ‘unfriendly’ responses. It should be noted, that when the present study was planned the Paul Ekman portraits were not published in Russia and were not available.
In the first study we evaluated the associations between the performance on the facial perception task and depression and anxiety level in patients awaiting on-pump surgery. In addition, we studied the postoperative dynamics of the performance on the facial perception test in our patient population. In the second study we used modified version of the same non-verbal screening tool in the group of neurotic patients in order to determine the reliability of the facial perception approach to screening diagnostics of depression and anxiety in neurotic patients.
Study 1:
Methods
Patients
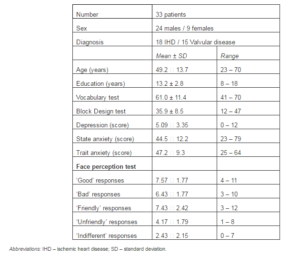
Thirty three adult inpatients were included into the study. Inclusion criteria were: (1) age 18 — 70 years old; (2) left ventricular ejection fraction > 50%. Exclusion criteria were: (1) a history of stroke or other serious neurological disease; (2) sedative drugs intake; (3) emergency or reoperative surgical procedures; (4) deficits of vision or hearing. The study design was explained to subjects, and each subject gave an oral informed consent, as is customary in Russia.
Procedure
The facial perception and other tests were administered to all subjects by the same neuropsychologist (A.P.) in a quiet testing room. The tests were given manually in a standard manner. Psychological scales were given to subjects at the end of the session. Subjects were evaluated 2 — 3 days before on-pump surgery, and nineteen patients were reevaluated in two — three weeks after on-pump operation (10 patients underwent coronary artery bypass grafting, and 9 patients – valvular replacement) in order to determine postoperative changes of the performance on the facial perception task and the dynamics of emotional state.
Facial Perception Task
Fourteen portraits of psychiatric patients and criminals were randomly selected from the Szondi test (Sobchik, 1995) and used for evaluation of facial perception in this study (figure 1). We asked subjects to select one of two responses concerning mood of a person at the portrait: good versus bad mood. Then we asked subjects to select one of three responses concerning friendliness of a person at the same portrait: friendly, unfriendly or indifferent. Commonly, the test session lasted about 10 minutes. Number of selected ‘good mood’ or ‘bad mood’ responses, number of friendly responses, number of unfriendly responses and number of indifferent responses were included into the analysis as separate variables.
Psychological Assessment
The short form (13-item) of the Beck Depression Inventory (BDI) was used for the evaluation of depression as this self-report tool is commonly used in Russia. It should be noted that the recommended cutoff scores for the short form of the BDI are lower in comparison with the 21-item version and are commonly equal to 5, 8, and 16 for determining mild, moderate, and severe depression, respectively (Stukenberg et al., 1990).

Figure 1: Fourteen Photographs from the Szondi Test, which were Used in the Present Study
Russian version of the Spielberger State-Trait Anxiety Inventory (Belova & Shepetova, 2002) was administered in order to evaluate state and trait anxiety. In addition, intelligence was evaluated by Vocabulary and Block Design subtests from the Russian version of Wechsler Adult Intelligence Scale (WAIS) (Filimonenko, & Timofeev, 1995).
Statistical Analysis
Analyses were performed using SPSS software for windows (SPSS 17.0, Chicago, IL, USA). After dividing patients in two groups according to the BDI score, we used t-tests for evaluation of intergroup differences. In addition, we performed correlation analysis using Pearson (2-tailed) test. The perioperative dynamics of emotional performance was tested by Repeated measures ANOVA.
Results
Baseline Patient Characteristics
The baseline patient characteristics are presented in table 1. Middle-aged and elderly males were the majority of this patient population. Overall, the depression level was mild or moderate in the studied patient group. The maximal score on the 13-item Beck Depression Inventory reached 12 points. Twenty four (73%) patients reported depression in a low range (0 — 6 points), and 27% of patients reported mild to moderate depression (7 — 12 points). Trait anxiety was somewhat elevated (46 — 64 points) in the majority of subjects (61%), and situational anxiety was moderately elevated (46 — 62) in the half (51%) of patients and reached maximum of 79 points in one case.
The means and range of negative and positive responses on facial perception task are presented in table 1. Overall, the responses of any kind during the facial perception task were normally distributed (Kolmogorov-Smirnov test ps>0.10), and the range was wide.
