Introduction
Aflatoxins are generally produced in animal feeds by toxigenic fungi such as Aspergillus flavus, Aspergillus parasiticus and the rare Aspergillus nomius (Kamkar, et al 2011). They are both acutely and chronically toxic, mutagenic, teratogenic and carcinogenic compounds for animal and human (Deshpande, 2002; Ghazani, 2009; Maktabi et al., 2011 and Mohamadi Sani et al., 2012). Aflatoxin M1 (AFM1) is the principle hydroxylated metabolite of aflatoxin B1 which is transformed at the hepatic level by means of cytochrome p450 enzymes and excreted into the milk in the mammary glands of both human and lactating animal after ingestion by the animal of pellets and forage contaminated with aflatoxin B1 (Oveisi et al., 2007; Prandini et al., 2009; Hampikyan et al., 2010; Ayoub et al., 2011). It has been reported that there is a linear relationship between AFM1 in milk and AFB1 in the feed consumed by the animals with approximately 1% to 6% of the ingested AFB1 appearing as AFM1 in milk (Dragacci et al., 1995; Battacone et al., 2005; Fallah, 2010). Milk is a major food commodity for introducing aflatoxin into human diet and evidence of hazardous human exposure to AFM1 through dairy products has been shown (Zinedine & Manes, 2009). Aflatoxin M1 is resistant to thermal inactivation and not destroyed completely by pasteurization, autoclaving and other food processing procedures (Youssef & Marth, 1989; and Maktabi, & Fazlara, 2011).
Since the consumption of milk and milk products by human populations is quite high there is a risk of exposure to AFM1 with infants and young children being at increased risk. Levels of AFM1 in the diet are therefore important and AFM1 in milk and dairy products should be controlled systematically to minimize such risk. Many countries have established regulations to control levels of AFB1 in feeds and maximum permissible levels of AFM1 in milk and cheese to reduce this risk (Sarimehmetoglu et al., 2004; Mahdiyeh et al., 2013). In the United States and Brazil (as well as in Codex) an action level for AFM1 in fluid milk has been set at 500 ng/L (Shundo & Sabino, 2006; Codex Standard, 2008; Motawee et al., 2009) while the European Union (Commission Regulation, EC, 2006) has established a lower maximum allowable level for AFM1 in milk of 50 ng/L and 250 ng/kg for cheese. Many other countries have followed the European Union standards (Dashti et al., 2009, Kamkar et al., 2011). In Egypt, the ministry of health established in 1990 that fluid milk and dairy products should be free from AFM1 and currently the maximum permissible levels follow the European Union standard (Egyptian Standard, 2007).
So, for any country (including Egypt) any increase in the proportion of AFM1 in milk and dairy products above the permissible limit of Codex and other countries can affect international trade of such milk products in global markets. Many studies have reported the occurrence of high levels of AFM1 in milk and cheese in many countries that exceeded these maximum allowed limits (Cirilli & Cirilli, 1988; Oruc & Sonal, 2001; Motawee, 2003; Motawee et al., 2004, 2009; Tekinsen & Tekinsen, 2005; Yapar, et al., 2008; Atasever, et al., 2010; Tsakiris, et al., 2013).
Domiati cheese is the most popular soft white pickled cheese in Egypt (accounting for 75% of cheeses produced and consumed in Egypt). It differs chiefly from other pickled cheese varieties, such as feta, Brinza, or Telema cheese, in that the milk is salted (from 5% to 14%) before renneting depending on season and cheese ripening temperature (Abou-Donia, 1986; ElBaradei et al., 2007). Domiati cheese can be made from either cow or buffalo whole milk or their mixture. The salted milk can be curdled fresh of sometimes after pasteurization. No starter culture is added. It can be consumed fresh but more often after pickling in salted whey or a brine solution for up to 2 to 4 months (Zhang et al., 2003). The objective of the present study was to determine the effect of milk pasteurization followed by manufacture and pickling of Domiati cheese on AFM1 levels in milk and cheese.
