Introduction
Prostate carcinoma (PCa) is one of the most common cancers in males. Clinical screening of PCa, mainly based on serum prostate-specific antigen (PSA), has increased the detection of early-stage PCa which is curable with a radical retropubic prostatectomy (RRP) [1]. After a successful RRP, the PSA usually becomes undetectable (
An increase in PSA level, however, cannot provide precise information regarding the location of relapse, which can be local (in the prostatic bed) or systemic (skeletal or in other organs). These two conditions involve different therapies: local salvage therapy (surgery/radiotherapy) in the first case and systemic therapy (hormonotherapy) in the second case [3,4].
The potential role of dynamic contrast-enhanced (DCE) MRI in the detection of PCa recurrences after RRP has already been demonstrated in some preliminary studies [5]. Recently, some authors have described the high sensitivity of DWI in detecting recurrent PCa after brachytherapy [6] and RRP [7].
In this paper, we present a case of PCa recurrence in the bladder wall after RRP: we report the main features at endorectal-MRI, including DWI and DCE-MRI, and discuss the role of this multi-parametric imaging modality in evaluating patients with a suspected local recurrence of PCa.
Case Report
A 78-year-old man was referred to our institution for biochemical relapse after RRP for a PCa. The PSA level at the moment of the RRP was 10.3 ng/mL; pathological examination showed a PCa in both prostatic lobes, with a Gleason score (GS) of 3+3, without extra-prostatic extension (pT2c,N0,Mx). Biochemical recurrence occurred 24 months after RRP, with a slow increase of the PSA level, from 0.97 ng/mL to 1.7 ng/mL over five years. Based on these clinical data, local recurrence was suspected and endorectal-MRI was performed for investigating the prostatic fossa. This exam was carried out using a 1.5T whole-body scanner with a pelvic phased-array surface coil (Signa HDxt; General Electric Company, Milwaukee, WI, USA) combined with a disposable endorectal coil (MedRad, Indianola, Pa), after a cleansing rectal enema. According to our routine protocol, a morphological study was obtained using Fast Relaxation Fast Spin-Echo (FRFSE) T2-weighted sequences in the sagittal, transverse and coronal planes, and a Fast Spin-Echo (FSE) T1-weighted sequence in the transverse plane, all including the bladder and the symphysis, with a slice thickness of 4 mm.
The DWI acquisition was performed in the axial plane, using a single-shot echo-planar imaging (SSEPI) sequence, with two b-values (0 and 600 s/mm2) and a slice thickness of 4 mm.
The DCE-MRI was obtained using a three-dimensional Spoiled Gradient Recalled (SPGR) T1-weighted sequence during the intravenous injection of a contrast bolus of 0.2 ml/kg of body weight of Gadoterate meglumine (Dotarem, Guerbet S.A., Roissy, France), at a flow rate of 3-4 ml/sec followed by 15 ml of saline solution. Twenty three-dimensional data sets, two before and eighteen after contrast administration, were acquired with a 19-second temporal resolution and a total duration of 4-5 minutes. The DWI and DCE-MRI images were processed on an independent workstation with dedicated software (Functool, 4.5.5, General Electric Company). Regions of interest (ROIs) positioned on the suspected areas were used to calculate the corresponding value of the apparent diffusion coefficient (ADC) for DWI, and Time-Intensity (T-I) curves for DCE-MRI.
The accurate analysis of the images showed a small nodule, approximately one centimeter in diameter, within the posterior bladder wall, delineated by the mucosa. This nodule appeared hyperintense on T2- weighted images (Fig. 1A and B) and on b=600 s/mm2 images of the DWI (Fig. 1C). The DCE-MRI showed a hypervascular nodule with a T-I curve characterized by a prominent rapid wash-in and a rapid wash-out (Fig. 2A, B).

Figure 1: The T2-weighted sequence in the transverse (A) and sagittal (B) planes shows a hyperintense nodule in the posterior wall of the bladder (white arrow). The sagittal view clearly demonstrates that the nodule is distant from the prostatectomy bed (arrowhead in B). (C) The b=600 s/mm2 image of the DWI sequence shows a distinctly hyperintense nodule (white arrow); the corresponding ADC value, calculated on the ADC map (not shown), isreduced (1.48 x 10-9 m2/s)
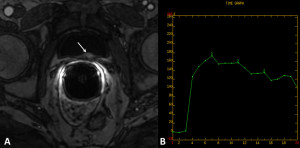
Figure 2: (A) At DCE-MRI, the nodule has intense contrast enhancement after contrast medium administration (white arrow). (B) The corresponding T-I curve is characterized by a prominent rapid wash-in and a rapid wash-out
Based on these imaging findings, PCa recurrence in the bladder wall was suspected. The cystoscopy confirmed the presence of a submucosal nodule in the posterior bladder wall and a radical resection was carried out. At histology, a diagnosis of fibromuscolar tissue infiltrated by PCa (GS 3+3) was made.
Discussion
Although this is a single case report, we believe that this study points out the ability of multi-parametric MRI to evaluate a local recurrence of PCa after RRP, which affected 20-50% of patients during a 10-year follow-up [1]. In this clinical setting, PSA remains the main tumor marker; the current consensus considers a PSA level >0.2 ng/mL indicative of residual/recurrent disease [1]. However, PSA alone is not able to differentiate local from distant recurrence, even if specific features of its increase can reliably accomplish this differentiation; a late biochemical recurrence (>24 months after local treatment), low PSA velocity (change of PSA over time) and a long PSA doubling time (>6 months) more likely indicate local recurrence; conversely, a rapid biochemical recurrence, high PSA velocity and a short PSA doubling time, more likely indicate distant recurrence [1,2].