Table 1: Baseline Patient Characteristics and Performance on Psychological Tests (Study 1)
Associations between Depression and Performance on the Facial Perception Task
Patients with mild to moderate depression (n=9) differed significantly on ‘bad mood’ and ‘unfriendly’ responses in comparison with the group without depression (n=24). Patients with depression reported 8.22±1.64 ‘bad mood’ responses in comparison with 5.88±1.36 ‘bad mood’ responses of the group without depression (t= -4.17, p<0.001). In addition, patients with depression reported 5.33±1.41 ‘unfriendly’ responses in comparison with 3.83±1.79 ‘unfriendly’ responses in the group without depression (t= -2.26, p=0.031).
The correlation between depression and ‘bad mood’ responses was r=0.45, p=0.008. And the correlation between depression and ‘unfriendly’ responses was 0.46, p=0.007. The correlations between anxiety and facial perception test scores were not significant.
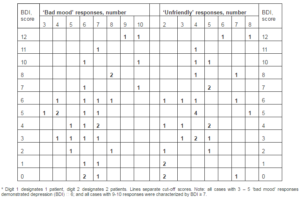
If cut-off score of 7 for ‘bad mood’ responses and 6 for Beck Depression Inventory would be used, than ‘bad mood’ responses criterion would be able to correctly classify 28 of 33 (84.8%) patients (table 2). The cut-off score of 5 ‘unfriendly’ responses allowed to correctly classify 72.7% patients.
Table 2: Baseline Distribution of ‘Bad Mood’ and ‘Unfriendly’ Responses in Cardiac Surgery Patients
Postoperative Changes of the Performance on the Facial Perception Task
Patients after both coronary artery bypass grafting and open heart surgery gave less ‘bad mood’ responses at the postoperative assessment in comparison with the preoperative assessment (F (1,18) = 4.43, p = 0.05) (figure 2). The intergroup differences almost reached significance (F (1, 22) = 3.93, p = 0.061). No significant changes of ‘unfriendly’ responses, depression or anxiety at postoperative follow-up were found in the present patient group.
The number of ‘bad mood’ responses at postoperative assessment tended to correlate with the postoperative depression (r=0.39, p=0.10). Interestingly, the correlation between the ‘bad mood’ responses and state anxiety at postoperative period reached significance (r=0.47, p=0.04).
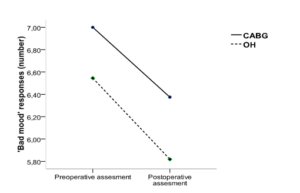
Figure 2: Changes of ‘Bad Mood’ Responses in Patients after Coronary Surgery (CABG) and Open Heart Surgery (OH)
Discussion
The most important finding of the present study was the significant association between the performance on the facial perception task and the self-reported depression. As we expected, patients with elevated depression level tended to give more negative responses on the facial perception task in comparison with patients without depression.
Several previous studies demonstrated significant decrease of depression and anxiety in patients after cardiac surgery at early postoperative period in comparison with the preoperative level (Toner et al., 1998; Kilminster et al., 1999; Krannich et al., 2007). In our previous study, which included another patient group, we found the similar significant decrease of depression at discharge in comparison with the preoperative assessment with the same non-significant trend for state anxiety (Golukhova et al., in press). In the present study we found only the decrease of ‘bad mood’ responses at postoperative period in comparison with the preoperative assessment, whereas verbal reports of depression and anxiety symptoms remained at the preoperative level. It should be noted, that the postoperative asthenia symptoms may be registered as the symptoms of depression when self-report diagnostic approaches are used, and therefore it appeared that the facial perception task was more sensitive to postoperative emotional changes in our patients in comparison with the traditional verbal questionnaires. Interestingly, the correlation between state anxiety and performance on facial perception test reached significance at postoperative period.
The important limitation of this study was the small number of patients with moderate depression. Therefore we undertook the second study, which included psychiatric patients with various levels of depression.
Study 2:
Methods
Patients
Sixty two adult inpatients of the Scientific Practical Psychoneurological Center (previously: Moscow City Hospital N 8 for Neuropsychiatry named after Z.P. Solovyov) were included into the study. The majority of patients of the Solovyov Hospital are neurotic patients or patients needing psychiatric treatment due to stressful life events. Inclusion criteria were: (1) age 18 — 70 years old; (2) education length ≥ 10 years. Exclusion criteria were: (1) psychotic symptoms; (2) a history of stroke or other serious neurological disease; (3) deficits of vision or hearing. The study design was explained to subjects, and each subject gave a written informed consent.
Procedure
The psychiatric diagnostics was conducted by the psychiatrists managing patients during hospitalization according to the clinical interview results. The 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10) is used in the Moscow City Hospital N 8 for Neuropsychiatry.