Materials and Methods
Cheese Making and Sampling
Domiati cheese was made (in duplicate) with some modifications according to Abou-Donia (1986) from 24 kg of cows milk (4% fat, 8% solids not fat). Milk was divided into two 12-kg batches that were spiked with 1.0 µg/kg of AFM1 (Sigma chemical Co. Deisenhofen, Germany) and dissolved in milk, then pasteurized at 63°C for 30 minutes and cooled to 34±1°C. Then 0.8 g CaCl2 was added to each batch to ensure good coagulation and curd formation and then each batch was divided into three 4-kg portions. To each portion of milk was then added 6%, 8% or 10% (wt./wt.) of NaCl, respectively. Then sufficient (1, 1.25 or 1.5 ml, respectively) standard strength calf rennet (Chr. Hansen’s. Inc. Denmark) was added to produce a firm curd in 2 to 3 h at 34°C. The coagulum was then ladled out into 4-L steel molds lined with coarse cloth and the molds turned twice (after ~ 1 to 2 hour) and then allowed to stand 18 to 20 h to allow for whey drainage. The cheeses were removed from their molds and placed in small tin containers that were then filled with the respective cheese whey to exclude air and allow for pickling of the cheese. The containers were closed and stored at 20°C for 3 months.
Determination of AFM1
Milk was sampled after spiking with AFM1 and then after pasteurization. Cheese as sampled after curd formation, then before placing cheese in the pickling tins (day 0) and then every 15 d. Whey samples were collected at the same times. Each sample (milk, curd, whey, cheese) was stored at 4°C and analyzed for AFM1 content within 12 h. Detection of AFM1 was by an enzymelinked immunoassay test kit (RIDASCREEN, R-Biopharm GmbH, Darmstadt, Germany) according to manufacturer’s instructions (Anonymous, 1999) as briefly described below. The test kit included microtiter plates with immobilized AFM1-antibody, AFM1-enzyme conjugate, enzyme substrate (urea peroxide) chromogen (tetramethyl benzidine), and stop reagent 1M H2SO4. Other chemicals used included reagent grade methanol, n-heptane and dichloromethane (Merck, Darmstadt, Germany), and phosphate buffer saline at pH 7.2 prepared by mixing 0.55 g NaH2PO4.H2O with 2.85 g of Na2HPO4.2H2O and 9 g NaCl and then filled up to 1000 ml with distilled water. Pure AFM1 from SIGMA (Deisenhofen, Germany) was used as a standard.
For milk and whey: Milk and whey samples (4 ml) were chilled to 4°C, centrifuged for 10 min at 3500 rpm, (Heraeus Megafuge 1.0, Thermo: Fisher Scientific, Inc., Waltham, MA, USA), and then the upper cream layer was completely removed by aspiration through a Pasteur pipette. The aqueous layer was then diluted 20 times (vol./vol. ) with deionized water, then 100 µl placed into a microtiter plate sample well.
For cheese and curd: Curd and cheese samples (10 g) were coarsely ground and thoroughly mixed, without the addition of liquid using (Ultraturrax, IKA- Werke, Staufen, Germany) and then 2.000 +0.005g weighed into a centrifugal glass vial and 40 ml of dichloromethane was added and extracted by stirring/shaking the vial for 15 min. then the suspension was filtered and 10 ml of the extract was evaporated at 60°C under a weak nitrogen stream. The oily residue was redissolved in 0.5 ml methanol, 0.5 ml PBS buffer and 1 ml heptane and mixed thoroughly. After centrifugation for 15 min at 2700 g, the upper heptane-layer was completely removed. An aliquot of the lower methanolic-aqueous phase was carefully poured off using a Pasteur pipette. One hundred microliters of this aliquot was brought up to a 10% methanol content by addition of 400 µl Ridascreen buffer 1 and 100 µl was used per well in the test. In order to obtain sample AFM1 concentration in ng/L, the concentration read from the calibration curve was further multiplied by a dilution factor 1 for milk and 10 for curd and cheese. Therefore, the mean detection limit for AFM1 in milk and whey was 5 ng/L and in curd and cheese was 50 ng/kg.