In our case, the patient’s clinical presentation strongly suggested the presence of local recurrence. In this context, digital rectal examination (DRE) and TRUS frequently demonstrate negative results, even if color/power-Doppler or contrast agents are used [8]. Computed tomography is useful in detecting systemic metastases, but its value in the evaluation of the prostatic fossa is very limited [9].
Endorectal-MRI has been used to detect local recurrence following RRP. While most local recurrences are perianastomotic or retrovesical, 30% may be found elsewhere in the pelvis, at sites which can be more readily assessed by MRI than by TRUS [1]. Previous studies on the use of T2-weighted MRI alone, reported variable values of sensitivity and specificity (48-95% and 52-100%, respectively) [1]; endorectal-MRI also allows clear evaluation of retained seminal vesicle remnants which are present in up to 20% of patients and can be the site of cancer recurrence.
Recent studies have suggested that newer MR techniques, including spectroscopy and DCE-MRI, can improve the detection of local recurrence [5]. In particular, DCE-MRI seems to be a very promising technique in detecting recurrent cancer foci. The authors of these studies found that DCE-MRI, either alone or in combination with T2-weighted MRI, significantly improved sensitivity and specificity to 84.1-88% and 89.3-100% respectively. In addition, Boonsirikamchai et al. [10] attempted to characterize cancer tissue by using T-I curves obtained starting from postprocessed DCE-MRI images.
DW-MRI has recently been introduced as an innovative diagnostic tool in the study of microstructural and functional changes in several pathologies of various organs without contrast medium [6]. In the field of urologic oncology, DW-MRI has various applications, mainly related to the characterization of focal renal masses and the detection of prostate and bladder cancer [7].
Nowadays, the Choline PET/CT may visualize the site of recurrence by revealing both local and systemic recurrence in a single step. In most of the studies, the detection rate is about 30-40% for disease relapse after radical treatment, even in the presence of low (
In our case, DW-MRI and DCE-MRI permitted correct evaluation of the nodular tissue in the bladder wall as a PCa recurrence allowing a radical surgical trans-urethral treatment. Moreover, our experience demonstrated that the combination of a T2-weighted sequence with DCE-MRI and DWI is able to detect a local recurrence of PCa, even in the presence of low levels of PSA (
In conclusion, the endorectal MRI with multiparametric approach (essentially with DCE) may have a role in the early phase of PSA relapse.
References
1. Katz S, Rosen M (2006) “MR imaging and MR spetroscopy in prostate cancer management,” Radiol Clin North A, 44(5):723-734.
GoogleScholar
2. Schiavina R, Borghesi M, Fiorentino M, Brunocilla E, Manferrari F, Vagnoni V, et al, (2013) “Identification of prostate cancer risk categories according to surgical margins status, pathological stage and Gleason score,” Int J Urol 20(11):1097-1103.
Publisher – GoogleScholar
3. Leonardo C, Simone G, Papalia R, Franco G, Guaglianone S, Gallucci M, (2009) “Salvage radical prostatectomy for recurrent prostate cancer after radiation therapy,” Int J Urol 16(6):584-586.
Publisher – GoogleScholar
4. Chalasani V, Iansavichene AE, Lock M, Izawa JI, (2009) “Salvage radiotherapy following radical prostatectomy,” Int J Urol 16(1):31-36.
Publisher – Google Scholar
5. Sciarra A, Panebianco V, Salciccia S, Osimani M, Lisi D, Ciccariello M, et al, (2008) “Role of Dynamic Contrast-Enhanced Magnetic Resonance (MR) Imaging and Proton MR Spectroscopic Imaging in the detection of local recurrence after radical prostatectomy for prostate cancer,” Eur Urol 54:589-600.
Publisher – Google Scholar
6. Tamada T, Sone T, Jo Y, Hiratsuka J, Higaki A, Higashi H, et al, (2011) “Locally recurrent Prostate Cancer After High-Dose-Rate Brachytherapy: The value of Diffusion-Weighted Imaging, Dynamic Contrast-Enhanced MRI, and T2-Weighted Imaging in localizing tumors,” AJR Am J Roentgenol 197:408-414.
Publisher – Google Scholar
7. Giannarini G, Nguyen DP, Thalmann GN, Thoeny HC, (2012) “Diffusion-weighted magnetic resonance imaging detects local recurrence after radical prostatectomy: initial experience,” Eur Urol 61(3):616-620.
Publisher – Google Scholar
8. Drudi FM, Giovagnorio F, Carbone A, Ricci P, Petta S, Cantisani V, et al, (2006) “Transrectal color doppler contrast sonography in the diagnosis of local recurrence after radical prostatectomy – comparison with MRI,” Ultraschall in Med 27:146-151.
Publisher – GoogleScholar
9. Older RA, Lippert MC, Gay SB, Omary RA, Hillman BJ, (1995) “Computed tomography appearance of the prostatic fossa following radical prostatectomy,” Acad Radiol 2(6):470-474.
Publisher – GoogleScholar
10. Boonsirikamchai P, Kaur H, Kuban DA, Jackson E, Hou P, Choi H, (2012) “Use of maximun slope images generated from dynamic contrast-enhanced MRI to detect locally recurrent prostate carcinoma after prostatectomy: a practical approach,” AJR Am J Roentgenol 198(3):W228-236.
Publisher – Google Scholar
11. Schiavina R, Ceci F, Borghesi M, Brunocilla E, Vagnoni V, Gacci M, Castellucci P, Nanni C, Martorana G, Fanti S, (2013) “The dilemma of localizing disease relapse after radical treatment for prostate cancer: which is the value of the actual imaging techniques?,” Curr Radiopharm 6;6(2):92-95.
Google Scholar




