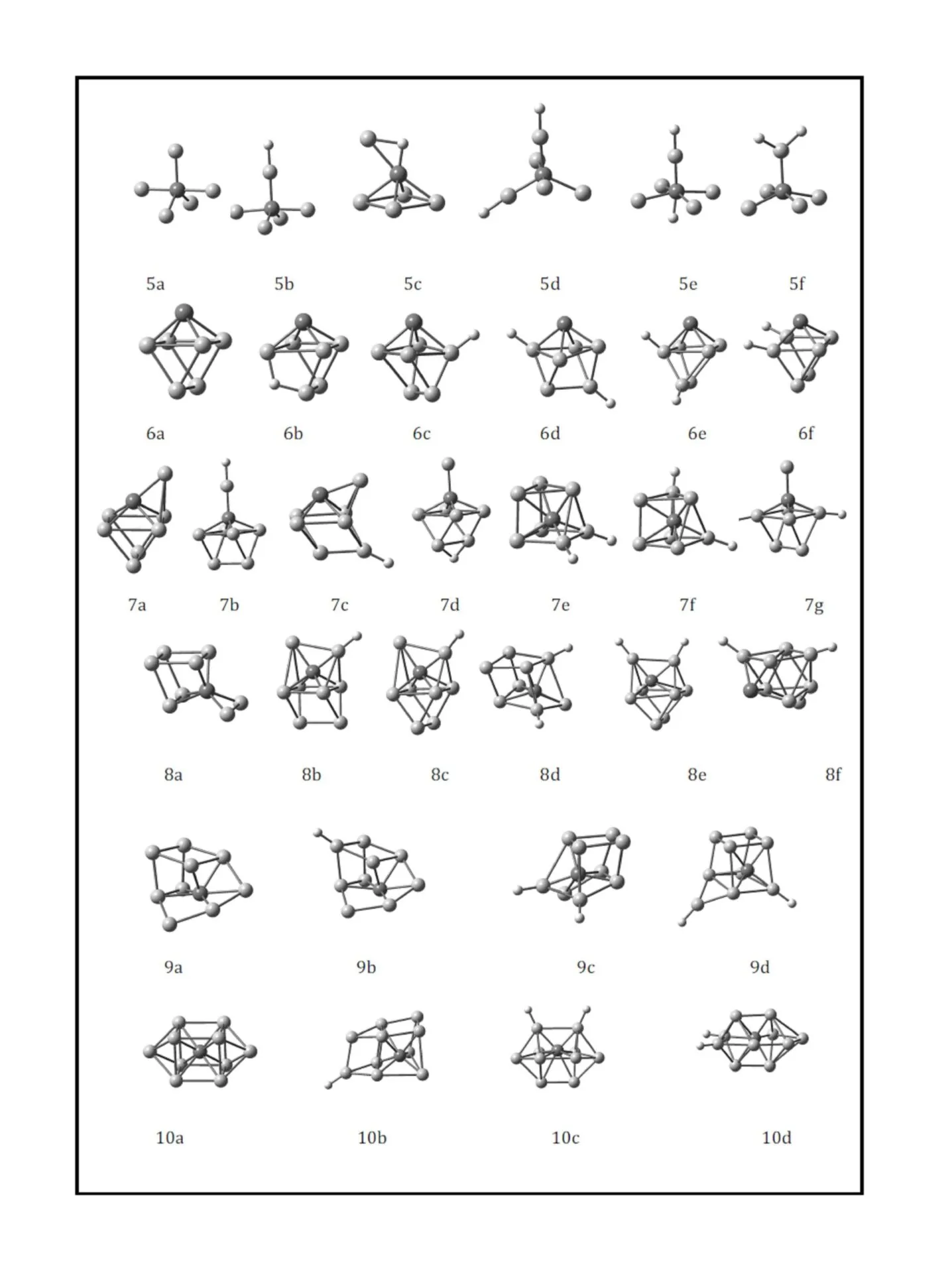
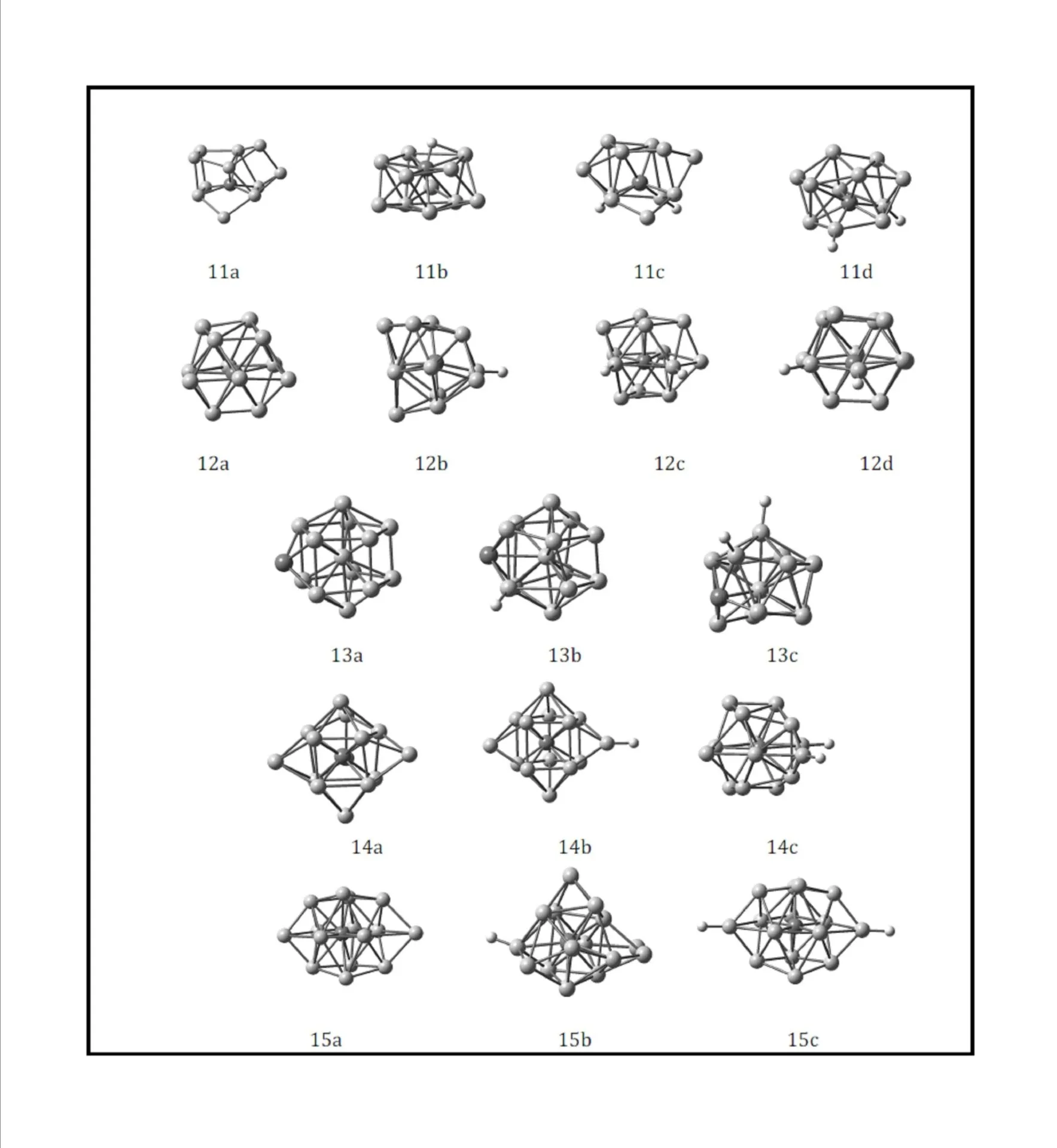
This work was financially supported by the National Natural Science Foundation of China (Grant No. 20603021), the Natural Science Foundation of Shanxi (Grant No. 2013011009-6), the Youth Academic Leader of Shanxi, Undergraduate Training Programs for Innovation and Entrepreneurship of Shanxi Province (Grant No. 2013145) and Shanxi Normal University (SD2013CXCY-65,105088) and Teaching Reform Project of Shanxi Normal University (SD2013JGXM-51).
References
1. Andersonsmall, H. H. (1997) Particles and Inorganic Clusters, New York press.
2. Behm, J. M., Brugh, D. J. and Morse, M. D. (1994) “Spectroscopic analysis of the open 3d subshell transition metal aluminides: AlV, AlCr, and AlCo,” Journal of Chemical Physics, 101 (8) 6487-6499.
Publisher – Google Scholar
3. Bond, G. C. (1987) Heterogeneous catalysis : principles and applications, Clarendon Press, Oxford, New York.
4. Chen, F. Y. and Johnston, R. L. (2008) “Energetic, Electronic, and Thermal Effects on Structural Properties of Ag-Au Nanoalloys,” ACS NANO, 2 (1) 165-171.
5. Guo, L. (2007) “Evolution of the electronic structure and properties of neutral and charged cobalt-doped aluminum clusters,” International Journal of Mass Spectrometry, 268 (1) 8-15.
Publisher – Google Scholar
6. Guo, L. (2008) “The cobalt-doped aluminum AlnCo (n = 8—16) and their anions: Structure, thermochemistry, and electron affinities,” Journal of Alloys and Compounds, 466 (1-2) 463-470.
Publisher – Google Scholar
7. Khanna, S. N. and Jena, P. (1994) “Designing ionic solids from metallic clusters,” Chemical Physics Letters, 219 (5) 479-483.