ELISA test and standard curve procedure: AFM1 standard solution was prepared containing (50, 100, 200, 400, and 800 ng AFM1/L) for making a calibration curve. Samples (50 µl) in microtiter plate wells (in duplicate) were incubated for 60 min at room temperature in the dark, to allow antibody binding sites in the wells to be occupied proportionally to AFM1 concentration. The liquid was then removed completely from the wells, which were washed twice with 250 µl of washing buffer and distilled water. In the next step, any remaining free binding sites were occupied by adding 100 µl of enzyme conjugate to the microtiter plate wells and incubated for another 60 min at room temperature (20 to 25°C) in the dark. Any unbound enzyme conjugate was then removed in a washing step. This was followed with 50µl of urea peroxide and 50 µl of tetramethylbenzidine and 30-min incubation at room temperature in the dark. , which then turned yellow on addition of 100 µl of the stop reagent. Yellow color was measured at 450 nm in ELISA reader (ELX-808, Winooski, VT, Inc., USA) against an air blank within 60 min, with AFM1 concentration being inversely proportional to A450.
Statistical Analysis
All the data were treated statistically using SAS (1996). To determine the effect of pasteurization, salting and storage treatments, a General Linear Model (GLM) was used with the equation: Yij = µ + αj + εij; where Yij is the AFM1 level, µ the general mean, αj is the salting or storage effect, and εij is the residual error. The mean and standard error (SE) were used to express the results of the composition and AFM1 levels.
Results and Discussion
Pasteurization of milk at 63 ºC for 30 min reduced AFM1 content by ≤ 10% (Table 1). This is in agreement with El-Deeb et al., (1992) who reported a 9.5% drop in AFM1. Mashaley et al., (1986) reported a 5.2 to 9.4% decrease in AFM1 and AFM2 in milk spiked with 5 and 10 µg/Kg and observed that the decrease during pasteurization was inversely proportional to the amount of toxin added. Similarly, Motawee & McMahon (2009) reported that pasteurization of milk caused ≤10% destruction of AFM1 during Feta cheese making. Deveci (2007) investigated milk pasteurization at 72 ºC for 2 min and reported losses of AFM1 of 12% and 9% in milk contaminated with 1.5 µg/Kg and 3.5 µg/L of AFM1 respectively. An earlier study by Kiermeier & Mashaley, (1977) had reported 12% losses of AFM1 in pasteurized milk at 75ºC for 40 sec. Thus, as shown through a number of studies, AFM1 is relatively resistant to heat treatments such as pasteurization (Van Egmond et al., 1977; Wiseman & Marth 1983; Govaris et al., 2002; Oruc et al., 2006; Anfossi et al., 2012). When this pasteurized milk was converted into Domiati cheese, there was a partitioning of AFM1 between curd and whey that was dependent on the level of salt added to the milk. For cheese made from milks containing 6%, 8% or 10% salt, the levels of AFM1 in the cheese curd was 2.73, 2.52 and 2.31 µg/kg, respectively. This increased concentration comes about as the casein and fat are concentrated as the cheese is made, while the level of AFM1 in the corresponding whey was 0.36. 0.38 and 0.40 µg/kg, respectively. Thus there was a mean transfer of 60%, 58% and 56% of AFM1 in the original milk into the cheese curd and 30%, 32% and 34% of AFM1 was transferred into the whey with salt additions to milk of 6%, 8% and 10%, respectively. Our results are in agreement with other previous studies (Abd-Allalh, 1983; Yousef & Marth, 1989; Dragacci & Fremy, 1996; Motawee, 2003; Motawee & McMahon, 2009; Rubio et al., 2011).
Even though ~85% of the milk ends up as whey, a concentration of AFM1 in cheese curd occurs because AFM1 has an affinity to casein protein fraction in milk and it is more soluble in water than in oil. Levels of AFM1 in Domiati cheese that are almost 3 times higher than in the milk from which the cheeses were made is in agreement with other studies on soft cheeses (Yousef & Marth 1989; Govaris et al., 2001; Prandini et al., 2009). Although, there have been other studies that have shown differing distributions of AFM1 in milk between curd and whey. Some of these differences occur when there are different levels of AFM1 in the milk. Some authors reported that half or more of the AFM1 transfers into the whey: 50%, 61%, 66%, 86%, and 100% according to Stubblefield and Shannon, 1974; Wiseman and Marth, 1983; Blanco et al., 1988; Stoloff et al.,1981; and Purchase et al., 1972, respectively. In contrast, others have reported that most of AFM1 transfers into the curd at levels of 66%, to 72%, 73% to 77%, 80%, and 100% according to Mashaley et al., 1986; El-Deeb et al., 1992; McKinney et al., 1973; and Blanco, et al., 1988, respectively. In a non-rennet cheese such as Ricotta cheese, most of the AFM1 (~94%) goes into the hot whey when as the whey proteins precipitate (Cattaneo et al., 2013). These varying transfer rates can be ascribed to factors such type and degree of milk contamination, differences in milk quality, presence of curd fines in the whey, the cheese manufacture process, as well as experimental AFM1 measurement techniques such as extraction method, methodology, and expression of the results.