The facial perception and cognitive tests were administered to all subjects by the same clinical psychologist (E.S.) in a quiet testing room. The tests were given manually in a standard manner and in the same order (see below). Psychological scales were given to subjects at the end of the session.
Facial Perception Task
In the present study we used 38 Szondi portraits (Sobchik, 1995). It should be noted, that 10 more portraits from the original Szondi test were not included because of closed eyes, obvious happy expression or hand jesticulation of the persons on the portraits. The facial perception task lasted in average 15 minutes.
As only two significant associations between performance on the 38-portrait task and psychological variable were found, we present here predominantly the results of the analysis of the responses concerning the 14 portraits (figure 1) which were used in the first study. As in the previous study, we asked subjects to select one of two responses concerning mood of a person at the portrait: good versus bad mood. In contrast to the first study, the second question concerning friendliness of a person at the same portrait allowed only two options: friendly or unfriendly. Number of selected ‘bad mood’ responses and number of ‘unfriendly’ responses were included into the analysis as separate variables.
Psychological Assessment
The short form (13-item) of the Beck Depression Inventory (BDI) was used for the evaluation of depression as this self-report tool is commonly used in Russia. The recommended cutoff scores for the short form of the BDI are lower in comparison with the 21-item version and are commonly equal to 5, 8, and 16 for determining mild, moderate, and severe depression, respectively (Stukenberg et al., 1990).
Russian version of the Spielberger State-Trait Anxiety Inventory (Belova & Shepetova, 2002) was administered in order to evaluate state and trait anxiety. In addition, intelligence was evaluated by Vocabulary and Block Design subtests from the Russian version of Wechsler Adult Intelligence Scale (WAIS) (Filimonenko, & Timofeev, 1995).
Statistical Analysis
Analyses were performed using SPSS software for windows (SPSS 17.0, Chicago, IL, USA). The analysis of demographic intergroup differences was conducted using ANOVA with patient groups as a fixed factor. As the number of ‘bad mood’ and ‘unfriendly’ responses did not distribute normally in the present study (Kolmogorov-Smirnov zs > 1.4;ps<0.05), the intergroup comparisons were conducted using Kruskal-Wallis test and Mann-Whitney test, and correlational analysis was performed using Spearman's (2 tailed) test.
Results
Characteristics of Patient Groups and Intergroup Comparisons
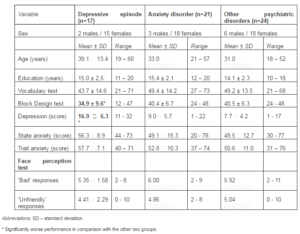
Patient characteristics are presented in table 3. Middle-aged and young females constituted the majority of patients in the present study. We divided patients into three groups according to the main psychiatric diagnosis. The first group included 17 patients with the moderate depressive episode (6 patients with the first episode, 9 patients with recurrent depressive disorder, and 2 patients with bipolar disorder). The second group included 21 patients with anxiety disorder (6 patients with panic attacks and 15 patients with mixed anxiety and depression disorder). The third group included 24 patients with various non-psychotic psychiatric conditions, and the majority of patients of this group were patients with the somatoform disorders (n=13). In addition, this group included patients with borderline or histrionic personality disorder (n=4), schizotypal disorder (n=3), eating disorder (n=3) and organic emotionally labile disorder (n=1).
The groups did not differ by age, education, verbal intelligence (Vocabulary subtest) or sex distribution. As it could be expected, Beck Depression Inventory score was significantly higher in the depressive episode group in comparison with the other two patient groups (F=16.3, p<0.001; after Bonferroni correction ps<0.001). In addition the group with depressive episode showed significantly lower nonverbal intelligence (Block Design subtest) in comparison with other two groups (F=3.44, p=0.039; after Bonferroni correction ps<0.09). Moreover, this group tended to differ on the Spielberger Trait Anxiety score in comparison with the mixed neurotic group (F=2.65, p=0.079; after Bonferroni correction p=0.077).
Patient groups did not show significant differences on the 14-portrait facial perception task (Kruskal-Wallis χ2 0.4). Interestingly, anxious group demonstrated significantly larger number of ‘unfriendly’ responses on the 38-portrait task in comparison with the depressive patients (15.2±5.7 vs 11.3±5.7; Mann-Whitney z=2.16, p=0.031).