Publisher – Google Scholar
8. Knight, W. D., Clemenger, K, de Heer, W. A., Saunders, W. A., Chou, M. Y. and Cohen, M. L. (1984) “Electronic Shell Structure and Abundances of Sodium Clusters,” Physical Review Letters, 52 (24) 2141-2143.
Publisher – Google Scholar
9. Koel, B. E. and Somorjai, G. A. (1985) Catalysis, Berlin press.
10. Laguna, A, Lasanta, T, Lopez-de-Luzuriaga, J. M., Monge, M, Naumov, P. and Olmos, M. E. (2010) “Combining Aurophilic Interactions and Halogen Bonding To Control the Luminescence from Bimetallic Gold-Silver Clusters,” Journal of the American Chemical Society, 132 (2) 456-457.
11. Lahr, D. L. and Ceyer, S. T. (2006) “Catalyzed CO Oxidation at 70 K on an Extended Au/Ni Surface Alloy,” Journal of the American Chemical Society, 128 (6) 1800-1801.
Publisher – Google Scholar
12. Lee, H. M., Ge, M, Sahu, B. R., Tarakeshwar, P. and Kim, K. S. (2003) “Geometrical and Electronic Structures of Gold, Silver, and Gold-Silver Binary Clusters: Origins of Ductility of Gold and Gold-Silver Alloy Formation,” Journal of Physical Chemistry B, 107 (37) 9994-10005.
Publisher – Google Scholar
13. Lu, C, Kuang, X. Y., Lu, Z. W., Mao, A. J. and Ma, Y. M. (2011) “Determination of Structures, Stabilities, and Electronic Properties for Bimetallic Cesium-Doped Gold Clusters: A Density Functional Theory Study,” Journal of Physical Chemistry A ,115, 9273-9281.
Publisher – Google Scholar
14. Menezes, W. J. C. and Knickelbein, M. B. (1991) “Bimetallic clusters of cobalt and aluminum: ionization potentials versus reactivity, and the importance of geometric structure,” Chemical Physics Letters, 183 (5) 357-362.
Publisher – Google Scholar
15. Menezes, W. J. C., Knickelbein, M. B. (1993) “The evolution of electronic structure in AlnCom,” Zeitschrift für Physik D Atoms, Molecules and Clusters, 26 (1) 322-324.
Publisher – Google Scholar
16. Nonose, S, Sone, Y, Onodera, K, Sudo, S. and Kaya, K. (1989) “Reactivity study of alloy clusters made of aluminum and some transition metals with hydrogen,” Chemical Physics Letters, 164 (4) 427-432
Publisher – Google Scholar
17. Ouyang, Y. F., Wang, J. C., Hou, Y. H., Zhong, X. P., Du, Y. and Feng, Y. P. (2008) “First principle study of AlX , (X=3d,4d,5d elements and Lu) dimmer,” Journal of Chemical Physics, 128 (7) 074305-1-6.
Publisher – Google Scholar
18. Passacantando, M, Ottaviano, L, D’Orazio, F, Lucari, F, Biase, M. D., Impellizzeri, G. and Pramann, A, Nakajima, A. and Kaya, K. (2001) “Photoelectron spectroscopy of bimetallic aluminum cobalt cluster anions: Comparison of electronic structure and hydrogen chemisorption rates,” Journal of Chemical Physics, 115 (12) 5404-5410.
Publisher – Google Scholar
19. Priolo, F. (2006) “Growth of ferromagnetic nanoparticles in a diluted magnetic semiconductor obtained by Mn+ implantation on Ge single crystals,” Physical Review B, 73 (19) 195207 (5).
Publisher – Google Scholar
20. Singh, A. K., Kumar, V. and Kawazoe, Y. J. (2005) “Thorium Encapsulated Caged Clusters of Germanium: Th@Gen, n =16, 18, and 20,” Journal of Physical Chemistry B, 109 (32) 15187-15189
Publisher – Google Scholar
21. Tian, F. Y., Jing, Q. and Wang, Y. X. (2008) “Structure, stability, and magnetism of ScnAl (n=1-8,12) clusters: Density-functional theory investigations,” Physical Review A, 77 (1) 013202 (8).
Google Scholar
22. Zanti, G. and Peeters, D. (2010) “DFT Study of Bimetallic Palladium-Gold Clusters PdnAum of Low Nuclearities (n + m ≤14),” Journal of Physical Chemistry A, 114, 10345-10356.
Publisher – Google Scholar
23. Zhao, Y. R., Kuang, X. Y., Zheng, B. B., Li, Y. F. and Wang, S. J. (2011) “Equilibrium Geometries, Stabilities, and Electronic Properties of the Bimetallic M2-doped Aun (M = Ag, Cu; n = 1-10) Clusters: Comparison with Pure Gold Clusters,” Journal of Physical Chemistry A, 115, 569-576.