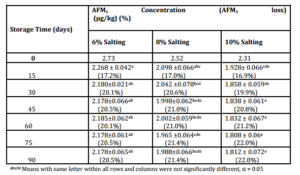
When considered on a serving size (60 g of cheese compared to 250 ml of liquid milk) there is considerably less dietary exposure to AFM1 when consuming Domiati cheese that has been pickled for 3 mo rather than milk. A serving of milk containing 500 ng/kg of AFM1 (US allowable limit) would give an exposure of 125 ng AFM1 while a serving of pickled Domiati cheese made from the same milk (pasteurized) would only give a exposure to ~45 ng AFM1. Even consuming the cheese fresh after only 2 wk storage at 20 ºC, would still give a dose of <50 ng AFM1 depending on the salting level used during cheese manufacture compared to their levels after manufacture. With continued storage through 90 d the losses of AFM1 were significantly different (P < 0.05) with reduction in AFM1. Continued slight transfer of AFM1 from curd into whey during 90 days of storage is in agreement with others (Brackett and Marth, 1982b; Mashally et al., 1986; Fremy et al., 1990; Dragacci et al., 1995; Govaris et al., 2001; Motawee, 2003; and Motawee & McMahon, 2009). In contrast, the levels of AFM1 during cheese ripening and storage of cheeses that are not stored in whey vary, as reported for Cheddar cheese (Brackett and Marth, 1982c), Brick and Limburger cheeses (Brackett et al 1982), Camembert and Tilsit cheeses (Kiermeir & Buchner, 1977) in which an increase in AFM1 during the early stage of ripening was observed with decreases thereafter for cheeses prepared from naturally contaminated milk. While in Gouda (Van Egmond et al., 1977) or Mozzarella (Brackett & Marth, 1982b) cheeses there was no appreciable change during ripening for 6 or 4 months, respectively. These various results may be due to several factors such as the type of cheese, and as suggested (Brackett & Marth, 1982a) proteolysis during cheese ripening may release the toxin, or high lipolytic action during ripening of cheeses such as Teleme and release of free fatty acids may enhance release of AFM1 from its hydrophobic bonds to casein (Brackett et al., 1982).
Table ( 1) – Aflatoxin M1 (AFM1) content and concentration in milk spiked with 1.00 µg AFM1 per kg and its partitioning between curd and its whey made using 4 kg of milk with 6%, 8% and 10% salt added to milk prior to manufacture of Domiati cheese.
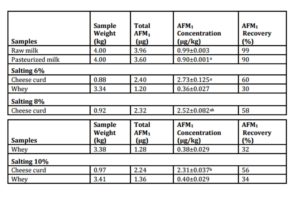
Table (2) – Concentration of aflatoxin M1 (AFM1) in Domiati cheese made from 4 kg milk spiked with 1.00 µg AFM1 per kg prior to pasteurization, and with salt additions to the milk of 6%, 8% and 10%, and subsequent during storage at 20oC in its whey, with percent loss of AFM1 shown in parentheses.
Conclusions
AFM1 levels in milk, Domiati cheese and whey were determined using an ELISA test kit. Pasteurization of milk caused ≤10% loss of AFM1. About 60%, 58%, and 56% of total AFM1 remained in cheese curd made using 6%, 8% and 10% salt respectively after manufacture directly. When considered on a serving size (60 g of cheese compared to 250 ml of liquid milk) there is considerably less dietary exposure to AFM1 when consuming Domiati cheese that has been pickled for 3 mo. rather than milk. A serving of milk containing 500 ng/kg of AFM1 (US maximum allowance) would give an exposure of 125 ng AFM1 while aserving of pickled Domiati cheee made from the same milk (pasteurized) would only give a exposure to ~45 ng AFM1. Even consuming the cheese fresh after only 2 wk storage at 20 ºC, would still give a dose of <50 ng AFM1 depending on the salting level used during cheese manufacture compared to their levels after manufacture. With continued storage through 90 d the losses of AFM1 were significantly different (P < 0.05) with reduction in AFM1.