Table 3: Baseline Patient Characteristics and Performance on Psychological Tests (Study 2)
Performance on the Facial Perception Task in the Depressive Episode Group
In accordance with the results of the Study 1, we expected that the majority of patients with depressive episode would give 8 and more “bad mood” responses and 6 and more “unfriendly” responses. This was not the case. Only 1 patient with depressive episode gave 8 ‘bad mood’ responses, whereas 7 patients gave few (2 — 5) negative responses. The similar trend was obvious for ‘unfriendly’ responses, and only 4 patients gave 6 and more ‘unfriendly’ responses.
Although we could not confirm our facial perception task to be an effective tool for the diagnostics of depression, we found the significant association between the severity of depression and number of ‘unfriendly’ responses both at 14-portrait and 38-portrait task versions (figure 3). The Spearman’s correlations were 0.58 (p=0.015) for 14 portraits and 0.56 (p=0.016) for 38 portraits.
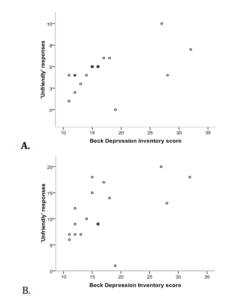
Figure 3: Associations between the Severity of Depression and ‘Unfriendly’ Responses on the 14-Portrait (A) and 38-Portrait (B) Facial Perception Task in Depressive Patients
Performance on the Facial Perception Task in the Anxiety Disorder Group
Only 6 of 21 patients with anxiety disorder gave 8 ‘bad mood’ responses, whereas 7 patients gave few (2 — 5 negative responses). And only 7 of 21 (33%) patients gave 6 and more ‘unfriendly’ responses.
Although we could not confirm our facial performance task to be an effective tool for the diagnostics of anxiety, we found the significant association between the severity of trait anxiety and number of ‘bad mood’ responses on the 14-portrait task version (figure 4). The Spearman’s correlation was 0.51 (p=0.018). The nonsignificant correlation between depression and number of bad mood responses was positive (R=0.25, p=0.27) as well.
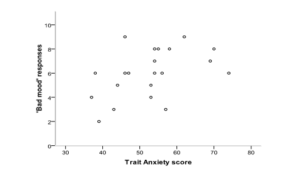
Figure 4: Association between the Trait Anxiety Score and ‘Bad Mood’ Responses on the 14-Portrait Facial Perception Task in Patients with Anxiety Disorder
Performance on the Facial Perception Task in the Patients with Various Neurotic Disorders
Eleven patients of the mixed group reported low level of depression according to the Beck Depression Inventory (1 — 6 points), and 13 patients reported moderate depression (8 — 17 points). The intergroup difference on the ‘bad mood’ responses was near to be significant (3.82±1.54 versus 11.0±2.6; Mann-Whitney z=1.90, p=0.058). The correlation between depression and ‘bad mood’ responses was significant (R=0.41, p=0.044; figure 5). In addition, state anxiety significantly correlated with the ‘bad mood’ responses as well (R=0.42, p=0.044).
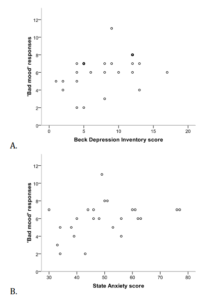
Figure 5: Associations between ‘Bad Mood’ Responses and Depression (A) or State Anxiety (B) Mixed Neurotic Group
In accordance with the results of the Study 1, we applied the cut-off score of 7 for ‘bad mood’ responses and 6 for Beck Depression Inventory. All 11 patients with low depression gave 1 — 7 ‘bad mood’ responses, however only 3 of 14 patients with elevated depression gave 8 or more ‘bad mood’ responses. Group differences did not reach significance (χ2=2.90, p=0.089). When the cut-off score of 5 was set, then 6 of 11 patients gave few (1 — 5) ‘bad mood’ responses and 11 of 13 patients gave 6 and more ‘bad mood’ responses. The intergroup differences reached significance (χ2=4.11, p=0.043). Overall, 71% of patients were correctly classified according to depression level when the cut-off score of 5 ‘bad mood’ responses was used.
It should be noted that no significant correlations between depression/anxiety and performance on the facial perception task were found in the general patient population (n=62) in the present study. When males were analyzed separately, trait anxiety significantly and positively correlated with the number of ‘bad mood’ responses (n=11; R=0.65, p=0.022).
Discussion
The results of the Study 2 did not confirm the Szondi portraits task to be an efficient tool for the screening diagnostics of depression. Nevertheless, in all studied groups we observed significant positive associations between the verbal reports of depressive and anxious symptoms and the performance on the facial perception test. Therefore, the presented nonverbal approach to the screening diagnosis of depression and anxiety appears to be promising.