Acknowledgements
The author thank Prof. Dr. Donald McMahon Professor of Dairy Chemistry and Processing Director of Western Dairy Center (Utah State University, Logan USA) and Prof. Dr .Mohamed Shalappy – Head of Dairy Department (Mansoura UniversityEgypt) for their cooperation. The author also, would like to thank Dr. Omar Farid (NODCAR) Egypt, for performing statistical analysis.
References
1.Abd-Allah, E. A. M., (1983). “Effect of milk processing on aflatoxin M1 content” Msc in Dairy Science, Faculty of Agriculture. Cairo University – Egypt.
2.Abou-Donia, S. A. (1986). “Egyptian Domiati Soft White Pickled Cheese” Review- New Zealand Journal of Dairy Science and Technology, 21 167-190.
3.Anfossi, L., Baggiani, C., Giovannoli, C., D’Arco, G., Passini, C. & Giraudi, G. (2012). “Occurrence of aflatoxin M1 in Italian cheese: Results of a survey conducted in 2010 and correlation with manufacturing, production season, milking animals,, and maturation of cheese” Food Control, 25 125-130.
4.Anonymous, (1999). “Enzyme Immunoassay for the Quantitative Analysis of Aflatoxin M1” Art. No R 1101. R-Biopharm GmbH, Darmstadt, Germany.
5.Atasever, M. A., Adiguzel, G., Atasever, M., & Ozturan, K., (2010). “Determination of Aflatoxin M1 Levels in Some Cheese Types Consumed in Erzurum- Turkey” Kafkas Universitesi Veteriner Fakultesi Dergisi, 16 87-91.
6.Ayoub, M. M, Azza Mahmoud, K. S. & Amal, A. R. (2011). “Evaluation of aflatoxin M1 in raw, processed milk and some milk products in Cairo with special reference to its recovery. Researcher” 2011; 3 (9).
7.Battacone, G., Nudda, A., Palomba, M., Pascale, M., Nicolussi, P. & Pulina, G. (2005). “Transfer of aflatoxin B1 from feed to milk andfrom milk to curd and whey in dairy sheep fed artificially artificially Contaminated concentrates” Journal of Dairy Science 88 3063-3069.
8. Blanco, J. L, Domingues, L., Gomez-Lucia, E., Garayzabal, J. F., Goyache, J. & Suarez, G. (1988). “Behaviour of aflatoxin M1 during the manufacture, ripening and storage of Manchego-type cheese” Journal of Food Protection, 53 1373- 1376.
9.Brackett, R. E. & Marth, E. H. (1982a). “Association of aflatoxin M1 with casein” Zeitschrift fur Lebensmitteluntersuchung und Furschung, 174 439-441.
10.Brackett, R. E., & Marth, E. H. (1982b). “Fate of aflatoxin M1 in Parmesan and Mozozzarella cheese” Journal of Food Protection, 45 597-600.
11.Brackett, R. E. & Marth, E. H. (1982c). “Fate of aflatoxin M1 in cheddar cheese and in process cheese spread” Journal of Food Protection, 45 549- 552.
12.Brackett, R. E., Applebaum, R. S., Wiseman, D. W. & Marth, E. H. (1982). “Fate of aflatoxin M1 in Brick and Limburger- like cheese” Journal of Food Protection, 45 553-556.
13.Cattaneo, T. M. P., Marinoni, L., Iametti, S. & Monti, L. (2013). “Behavior of Aflatoxin M1 in dairy wastes subjected to different technological treatments: Ricotta cheese production, ultrafiltration and spray-drying” Food Control, 32 77-82.
14.Codex Standard. 193 (2008). Codex general Standard for contaminations and toxins in foods. Codex Stan. 193 (1995), Review. 3 (2008).