From neurological point of view, depression and anxiety are characterized by the similar hyperactivity of ‘negative brain’ structures (Davidson et al., 2000; Carretie et al., 2009). These negative emotional conditions highly correlate. For instance, in the Study 1 the correlation coefficients between Beck Depression score and Spielberger State and Trait Anxiety scores were 0.50 and 0.59 (n=33, ps<0.01), respectively. In the Study 2, in the combined patient group (n=62) the same correlations were 0.56 and 0.60 (ps<0.001). Therefore, the performance on the facial perception task significantly correlated with the severity of both negative self-reported psychiatric conditions.
In both studies, ‘bad mood’ responses demonstrated more significant associations with the severity of depression or anxiety in comparison with ‘unfriendly’ responses. Both types of responses were significantly associated with the depression level in the present Study 1. Nevertheless, only ‘bad mood’ responses showed significant postoperative dynamics, which was consistent with the results of a range of previous studies of neuropsychological outcomes of cardiac surgery (Toner et al., 1998; Kilminster et al., 1999; Krannich et al., 2007).
In the Study 2, ‘bad mood’ responses significantly and positively correlated with trait anxiety in the anxiety disorder patients, and with depression and state anxiety in the mixed group of neurotic patients. In addition, ‘bad mood’ responses showed significant positive correlation with trait anxiety in male subgroup. ‘Unfriendly’ responses demonstrated significant association with the anxiety disorder, which could be expected according to the previous study (Yoon, & Zinbarg, 2007). Interestingly, the severity of self-report depression significantly and positively correlated with the number of ‘unfriendly’ responses in patients with a depressive episode.
No negative significant associations between performance on the facial perception task and psychological variables were found in either study. Overall, both types of responses may be useful as a nonverbal measure of psychological distress in future studies. Nevertheless, ‘bad mood’ responses showed higher efficiency in our two studies.
The main limitation of the present study was using Szondi portraits instead of the more scientifically advanced Ekman’s methodology. Indeed, when the present study was designed books of Paul Ekman (2010a, 2010b) were not translated and published in Russia, and therefore this methodology was hardly available at that period. At the same time, Szondi portraits is a popular and well-known tool in clinical psychological practice in Russia. It is obvious now, that the Ekman’s portraits would be a better choice for non-verbal diagnostic screening tools for depression and anxiety in future studies as the pictures from the Szondi test are un-standard and contain much unnecessary information (e.g., hair, cloth, etc.).
The other limitation is the reversed pattern of gender distribution in the Study 1 and Study 2. Male subjects predominantly constituted the patient cohort in the Study 1, and female subjects were the majority in the Study 2.
It should be noted, that females demonstrated not only quantitative, but also qualitative differences in depression and anxiety symptoms in a range of previous studies. It was consistently shown that females were prone to depressive and anxiety disorders twice as frequent as males were in both adolescent and adult populations (McGuinness et al., 2012; Rai et al., 2013; Sonnenberg et al., 2013). Moreover, females more frequently demonstrate somatic and autonomic symptoms of depression, which include appetite and sleep disturbances, headache and fatigue (Alexandrino-Silva et al., 2013; Silverstein et al., 2013), whereas males more commonly show psychomotor retardation or agitation (Alexandrino-Silva et al., 2013). Hence, depressive syndromes are somewhat gender-specific.
In previous studies it was shown that females outperformed males on emotional perception tasks (Weisenbach et al., in press; Kessels et al., in press). Neuroimaging studies consistently showed that brain activation during face perception tasks was more prominent and diffuse in males in comparison with females, and these findings indicated higher efficiency of facial perception networks in females (Ino et al., 2010; Weisenbach et al., in press). Interestingly, Ohrmann and colleagues (2010) observed a reversed pattern of brain activation during facial emotion processing in females and males with panic disorder.
In our patient cohorts, the facial perception task showed somewhat higher efficiency as a tool for the diagnostics of depression and anxiety. In male subjects these associations were significant in both studies. However, in females the associations between facial perception and emotional status were less consistent and depended on the psychiatric condition. Hence, further studies should consider the associations between depressive symptoms and facial emotional perception bias separately in males and females.
We used a 13-item version of Beck Depression Inventory instead of the original 21-item version, and this could be considered as another limitation of our study. We have chosen the short version of Beck Inventory in order to minimize discomfort of the emotional evaluation during preoperative period in cardiac surgery patients. At least two previous studies showed that the reliability and validity of the short version of Beck Inventory as a diagnostic tool for depression is good (Scogin et al., 1988; Furlanetto et al., 2005).