15.Commission Regulation (EC) No 1881. (2006). Setting maximum levels for certain Contaminations in foodstuffs. Official Journal of the European Union L 364, pp. 5-24. 19 December, 2006.
16.Cirilli, G., Aldana, G. & Cirilli, G. S. (1988). “Contamination of Dairy products by hydroxyl-aflatoxin” Microbiologie, Aliments, Nutrition, 6 217-219.
17.Dashti, B., Al-Hamli, S., Alomirah, H., Al-zenki, S., BuAbbas, A. & Sawaya, W. (2009). “Level of aflatoxin M1 in milk, cheese consumed in Kuwait and occurrence of total aflatoxin in local and imported animal feed” Food Control, 20 (7) 686-690.
18.Deshpande, S. S. (2002). Fungal toxins. In Handbook of Food Toxicology, (S. S. Deshpande, ed.) pp. 387-356. Marcel Decker, New York.
19.Deveci, O. (2007). “Change in the concentration of aflatoxin M1 during manufacture and storage of White Pickled cheese” Food Control, 18 1103- 1107.
20.Dragacci, S. & Fremy, J. M. (1996). “Application of immunoaffinity column cleanup to aflatoxin M1 determination and survey in cheese” Journal of Food Protection, 59 1011-1013.
21.Dragacci, S., Gleizes, E., Fremy, J. M. & Candlish, A. G. (1995). “Use immunoaffinity chromatography as a purification step for the determination of aflatoxin M1 in cheeses” Journal of Food Additives and Contaminants, 12 59-65.
22.Egyptian Standard 1- 1875 (2007) “Maximum levels of mycotoxin for foods and feeds, part-1: Aflatoxins” Egyptian Organization for Standardization and Quality.
23.El-Baradei, G., Delacroix-Buchet, A. & Ogier, C. (2007). “Biodiversity of Bacterial Ecosystems in Traditional Egyptian Domiati Cheese” Applied. Environment and Microbiology, p. 1248- 1255.
24.El-Deeb, S. A., Zaki,N., Shoukry, Y. M. R., & Kheadr E. E. (1992). “Effect of some technological processes on stability and distribution of Aflatoxin M1 in milk” Egyptian Journal of Food Science, 20 29- 42.
25.Fallah, A. (2010). “Assessment of aflatoxin M1 contamination in pasteurized and UHT milk marketed in central part of Iran” Food and Chemical Toxicology, 48 988-991.
26.Fermy, J. M., Roiland, J. C., & Gaymard, D. (1990). “Behavior of 14C aflatoxin M1 during Camembert cheese making” Journal of Environmental Pathology, Toxicology and Ecology, 10 95-98.
27.Ghazani, M. H. (2009). “Aflatoxin M1 contamination in pasteurized milk in Tabriz (northwest of Iran)” Food and Chemical Toxicolology, 47 1624-1625.
28.Govaris, A, Roussi, P. A, Koidis & Botsoglou, N. A. (2001). “Distribution and Stability of aflatoxin M1 during processing, Ripeneng and storage of Telemes cheese” Food Additives and Contaminants, 18 (5) 437-443.
29.Govaris, A., Roussi, V., Koidis PA. & Botsoglou, NA. (2002). “Distribution and stability of aflatoxin M1 during production and storage of yoghurt” Food Additives and Contaminants, 19(11) 1043-1050.
30.Guo, Y., Yuan, Y. & Yue, T. (2013). “Aflatoxin M1 in milk products in China and Dietary Risk Assessment” Journal of Food Protection, 76 (5) 849-853.
31.Hampikyan, H., Bingol, E. B., Cetin, O. & Colack, H. (2010). “Determination of Aflatoxin M1 level in Turkish white, Kashar and tulum cheeses” Journal of Food Agriculture & Environmental Science, 8 (1) 13-15.
32.Kamkar, A., Jahed Khaniki, Gh.R., and Alavi, S. A. (2011). “Occurrence of Aflatoxin M1 in Raw Milk Produced in Ardabil of Iran. Iran” Journal of Environmental Health Science & Engineering, 8 (2) 123-128.
33.Karimi, G., Hassanzadeh, M., Teimuri, M., Nazari, F. & Nili, A. (2007). “Aflatoxin M1 contamination in pasteurized milk in Mashhad, Iran” Iranian Journal of Pharmacology Science, 3 153-156.