Conclusion
Our data evidence is that facial perception tasks are a perspective approach for non-verbal screening diagnostics of depression and anxiety. Future studies are needed in order to investigate effects of gender, age and cognitive status as potential contributors to the performance on facial perception screening tools.
References
Alexandrino-Silva, C., Wang, Y. P., Carmen Viana, M., Bulhões, R. S., Martins, S. S. & Andrade, L. H. (2013). “Gender Differences in Symptomatic Profiles of Depression: Results from the São Paulo Megacity Mental Health Survey,”Journal of Affective Disorders, 147 (1-3) 355-64.
Publisher – Google Scholar
Belova, A. N. & Shepetova, O. N. (2002). ‘Scales, Tests and Questionnaires in Medical Rehabilitation,’ Antidor, Moscow, Russia.
Google Scholar
Chen, C. H., Lennox, B., Jacob, R., Calder, A., Lupson, V., Bisbrown-Chippendale, R., Suckling, J. & Bullmore, E. D. (2006). “Explicit and Implicit Facial Affect Recognition in Manic and Depressed States of Bipolar Disorder: A Functional Magnetic Resonance Imaging Study,” Biological Psychiatry, 59 (1) 31-39.
Publisher – Google Scholar
Connerney, I., Sloan, R. P., Shapiro, P. A., Bagiella, E. & Seckman, C. (2010). “Depression Is Associated with Increased Mortality 10 Years after Coronary Artery Bypass Surgery,” Psychosomatic Medicine, 72 (9) 874-81.
Publisher – Google Scholar
Crews, W. D., Jr & Harrison, D. W. (1994). “Sex Differences and Cerebral Asymmetry in Facial Affect Perception as a Function of Depression,” Psychobiology, 22: 112-116.
Publisher
Davidson, R. J., Jackson, D. C. & Kalin, N. H. (2000). “Emotion, Plasticity, Context, and Regulation: AmiltonVes from Affective Neuroscience,” Psychological Bulletin, 126 (6) 890-909.
Publisher – Google Scholar
Doering, L. V., Martinez-Maza, O., Vredevoe, D. L. & Cowan, M. J. (2008). “Relation of Depression, Natural Killer Cell Function, and Infections after Coronary Artery Bypass in Women,” European Journal of Cardiovascular Nursery, 7 (1) 52-8.
Publisher – Google Scholar
Ekman, P. (2010a). ‘Psychologia Emotziy. Izdatelstvo “Piter”,’ Sankt-Petersburg, Russia.
Ekman, P. & Friesen, W. V. (2010b). ‘Uznay Lzhetza Po Vyrazheniu Litza. Izdatelstvo “Piter”,’ Sankt-Petersburg, Russia.
Filimonenko, Y. & Timofeev, V. (1995). ‘Manual for the Russian Version of the Wechsler Adult Intelligence Scale,’Imaton, Sankt-Petersburg, Russia.
Google Scholar
Furlanetto, L. M., Mendlowicz, M. V. & Romildo Bueno, J. (2005). “The Validity of the Beck Depression Inventory-Short form as a Screening and Diagnostic Instrument for Moderate and Severe Depression in Medical Inpatients,” Journal of Affective Disorders, 86 (1) 87-91.
Publisher – Google Scholar
Golukhova, E. Z., Polunina, A. G. & Guekht, A. B. (In Press). ‘Psychoemotional Status in Patients after On-Pump Surgery at Early and Delayed Postoperative Period,’ Kreativnaya Kardiologia.
Goyal, T. M., Idler, E. L., Krause, T. J. & Contrada, R. J. (2005). “Quality of Life Following Cardiac Surgery: Impact of the Severity and Course of Depressive Symptoms,” Psychosomatic Medicine, 67 (5) 759-65.
Publisher – Google Scholar
Gur, R. C., Erwin, R. J., Gur, R. E., Zwil, A. S., Heimberg, C. & Kraemer, H. C. (1992). “Facial Emotion Discrimination: II. Behavioral Findings in Depression,” Psychiatry Research, 42 (3) 241-51.
Publisher – Google Scholar
Hale, W. W. 3rd. (1998). “Judgment of Facial Expressions and Depression Persistence,” Psychiatry Research, 80 (3) 265-74.
Publisher – Google Scholar
Ino, T., Nakai, R., Azuma, T., Kimura, T. & Fukuyama, H. (2010). “Gender Differences in Brain Activation during Encoding and Recognition of Male and Females Faces,” Brain Imaging and Behavior, 4 (1) 55-67.
Publisher – Google Scholar
Kessels, R. P. S., Montagne, B., Hendriks, A. W., Perrett, D. I. & De Haan, E. H. (In Press) (2013). “Assessment of Perception of Morphed Facial Expressions Using the Emotion Recognition Task: Normative Data from Healthy Participants Aged 8-75,” Journal of Neuropsychology.
Publisher – Google Scholar
Kilminster, S., Mcmillan, T., Mcmillan, T. & Holt, D.W. (1999). “Neuropsychological Change and S100 Protein Release in 130 Unselected Patients Undergoing Caradiac Surgery,” Stroke, 30 (9) 1969-1974.
Publisher – Google Scholar
Krannich, J. H. A., Weyers, P., Lueger, S., Herzog, M., Bohrer, T. & Elert, O. (2007). “Presence of Depression and Anxiety before and after Coronary Artery Bypass Graft Surgery and Their Relationship to Age,” BMC Psychiatry, 7: 47.
Publisher – Google Scholar
Leppanen, J. M., Milders, M., Bell, J. S., Terriere, E. & Hietanen, J. K. (2004). “Depression Biases the Recognition of Emotionally Neutral Faces,” Psychiatry Research, 128 (2) 123-33.
Publisher – Google Scholar
Leung, J. M., Sands, L. P., Mullen, E. A., Wang, Y. & Vaurio, L. (2005). “Are Preoperative Depressive Symptoms Associated with Postoperative Delirium in Geriatric Surgical Patients?,” Journal of Gerontology: Medical Sciences, 60A (12) 1563-1568.
Publisher – Google Scholar
Lie, I., Arnesen, H., Sandvik, L., Hamilton, G. & Bunch, E. H. (2007). “Effects of a Home-Based Intervention Program on Anxiety and Depression 6 Months after Coronary Artery Bypass Grafting: A Randomized Controlled Trial,” Journal of Psychosomatic Research, 62 (4) 411-8.
Publisher – Google Scholar
Mcguinness, T. M., Dyer, J. G. & Wade, E. H. (2012). “Gender Differences in Adolescent Depression,” Journal of Psychosocial Nursing and Mental Health Services, 50 (12) 17-20.
Publisher – Google Scholar
Milani, R. V. & Lavie, C. J. (2007). “Impact of Cardiac Rehabilitation on Depression and Its Associated Mortality,”American Journal of Medicine 120 (9) 799-806.
Publisher – Google Scholar
Mitchell, A. J., Vaze, A. & Rao, S. (2009). “Clinical Diagnosis of Depression in Primary Care: A Meta-Analysis,” Lancet,374 (9690) 609-19.
Publisher – Google Scholar
Ohrmann, P., Pedersen, A., Braun, M., Bauer, J., Kugel, H., Kersting, A., Domschke, K., Deckert, J. & Suslow, T. (2010). “Effect of Gender on Processing Threat-Related Stimuli in Patients with Panic Disorder: Sex Does Matter,”Depression and Anxiety, 27 (1) 1034-43.
Publisher – Google Scholar
Price, J. L. & Drevets, W. C. (2010). “Neurocircuitry of Mood Disorders,” Neuropsychopharmacology Reviews, 35 (1) 192-216.
Publisher – Google Scholar
Rai, D., Zitko, P., Jones, K., Lynch, J. & Araya, R. (2013). “Country- and Individual-Level Socioeconomic Determinants of Depression: Multilevel Cross-National Comparison,” British Journal of Psychiatry, 202 (3) 195-203.
Publisher – Google Scholar
Scogin, F., Beutler, L., Corbishley, A. & Hamblin, D. (1988). “Reliability and Validity of the Short Form Beck Depression Inventory with Older Adults,” Journal of Clinical Psychology, 44 (6) 853-7.
Publisher – Google Scholar
Silverstein, B., Edwards, T., Gamma, A., Ajdacic-Gross, V., Rossler, W. & Angst, J. (2013). “The Role Played by Depression Associated with Somatic Symptomatology in Accounting for the Gender Difference in the Prevalence of Depression,” Social Psychiatry and Psychiatric Epidemiology, 48 (2) 257-63.
Publisher – Google Scholar
Smith, P. J., Attix, D. K., Weldon, B. C., Greene, N. H. & Monk, T. G. (2009). “Executive Function and Depression as Independent Risk Factors for Postoperative Delirium,” Anesthesiology, 110 (4) 781-7.