34.Kiermeier, F., & Buchner, M. (1977). “On the aflatoxin M1 content of cheese during ripening and storage” Zeitschrift fur Lebensmitteluntersuchung und Furschung, 164 87-91.
35.Kiermeier, F. & Mashaley, R. (1977). “Einfluss der molkerei technischen behandlung der rohmich auf des aflatoxin M1 gehalt der daraus hergestellen produkte” Zeitschrift fur Lebensmitteluntersuchung und Furschung, 164 183-187.
36.Mahdiyeh L. R, Reza, K. D, Mahya, M, Masoud, H. K, Khosro, I. & Mortiza, A.A. (2013). “Determination of Aflatoxin M1 Level in Raw Milk Samples Gilan, Iran” Advanced Studies in Biology, 5 (4) 151- 156.
37.Maktabi, S., Hajikolaie, M. R., Ghorbanpour, M. & Pourmehdi, M. (2011). “Determination of Aflatoxin M1 in UHT, pasteurized and GSM Milks in Ahvaz (South-west of Iran) Using ELISA” Global Veterinaria, 7 (1) 31-34.
38.Maktabi, S., & Fazlara, A. (2011). Contamination of Ice cream by Aflatoxin M1 in Iran. Amercan-Eurasian Journal of Toxicology Science. 3 (3) 120-123.
39.Mashally., RI, El-Deeb., SA. & Safwat. NM. (1986). “Distribution and stability of AFM1 during processing and storage of Karish cheese”Alexandria Journal of Agriculture- Research, 31(3) 219-228.
40.McKinney, J. D., Cavanau, G. C., Bell, J.T., Hoversland, A. S., Nelson, D. M., Pearson, J. & Selkirk, R. J. (1973). “Effects of ammoniation on aflatoxins in rations fed lactating cows” Journal of American Society, 50 79-84.
41.Mohamadi, H. & Alizadeh, M. (2010). “A study of the Occurrence of Aflatoxin M1 in Dairy Products Markted in Urmia, Iran” Journal of Agriculture Science and Technology, 12 579-583.
42.Mohamadi Sani, A., Khezri, M. & Moradnia, H. (2012). “Determination of Aflatoxin M1 in Milk by ELISA Technique in Mashad (Northeast of Iran)” International Scholarly Research Network ISRN Toxicology. Vol.2012, Article ID 121926. p 1-4.
43.Motawee, M. M., (2003). Detection, Determination and Detoxification of some Mycotoxins in Some Dairy Products. Ph.D thesis Mansoura, Univwesity-Egypt. 2003.
44.Motawee, M. M., & McMahon, D. J. (2009). “Fate of Aflatoxin M1 during Manufacture and Storage of Feta Cheese” Journal of Food Science, 74 (5) T42- 45.
45.Motawee, M. M., Bauer, J. & McMahon, D. J. (2009). “Survey of Aflatoxin M1 in Cow, Goat, Buffalo and Camel Milks in Ismailia- Egypt” Bulletin of Environmental Contamination and Toxicology, 83 (5) 766-769.
46.Motawee, M., Meyer, K. & Bauer, J. (2004). “Incidence of aflatoxins M1 and B1 in raw milk and some dairy products in Damietta, Egypt” Journal of Agriculture Science, Mansoura -Egypt , 29 711-718.
47.Nilchian, Z. & Rahimi, E. (2012). Aflatoxin M1 in Yoghurts, Cheese and Ice —Cream in Shahrekord- Iran. Journal of World Applied Science. 19 (5) 621-624.
48.Oveisi, M. R., Jannat, B., Sadeghi, N., Hajimahmoodi, M. & Nikzad, A. (2007). “Presence of aflatoxin M1 in milk and infant milk products in Tehran, Iran” Food Control, 18 1216-1218.
49.Oruc, H. H, Cibik, R, Yilmaz, E. & Kalkanli, O. (2006). “Distribution and stability of aflatoxin M1 during processing and ripening of traditional white pickled cheese” Food Additives and Contaminants, 23 (2) 190-195.
50.Oruc, H. H., & Sonal, S. (2001). “Determination of aflatoxin M1 level in cheese and milk consumed in Bursa, Turkey” Veterinary & Human Toxicology, 43 (5) 292-293.
51.Purchase, I. F. H., Steyn, M., Rinsma, R. & Tustin, R. C. (1972). “Reduction of aflatoxin M1 in milk by processing” Food Cosmetics Toxicology, 10 383-387.
52.Prandini, A., Tansini, G., Sigolo, S., Filippi, L., Laporta, M. & Piva, G. (2009). “Review: on the occurrence of aflatoxin M1 in milk and dairy products” Food and Chemical Toxicology, 47 984-991.
53.Rubio, R., Moya, V. J., Berruga, M. I., Molina, M. P. & Molina, A. (2011). “Aflatoxin M1 in the intermediate dairy products from Manchego cheese production: distribution and stability” Mljekarstvo, 61(4) 283-290.
54.Sarimehmetoglu, B., Kuplulu, O. & Celik, T. H. (2004). “Detection of aflatoxin M1 in cheese samples by ELISA” Food Control, 15 45-49.
55.SAS., (1996). “Statistical analysis systems users guide” (version 7), SAS institute Inc., N.C., USA.
56.Stoloff, L., Wood, G & Carter, G. (1981). “Aflatoxin M1 in manufactured dairy products” Produced in the United States in 1979. Journal of Dairy Science, 64 2426-2430.
57.Stubblefield, R. D. & Shannon, G. M. (1974). “Aflatoxin M1: Analysis in dairy products and distribution in dairy foods made from artificially contaminated milk” Association of Analytical Chemists, 57 847-851.
58.Sepehr, Sh., Amin, J., Masoomeh, Gh. & Sahab, Sh. (2012). “Detection and Occurrence of Aflatoxin M1 level in milk and Milk White Cheese Produce in Esfahan State Iran” Research Journal of Biological Science, 7 (5) 225-229.
59.Shundo, L. & Sabino, M. (2006). “Aflatoxin M1 in milk by immunoaffinity column Cleanup with TLC/HPLC determination” Brazilian Journal of Microbiology, 37 164-167.
60.Tekinsen, K. K. & Tekinsen, O. C. (2005). “Aflatoxin M1 in white pickle and Van otlu (herb) cheeses consumed in southeastern Turkey” Food Control, 16 565-568.
61.Tsakiris, I. N., Tzatzarakis, M. N., Alegakis, A. K., Vlachou, M. I., Renieri, E. A. & Tsatsakis, A.M. (2013). “Risk assessment scenarios of children’s exposure to aflatoxin M1 residues in different milk type from the Greek market” Food & Chemical Toxicology, 56 261-265. 61.Van Egmond, H. P., editor, (1989). Aflatoxin M1: occurrence, toxicity, regulation London; Elsevier. Mycotoxins in dairy products; P.11-15.
62.Van Egmond, H. P, Paulsch, H. A. Veringa & Schuller, P. L. (1977). “Effect of processing on the aflatoxin M1 content of milk and milk products. Extract des” Archives de L ‘institute Pasteur de Tunis, 3-4, 381-390.
63.Wiseman, D. W. & Marth,E. H. (1983). “Behavior of aflatoxin M1 during manufacture and Storage of Quesblanco and bakers’ cheese” Journal of Food Protection, 46 910-913.
64.Yapar, K., Elmali, M, Kart, A. & Yaman, H. (2008). “Aflatoxin M1 level in different type of cheese products produced in Turkey” Medycyna Weterynaryjna, 64 53-55.
65.Yousef, A. E. & Marth, E.H. (1989). “Stability and degradation of Afaltoxin M1. In Mycotoxins in Dairy products” (H. P. Van Egmond, ed.), pp. 127- 161.Elsevier Applied Science, London.
66.Zhang, X., Kilmer, R. L. & Muhammah, A. (2003). A descriptive Analysis of Egypt and Saudi Arabia who Import United States Dairy Products, monograph MGTC 03-8. International Agricultural Trade and Policy Center, Institute of Food and Agricultural Science. University of Florida, Gaines-ville, FL.
67.Zinedine, A. & Manes, J. (2009). “Occurrence and legislation of mycotoxins in food and feed from Morocco” Food Control, 20 334-344.