Publisher – Google Scholar
Sobchik, L. N. (1995). ‘Szondi Test: Russian Version,’ Izdatelstvo Folium, Moscow, Russia.
Sonnenberg, C. M., Deeg, D. J., Van Tilburg, T. G., Vink, D., Stek, M. L. & Beekman, A. T. (2013). “Gender Differences in the Relation between Depression and Social Support in Later Life,” International Psychogeriatry, 25 (1) 61-70.
Publisher – Google Scholar
Stukenberg, K. W., Dura, J. R. & Kiecolt-Glaser, J. K. (1990). “Depression Screening Scale Validation in an Elderly, Community-Dwelling Population,” Journal of Consulting and Clinical Psychology, 2 (2) 134-138.
Publisher – Google Scholar
Szekely, A., Balog, P., Benkц, E., Breuer, T., Szekely, J., Kertai, M. D., Horkay, F., Kopp, M. S. & Thayer, J. F. (2007). “Anxiety Predicts Mortality and Morbidity after Coronary Artery and Valve Surgery—A 4-Year Follow-Up Study,”Psychosomatic Medicine, 69 (7) 625-31.
Publisher – Google Scholar
Toner, I., Taylor, K. M., Newman, S. & Smith, P. L. C. (1998). “Cerebral Functional Changes Following Cardiac Surgery: Neuropsychological and EEG Assessment,” European Journal of Cardio-Thoracic Surgery, 13 (1) 13-20.
Publisher – Google Scholar
Tully, P. J., Bennetts, J. S., Baker, R. A., Mcgavigan, A. D., Turnbull, D. A. & Winefield, H. R. (2011a). “Anxiety, Depression, and Stress as Risk Factors for Atrial Fibrillation after Cardiac Surgery,” Heart and Lung, 40 (1) 4-11.
Publisher – Google Scholar
Tully, P. J., Pedersen, S. S., Winefield, H. R., Baker, R. A., Turnbull, D. A. & Denollet, J. (2011b). “Cardiac Morbidity Risk and Depression and Anxiety: A Disorder, Symptom and Trait Analysis among Cardiac Surgery Patients,”Psychology, Health and Medicine, 16 (3) 333-45.
Publisher – Google Scholar
Uher, R., Perlis, R. H., Placentino, A., Dernovsek, M. Z., Henigsberg, N., Mors, O., Maier, W., Mcguffin, P. & Farmer, A. (2012). “Self-Report and Clinician-Rated Measures of Depression Severity: Can One Replace the Other,” Depression and Anxiety, 29 (12) 1043-9.
Publisher – Google Scholar
Wagle, A. C., Ho, L. W., Wagle, S. A. & Berrios, G. E. (2000). “Psychometric Behavior of BDI in Alzheimer’s Disease Patients with Depression,” International Journal of Geriatric Psychiatry, 15 (1) 63-69.
Publisher – Google Scholar
Weisenbach, S. L., Rapport, L. J., Briceno, E. M., Haase, B. D., Vederman, A. C., Bieliauskas, L. A., Welsh, R. C., Starkman, M. N., Mcinnis, M. G., Zubieta, J. K. & Langenecker, S. A. (In Press) (2012). “Reduced Emotion Processing Efficiency in Healthy Males Relative to Females,” Social Cognitive and Affective Neuroscience.
Publisher – Google Scholar
Williams, J. B., Alexander, K. P., Morin, J. F., Langlois, Y., Noiseux, N., Perrault, L. P., Smolderen, K., Arnold, S. V., Eisenberg, M. J., Pilote, L., Monette, J., Bergman, H., Smith, P. K. & Afilalo, J. (2013). “Preoperative Anxiety as a Predictor of Mortality and Major Morbidity in Patients Aged >70 Years Undergoing Cardiac Surgery,” American Journal of Cardiology, 111 (1) 137-42.
Publisher – Google Scholar
Yoon, K. L. & Zinbarg, R. E. (2007). “Threat Is in the Eye of the Beholder: Social Anxiety and the Interpretation of Ambiguous Facial Expressions,” Behavior Research and Therapy, 45 (4) 839-847.
Publisher – Google Scholar
Zimmerman, M., Martinez, J., Attiullah, N., Friedman, M., Toba, C. & Boerescu, D. A. (2012). “Why Do Some Depressed Outpatients Who are Not in Remission According to the Depression Rating Scale Nonetheless Consider Themselves to be in Remission,” Depression and Anxiety 29 (10) 891-5.
Publisher – Google Scholar










